Advanced One-Step Synthesis of Revefenacin Intermediate for Commercial API Manufacturing
The pharmaceutical landscape for Chronic Obstructive Pulmonary Disease (COPD) treatment has been significantly advanced by the introduction of Revefenacin, marketed as Yupelri. As the first long-acting muscarinic antagonist (LAMA) delivered via nebulizer, its demand necessitates robust and efficient supply chains for its key precursors. Patent CN113563255A discloses a groundbreaking preparation method for the critical intermediate of Revefenacin, specifically addressing the historical bottlenecks of multi-step synthesis and environmental hazards. This technical insight report analyzes the transition from cumbersome legacy routes to a streamlined, one-step nucleophilic substitution process, offering a compelling value proposition for R&D directors and procurement strategists alike.
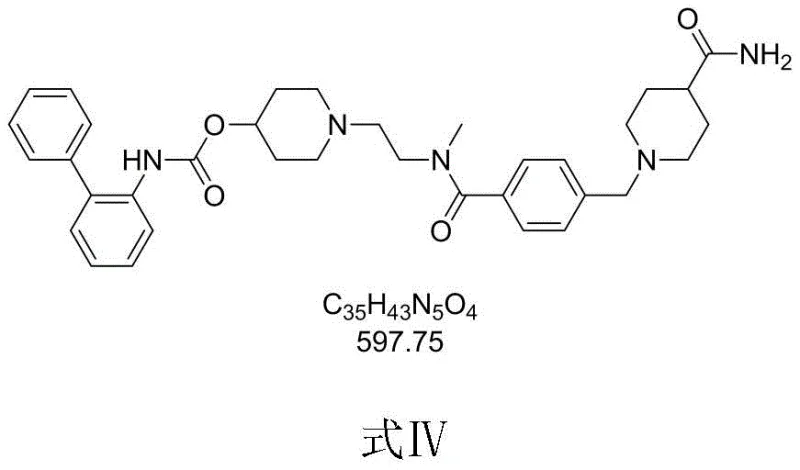
Revefenacin represents a significant therapeutic milestone, yet its complex molecular architecture has traditionally posed challenges for scalable manufacturing. The patent highlights that previous synthetic strategies were plagued by excessive step counts and the reliance on hazardous reagents. By focusing on the efficient construction of the piperidine-ethylamine side chain onto the biphenyl carbamate core, this new methodology promises to redefine the production economics of this high-value respiratory drug intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of the key intermediate relied on convoluted pathways that introduced significant operational risk and cost. As illustrated in earlier patent literature, traditional routes often involved the oxidation of alcohol precursors using toxic agents like Py·SO3 or the necessity of catalytic hydrogenation steps using Palladium on Carbon (Pd/C). These processes not only extend the production timeline but also introduce heavy metal contamination risks that require rigorous and costly purification protocols to meet pharmaceutical standards.
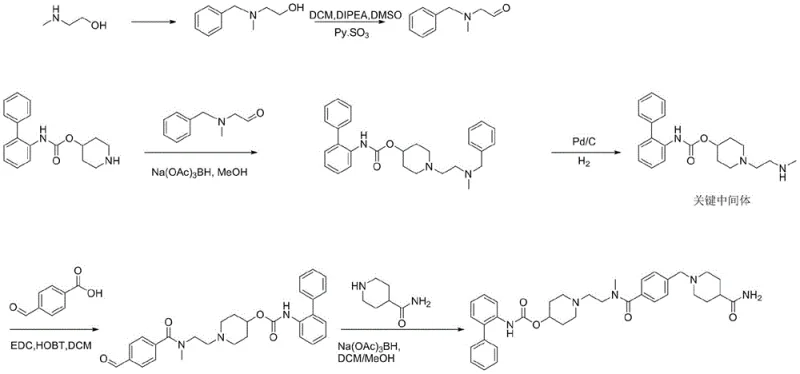
Furthermore, the conventional approaches frequently suffered from low overall yields due to the accumulation of losses across multiple isolation steps. The use of chlorinated solvents and strong oxidants raised substantial environmental, health, and safety (EHS) concerns, complicating waste disposal and regulatory compliance. For a commercial manufacturer, these factors translate into higher operating expenditures (OPEX) and a fragile supply chain vulnerable to raw material shortages of specialized reagents.
The Novel Approach
In stark contrast, the method disclosed in CN113563255A simplifies the entire operation into a single, direct substitution reaction. The core transformation involves reacting piperidin-4-yl [1,1'-biphenyl]-2-carbamate (Formula II) directly with a halo-alkylamine derivative (Formula III). This eliminates the need for pre-functionalization of the amine side chain or post-reaction hydrogenation. The reaction proceeds efficiently in alcoholic solvents, utilizing common inorganic or organic bases to drive the nucleophilic attack.
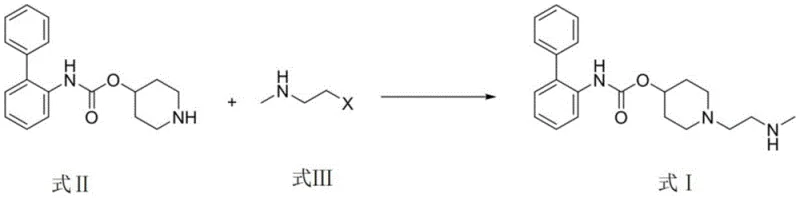
This streamlined approach drastically reduces the process mass intensity (PMI) by removing intermediate isolation steps. The avoidance of toxic oxidants and noble metal catalysts not only enhances the environmental profile but also simplifies the downstream processing. The result is a high-yielding, robust process that is inherently safer and more cost-effective, aligning perfectly with the principles of green chemistry and modern pharmaceutical manufacturing efficiency.
