Advanced Synthesis of (2S)-3-Deuterium-L-Valine Esters for Deuterated Drug Development
The pharmaceutical industry is witnessing a paradigm shift towards deuterated drugs, driven by the potential to improve pharmacokinetic profiles and reduce dosing frequency without compromising efficacy. A pivotal development in this domain is disclosed in patent CN113461573B, which details a robust and environmentally friendly preparation method for (2S)-substituted-3-deuterium-L-valine ester compounds and their analogues. This technology addresses the critical challenge of precise deuteration at specific sites, a hurdle that has historically limited the widespread adoption of deuterated therapeutics. By leveraging a novel synthetic route that avoids toxic solvents like benzene and achieves high yields under mild conditions, this process offers a viable pathway for the commercial production of high-purity deuterated amino acid intermediates essential for next-generation drug discovery.
The strategic importance of this synthesis lies in its ability to introduce a deuterium atom at the C-3 position of the valine scaffold with high fidelity. Deuterium substitution can alter the metabolic stability of drug candidates by strengthening the carbon-deuterium bond compared to the carbon-hydrogen bond, a phenomenon known as the kinetic isotope effect. Consequently, reliable access to building blocks like (2S)-substituted-3-deuterium-L-valine is paramount for R&D teams aiming to optimize lead compounds. The methodology described in the patent not only ensures structural integrity but also aligns with modern green chemistry principles, making it an attractive option for sustainable pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterated amino acids has been plagued by significant technical and operational drawbacks that hinder industrial scalability. Conventional routes often rely on harsh reaction conditions and the extensive use of hazardous solvents, particularly benzene, which poses severe health risks and environmental liabilities. Benzene is a combustible, carcinogenic liquid that requires stringent safety protocols for handling, storage, and waste disposal, thereby inflating operational costs and complicating regulatory compliance. Furthermore, traditional deuteration methods frequently suffer from low efficiency and poor regioselectivity, leading to complex impurity profiles that necessitate expensive purification steps such as preparative HPLC. These inefficiencies result in suboptimal yields and extended production timelines, creating bottlenecks in the supply chain for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach outlined in CN113461573B presents a streamlined and safer alternative that effectively circumvents these legacy issues. The process eliminates the need for benzene entirely, utilizing a combination of polar aprotic solvents like DMF, acetonitrile, or acetone, alongside hydrocarbon solvents like toluene for the radical step. This shift not only enhances operator safety but also simplifies the environmental footprint of the manufacturing process. The reaction sequence is designed for high yield and simplicity, avoiding the need for complex chromatographic purification in favor of standard workup procedures. By employing a specific oxalate intermediate strategy, the method ensures precise deuterium incorporation, delivering a product suitable for direct application in the synthesis of deuterated drugs without the burden of excessive impurities.
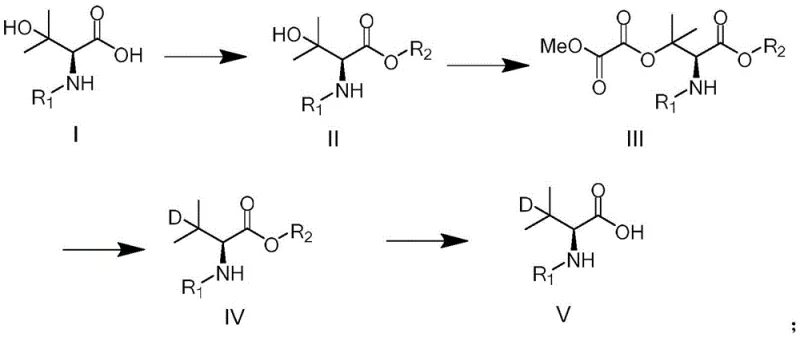
The comprehensive reaction pathway illustrates the elegance of this new methodology, transitioning smoothly from the starting hydroxy-valine derivative through esterification and activation to the final deuterated product. Each step is optimized for maximum conversion, ensuring that the overall process remains economically viable and technically robust. This represents a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing, as it reduces solvent consumption and waste generation while improving throughput.
