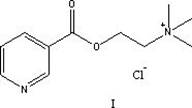
Revolutionizing Pyridine Choline Ester Production via Novel One-Pot Catalytic Strategy
The pharmaceutical and fine chemical industries are constantly seeking process intensification strategies that can reduce operational complexity while maintaining rigorous quality standards. A significant breakthrough in this domain is documented in Chinese Patent CN115872928A, which discloses a highly efficient one-pot method for the preparation of pyridine chloride 3-choline formate, a critical intermediate known for its vasodilatory properties and potential applications in hair loss treatment. This innovative approach fundamentally reimagines the synthetic pathway by utilizing 2-chloroethanol not merely as a passive solvent, but as a dual-function reagent that participates directly in the molecular construction. By integrating the esterification and quaternization steps into a single operational vessel, the technology effectively bypasses the cumbersome isolation procedures that have historically plagued the production of this valuable choline ester derivative. The strategic design of this reaction system offers a compelling value proposition for manufacturers aiming to streamline their supply chains and enhance overall process economics without compromising on the stringent purity profiles required for pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyridine chloride 3-choline formate has been hindered by convoluted multi-step protocols that introduce significant inefficiencies and cost burdens into the manufacturing workflow. Prior art, such as the methodology described in US Patent 3,337,569A and subsequent Chinese patents like CN107540605A, typically relies on a sequential approach where the nicotinic acid moiety is first esterified, often requiring conversion to an iodo-intermediate using sodium iodide before undergoing quaternization with trimethylamine. A particularly prohibitive aspect of these legacy routes is the final anion exchange step, which frequently necessitates the use of expensive silver chloride (AgCl) to replace the iodide counterion with the desired chloride, creating a substantial economic bottleneck due to the high cost of precious metal salts. Furthermore, these traditional pathways demand multiple solvent switches, extensive extraction procedures using dichloromethane, and repeated concentration steps, all of which accumulate operational time, increase waste generation, and elevate the risk of product degradation or impurity formation during prolonged handling.
The Novel Approach
In stark contrast to the fragmented nature of conventional synthesis, the methodology outlined in CN115872928A introduces a streamlined one-pot cascade reaction that elegantly resolves the aforementioned logistical and economic challenges. This novel approach leverages the unique reactivity of 3-pyridinecarbonyl chloride and trimethylamine directly within a 2-chloroethanol medium, allowing the esterification and subsequent quaternization to proceed concurrently or sequentially within the same reaction vessel without the need for intermediate isolation. By eliminating the requirement for iodine intermediates and the costly silver salt exchange, the process drastically simplifies the material flow and reduces the dependency on high-value reagents that offer no structural contribution to the final molecule. The result is a robust synthetic protocol that not only accelerates the production timeline but also inherently improves the environmental footprint by minimizing solvent consumption and waste generation, making it an exceptionally attractive option for industrial-scale manufacturing of high-purity pharmaceutical intermediates.
Mechanistic Insights into One-Pot Esterification-Quaternization
The core mechanistic advantage of this technology lies in the dual functionality of 2-chloroethanol, which serves simultaneously as the nucleophilic reactant for ester formation and the polar protic solvent that facilitates the subsequent quaternization of the amine. In the initial phase of the reaction, the hydroxyl group of the 2-chloroethanol attacks the electrophilic carbonyl carbon of the 3-pyridinecarbonyl chloride, displacing the chloride leaving group to form the ester linkage. As the reaction progresses, typically under mild heating conditions between 40°C and 60°C, the tertiary amine (trimethylamine) present in the system engages in a nucleophilic substitution reaction with the terminal chloro-group of the newly formed ester chain. This intramolecular or pseudo-intramolecular environment promotes the formation of the quaternary ammonium cation, effectively locking the choline structure onto the pyridine ring in a single continuous operation. The presence of a basic catalyst, such as sodium hydroxide or potassium hydroxide, can further accelerate this transformation by neutralizing the hydrochloric acid byproduct generated during the acylation step, thereby driving the equilibrium towards the desired product and preventing acid-catalyzed decomposition of sensitive intermediates.
From an impurity control perspective, the one-pot nature of this synthesis offers distinct advantages by minimizing the exposure of reactive intermediates to external environments where side reactions could occur. Traditional multi-step processes often suffer from the accumulation of byproducts during isolation and purification stages, such as hydrolysis of the acid chloride or degradation of the iodo-intermediate, which can be difficult to remove in the final steps. In this optimized route, the reaction conditions are tightly controlled within a closed system, and the final product precipitates directly from the reaction mixture upon cooling, acting as a self-purifying mechanism that excludes soluble impurities and unreacted starting materials from the crystal lattice. This crystallization-driven purification ensures that the resulting solid possesses high structural integrity and purity, often exceeding 97% HPLC purity without the need for complex chromatographic separations, thus providing R&D teams with a reliable pathway to generate reference standards and clinical trial materials with consistent quality attributes.