Mechanistic Insights into Nucleophilic Substitution
The heart of this synthesis lies in a classic yet optimized nucleophilic substitution mechanism. The secondary amine within the piperidine ring of Formula II acts as the nucleophile, attacking the electrophilic carbon adjacent to the halogen (X = Cl, Br, I) in Formula III. The choice of solvent plays a pivotal role here; polar protic solvents like ethanol or methanol facilitate the dissolution of the salt forms of the alkylating agent while stabilizing the transition state. The patent data indicates that ethanol provides an optimal balance, maximizing the reaction rate while minimizing side reactions.
Impurity control is achieved through precise stoichiometric management and temperature regulation. A major side reaction involves the over-alkylation of the newly formed tertiary amine, leading to a quaternary ammonium salt or bis-alkylated byproduct (often structurally related to Formula IV variants). By maintaining the reaction temperature between 0°C and 10°C and carefully controlling the molar ratio of the base (such as DIPEA or potassium carbonate) to the alkylating agent, the formation of these impurities is suppressed. This kinetic control ensures that the mono-alkylated product (Formula I) is obtained with high purity, reducing the burden on final crystallization steps.
How to Synthesize Revefenacin Intermediate Efficiently
The operational simplicity of this route makes it highly attractive for technology transfer. The process begins with the suspension of the carbamate starting material in ethanol, followed by the addition of the halo-amine salt. The critical control point is the slow, batch-wise addition of the base under cooling to manage the exotherm and prevent local hotspots that could trigger degradation. Following the reaction, a straightforward acid quench and solvent swap allows for high-purity crystallization.
- Dissolve the carbamate starting material (Formula II) in an alcoholic solvent such as ethanol or methanol.
- Add the halo-alkylamine salt (Formula III) and cool the mixture to 0-10°C.
- Slowly add a base like DIPEA or potassium carbonate, maintain temperature for 3-6 hours, then quench and crystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this novel synthetic route offers tangible strategic benefits beyond mere technical elegance. The elimination of complex multi-step sequences directly correlates to a reduction in manufacturing lead times and working capital requirements. By consolidating the synthesis into a single reactor train, facilities can increase throughput without expanding physical footprint, effectively debottlenecking production capacity for this high-demand intermediate.
- Cost Reduction in Manufacturing: The most significant economic driver is the removal of expensive catalytic systems and toxic oxidants. Traditional routes requiring Palladium catalysts incur high costs not just for the reagent but for the specialized recovery and scavenging processes needed to meet strict residual metal limits. By adopting a metal-free substitution strategy, manufacturers can drastically lower raw material costs and simplify waste treatment protocols. Furthermore, the use of commodity solvents like ethanol instead of specialized chlorinated hydrocarbons reduces solvent procurement and recycling expenses.
- Enhanced Supply Chain Reliability: The starting materials for this route, specifically the biphenyl carbamate and simple halo-alkylamines, are commercially available bulk chemicals with stable supply chains. Unlike specialized oxidants or custom-synthesized precursors required in older methods, these inputs are less susceptible to market volatility. This stability ensures consistent production scheduling and reduces the risk of supply disruptions, a critical factor for maintaining the continuity of API supply for global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind. The reaction conditions (0-15°C) are easily achievable with standard jacketed reactors, avoiding the need for cryogenic cooling or high-pressure hydrogenation vessels. From an EHS perspective, the absence of toxic oxidants and heavy metals simplifies regulatory filings and environmental permitting. This 'green' profile facilitates faster approval in regulated markets and aligns with the increasing corporate sustainability goals of major pharmaceutical buyers.
Frequently Asked Questions (FAQ)
Understanding the nuances of this new process is essential for stakeholders evaluating its adoption. The following questions address common technical and operational concerns regarding the synthesis of the Revefenacin intermediate, drawing directly from the experimental data and comparative analysis provided in the patent documentation.
Q: What are the primary impurities in this synthesis?
A: The main impurity is the bis-alkylated byproduct (Formula IV structure variant), formed if reaction conditions are not strictly controlled. Optimization of base and stoichiometry minimizes this.
Q: Why is ethanol preferred over dichloromethane?
A: Ethanol provides a superior balance of solubility and reaction rate, significantly reducing the formation of byproducts compared to chlorinated solvents while being more environmentally friendly.
Q: Is catalytic hydrogenation required?
A: No, unlike prior art routes which require Pd/C hydrogenation, this novel method relies solely on nucleophilic substitution, eliminating the need for expensive metal catalysts and high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Revefenacin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthetic route requires a partner with deep technical expertise and proven execution capabilities. Our team has extensively analyzed the pathway described in CN113563255A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of intermediate meets the exacting standards required for respiratory API manufacturing.
We invite you to discuss how this optimized synthesis can impact your project timelines and cost structures. Contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments to support your next phase of development and commercialization.