Mechanistic Insights into Radical Deuteration Strategy
The core innovation of this synthesis resides in the strategic use of a radical-mediated deuteration mechanism to install the isotopic label. The process begins with the activation of the side-chain hydroxyl group of the valine derivative. By reacting the hydroxy-ester intermediate with methyl oxalyl chloride in the presence of an organic base such as N-methylimidazole or triethylamine, an oxalate ester is formed. This oxalate moiety acts as a superior radical leaving group, priming the molecule for the subsequent isotopic exchange. The choice of methyl oxalyl chloride is critical, as it provides the necessary electronic environment to facilitate homolytic cleavage under radical conditions without affecting the stereochemistry at the alpha-carbon, thus preserving the chiral integrity of the L-valine scaffold.
Following activation, the key deuteration step employs a radical initiator system, typically comprising azobisisobutyronitrile (AIBN) and a deuterium source such as triphenyltin deuteride (Ph3SnD) or triphenylsilyl deuteride (Ph3SiD). Upon heating, the initiator generates radicals that abstract the oxalate group, creating a transient carbon-centered radical at the C-3 position. This reactive intermediate rapidly captures a deuterium atom from the tin or silicon reagent, effectively replacing the oxygen functionality with a deuterium atom. The use of toluene or dioxane as the solvent in this step provides an ideal medium for radical propagation, ensuring high conversion rates. This mechanistic pathway allows for precise control over the deuteration site, minimizing scrambling and ensuring that the isotopic label is located exactly where it is needed for metabolic modulation.
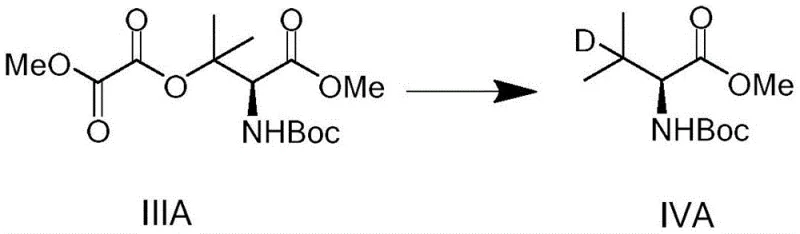
The specificity of this radical mechanism is further evidenced by the high purity of the resulting Formula IV compounds, which consistently demonstrate purity levels exceeding 98% in experimental examples. The ability to tune the reaction by selecting different initiators or deuterated reagents offers flexibility for process optimization, allowing manufacturers to balance cost and performance. For instance, while triphenyltin deuteride is highly effective, alternatives can be explored to mitigate heavy metal residues if necessary, although the patent indicates that standard purification methods suffice to remove such byproducts. This level of mechanistic control is essential for producing high-purity OLED material precursors or pharmaceutical intermediates where isotopic enrichment is a critical quality attribute.
How to Synthesize (2S)-substituted-3-deuterium-L-valine Ester Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly during the radical deuteration phase. The process is divided into distinct stages: initial esterification of the carboxylic acid, activation of the hydroxyl group via oxalylation, radical substitution with deuterium, and final hydrolysis to the free acid if required. Operators must ensure strict moisture control during the oxalylation step to prevent hydrolysis of the activated intermediate. Furthermore, the radical step demands precise temperature control, typically involving reflux conditions in toluene, to maintain the steady generation of radicals without inducing side reactions. The detailed standardized synthesis steps see the guide below for specific molar ratios and solvent volumes optimized for scale-up.
- Perform esterification of the starting hydroxy-valine acid (Formula I) using an alkylating agent like methyl iodide or dimethyl sulfate to form the ester intermediate (Formula II).
- React the ester intermediate with methyl oxalyl chloride in the presence of an organic base to install the oxalate leaving group, yielding Formula III.
- Execute the key radical deuteration step by treating Formula III with a deuterated reagent such as triphenyltin deuteride (Ph3SnD) and an initiator like AIBN to replace the oxalate with deuterium (Formula IV).