How to Synthesize Pyridine Choline Ester Efficiently
The implementation of this advanced synthetic route requires precise control over reaction parameters to maximize yield and ensure reproducibility at scale. The process begins with the charging of 2-chloroethanol into a reactor, followed by the addition of 3-pyridinecarbonyl chloride and a solution of trimethylamine, establishing the necessary stoichiometric balance for the cascade reaction. Maintaining the temperature within the optimal range of 50°C to 55°C is critical to balancing reaction kinetics with thermal stability, while the reaction time is typically monitored over a period of 4 to 8 hours to ensure complete conversion of the starting materials. Upon completion, the reaction mass is cooled to induce crystallization, allowing the target compound to precipitate as a solid which can be easily recovered via filtration, representing a significant departure from the liquid-liquid extraction methods of the past.
- Charge 2-chloroethanol into a reactor and add 3-pyridinecarbonyl chloride along with a trimethylamine solution.
- Heat the mixture to 40-60°C and maintain reaction for 4-8 hours to facilitate simultaneous esterification and quaternization.
- Cool the reaction mass to 20-30°C to induce crystallization, then filter and dry the solid precipitate to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthesis technology translates into tangible strategic benefits that extend far beyond simple laboratory efficiency. The most immediate impact is observed in the drastic reduction of raw material costs, driven primarily by the complete elimination of silver chloride and sodium iodide from the bill of materials. Silver salts represent a significant capital expenditure in traditional pharmaceutical manufacturing, and their removal from the process flow liberates working capital and reduces exposure to the volatility of precious metal markets. Additionally, the simplification of the workflow from a multi-step sequence involving extractions and solvent swaps to a single reaction-followed-by-filtration operation significantly lowers the consumption of organic solvents like dichloromethane and THF, contributing to substantial cost reductions in waste disposal and solvent recovery operations.
- Cost Reduction in Manufacturing: The economic model of this new process is fundamentally superior due to the removal of high-cost reagents and the consolidation of unit operations. By avoiding the use of silver-based anion exchange resins or salts, manufacturers can achieve a leaner cost structure that is less susceptible to fluctuations in the price of specialty chemicals. Furthermore, the high yield profile, consistently demonstrated in the range of 90% to 96% in experimental examples, ensures that raw material utilization is maximized, minimizing the loss of valuable pyridine derivatives and reducing the overall cost per kilogram of the active intermediate produced.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the shortened cycle time inherent in a one-pot process allows for faster turnover of production batches, enabling suppliers to respond more agilely to market demand fluctuations. The reduced complexity of the operation also lowers the risk of batch failures caused by human error during multiple transfer and isolation steps, thereby enhancing the reliability of supply for downstream customers who depend on consistent availability of this key intermediate for their own formulation pipelines. The ability to produce high-purity material through simple filtration rather than complex chromatography also reduces the dependency on specialized equipment, broadening the base of capable contract manufacturing organizations.
- Scalability and Environmental Compliance: The scalability of this process is exceptional, as the reaction conditions are mild and do not require extreme pressures or temperatures that would necessitate specialized high-pressure reactors. The reduction in solvent usage and the elimination of heavy metal waste streams (such as silver residues) align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. This compliance advantage mitigates regulatory risk for manufacturers and ensures long-term sustainability of the supply chain, making it a future-proof solution for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, drawing directly from the experimental data and process descriptions provided in the patent literature. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this technology for their specific production needs and for procurement professionals assessing the long-term value proposition of suppliers utilizing this method.
Q: How does the new one-pot method improve upon traditional silver salt exchange processes?
A: The novel method eliminates the need for expensive silver chloride (AgCl) anion exchange and iodine intermediates, replacing a multi-step isolation process with a direct filtration, thereby drastically cutting raw material costs and processing time.
Q: What are the typical purity and yield specifications achievable with this technology?
A: Experimental data from the patent indicates that this optimized route consistently achieves HPLC purity levels exceeding 97.7% with isolated yields ranging between 90% and 96%, demonstrating superior efficiency over conventional multi-step routes.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the process is highly scalable due to its one-pot nature which minimizes unit operations, reduces solvent switching requirements, and simplifies downstream processing to a basic cooling and filtration step suitable for large-scale reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine Choline Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising efficiencies of the one-pot synthesis described in CN115872928A can be fully realized in an industrial setting. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify identity, assay, and impurity profiles for every batch released.
We invite global partners to engage with our technical procurement team to discuss how this optimized manufacturing route can drive value for your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this streamlined process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the quality and cost-efficiency of your supply chain for vital therapeutic intermediates.