- Conclude the synthesis by hydrolyzing the ester group of Formula IV under basic conditions to obtain the final deuterated valine acid (Formula V).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial advantages that translate directly into operational efficiency and risk mitigation. The elimination of benzene, a highly regulated and hazardous solvent, removes a significant logistical burden associated with sourcing, handling, and disposing of toxic chemicals. This change alone streamlines the supply chain by reducing the need for specialized containment infrastructure and lowering insurance premiums related to hazardous material storage. Moreover, the use of commodity chemicals such as methyl iodide, dimethyl sulfate, and methyl oxalyl chloride ensures that raw material availability is stable and pricing is predictable, shielding manufacturers from the volatility often seen with exotic reagents.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization through high-yield reactions that minimize material waste. By avoiding the use of preparative high-performance liquid chromatography (HPLC) for purification and relying instead on conventional extraction and crystallization techniques, the method drastically reduces downstream processing costs. The high atom economy of the radical deuteration step ensures that expensive deuterated reagents are utilized efficiently, preventing unnecessary expenditure on isotopic materials. Additionally, the reduced solvent volume requirements compared to traditional methods lower both procurement costs and waste treatment fees, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: The reliance on widely available solvents like DMF, acetone, and toluene enhances supply chain resilience. Unlike specialized solvents that may face shortages or long lead times, these commodities are produced at massive scales globally, ensuring continuous availability for production runs. The robustness of the reaction conditions, which tolerate mild temperatures and standard atmospheric pressure, further reduces the risk of batch failures due to equipment limitations. This reliability is crucial for maintaining consistent delivery schedules to downstream pharmaceutical clients who depend on just-in-time inventory models for their drug development pipelines.
- Scalability and Environmental Compliance: The method is explicitly designed for industrial mass production, with reaction parameters that scale linearly from laboratory to plant scale. The absence of benzene and the use of recyclable solvents align with increasingly stringent environmental regulations, facilitating easier permitting and compliance auditing. The simplified workup procedures, involving standard aqueous washes and drying steps, are easily adaptable to large-scale reactors without requiring complex engineering modifications. This scalability ensures that the supply of deuterated intermediates can be ramped up quickly to meet surging demand in the deuterated drug sector without compromising on quality or safety standards.
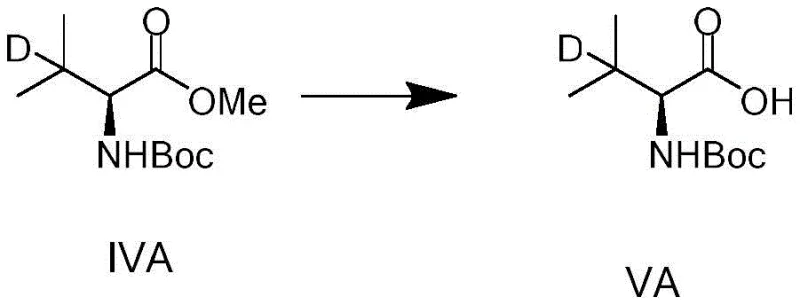
The final hydrolysis step, converting the ester to the free acid, further demonstrates the versatility of the platform. As shown in the reaction scheme, this transformation can be achieved using various bases like lithium hydroxide or sodium hydroxide in mixed solvent systems, providing flexibility to tailor the final salt form or physical properties of the product. This adaptability is a key asset for supply chain managers who may need to deliver the intermediate in different forms depending on the specific requirements of the customer's subsequent synthetic steps.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deuterated valine synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the removal of benzene significant in this synthesis process?
A: Benzene is a known carcinogen with high toxicity and strict regulatory limits. Eliminating benzene from the solvent system significantly reduces environmental hazards, lowers safety compliance costs, and simplifies waste treatment, making the process more suitable for large-scale industrial production.
Q: What is the role of the oxalate group in the deuteration step?
A: The oxalate group serves as an excellent radical leaving group. By converting the hydroxyl group into an oxalate ester (Formula III), the molecule becomes susceptible to radical substitution. This allows the deuterium atom from reagents like Ph3SnD to precisely replace the oxygen functionality at the specific C-3 position with high regioselectivity.
Q: Can this method be scaled for commercial API manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrial mass production. It utilizes common solvents like toluene, DMF, and acetone, avoids hazardous benzene, and demonstrates high yields across all steps, which are critical factors for scalable commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S)-substituted-3-deuterium-L-valine Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of deuterated chemistry in modern drug development and are committed to supporting our partners with cutting-edge synthetic solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (2S)-substituted-3-deuterium-L-valine ester meets the highest standards of isotopic enrichment and chemical purity required for clinical applications.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains for deuterated intermediates. By leveraging our technical expertise and the advanced processes described in CN113461573B, we can help you achieve significant efficiencies in your manufacturing operations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your drug development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
