Scalable Metal-Free Synthesis of Alpha-Unsaturated Aryl Ketones Using Dimethyl Sulfoxide for Industrial Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing carbon-carbon double bonds adjacent to carbonyl groups, a structural motif prevalent in countless bioactive molecules. A significant breakthrough in this domain is detailed in patent CN107759458B, which discloses a novel, environmentally benign method for synthesizing α-unsaturated aryl ketone compounds directly from arylethanones and dimethyl sulfoxide (DMSO). This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a more sustainable, metal-free oxidative methylenation strategy. By utilizing DMSO not merely as a solvent but as the active methylene reagent in the presence of a carboxylate base and persulfate oxidant, this process achieves high selectivity and yield under relatively mild conditions. For R&D directors and procurement specialists, this innovation offers a compelling alternative to legacy methods that rely on hazardous reagents and complex workup procedures, promising significant improvements in both cost-efficiency and supply chain reliability for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α-unsaturated ketones has relied heavily on methodologies that introduce substantial environmental and operational burdens. As illustrated in prior art, such as the reaction utilizing N,N-dimethylacetamide with ferric chloride and potassium persulfate, the dependence on transition metal salts creates severe downstream processing challenges.
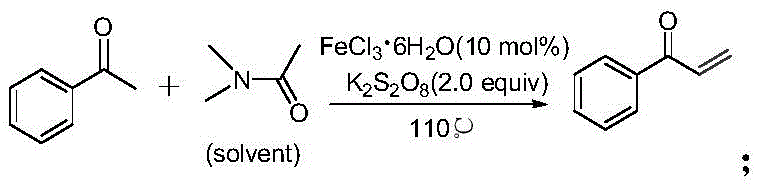
These metal-catalyzed routes often suffer from moderate yields and necessitate rigorous purification steps to remove trace heavy metals, which is a critical compliance hurdle for API manufacturing. Furthermore, alternative approaches employing trioxymethylene and trifluoroacetic acid secondary amine salts, while effective in laboratory settings, are plagued by the high cost of fluorinated reagents and safety concerns associated with handling unstable formaldehyde sources. These conventional pathways frequently require stoichiometric amounts of expensive catalysts and generate significant chemical waste, rendering them economically unviable for large-scale industrial production where cost reduction in pharmaceutical intermediate manufacturing is paramount.
The Novel Approach
The methodology described in CN107759458B elegantly circumvents these issues by employing a one-pot reaction system where dimethyl sulfoxide acts as the methylene donor. This approach eliminates the need for transition metals entirely, replacing them with inexpensive, commercially available sodium acetate and potassium persulfate. The general reaction scheme demonstrates the direct conversion of arylethanones into the desired α-unsaturated products with remarkable efficiency.
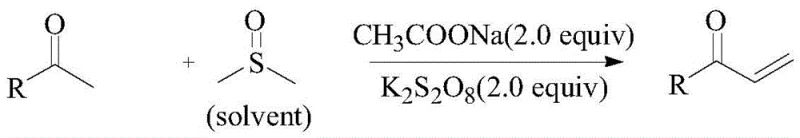
In this optimized protocol, the reaction proceeds smoothly at temperatures between 110°C and 130°C, typically reaching completion within 6 to 12 hours. The use of DMSO as both the solvent and reagent drastically simplifies the reaction matrix, reducing the volume of organic solvents required and minimizing waste generation. For example, the conversion of acetophenone to phenyl vinyl ketone achieves an impressive 85% isolated yield under standard conditions, while substrates like 2-acetylnaphthalene can reach yields as high as 90%. This simplicity and high atom economy make the process exceptionally attractive for commercial scale-up of complex pharmaceutical intermediates, offering a green chemistry solution that aligns with modern regulatory standards.
Mechanistic Insights into Non-Metal Catalyzed Oxidative Methylenation
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and impurity control. Unlike many persulfate-mediated reactions that proceed via radical pathways, experimental evidence suggests this specific transformation follows an ionic mechanism. The proposed cycle begins with the deprotonation of the arylethanone by the carboxylate base to form a stabilized enolate species. Simultaneously, the persulfate oxidant facilitates the activation of DMSO, likely generating a reactive sulfonium intermediate through intramolecular rearrangement and dehydration.
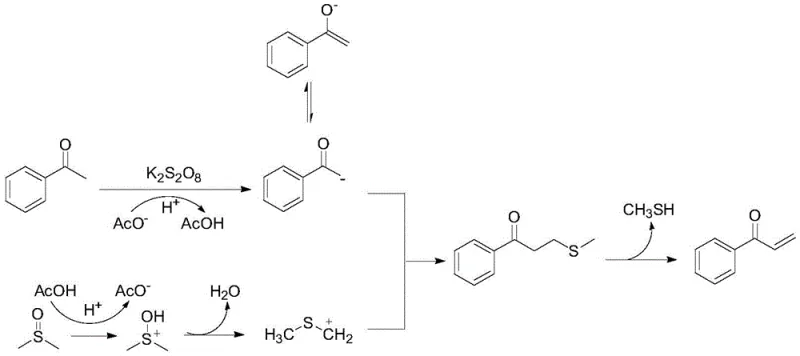
This electrophilic sulfonium species then reacts with the nucleophilic enolate to form a thioether derivative intermediate. Subsequent thermal elimination of methyl mercaptan from this intermediate yields the final α-unsaturated ketone product. Crucially, control experiments utilizing radical inhibitors such as BHT and TEMPO showed negligible impact on reaction yields, confirming the non-radical nature of the pathway. Additionally, isotopic labeling studies using deuterated DMSO confirmed that the methylene group in the product originates exclusively from the solvent, validating the role of DMSO as the carbon source. This mechanistic clarity allows for precise tuning of reaction parameters to minimize side reactions, ensuring the production of high-purity OLED material precursors or drug intermediates with consistent quality.
How to Synthesize Phenyl Vinyl Ketone Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and temperature control to maximize throughput. The standard protocol involves mixing the arylethanone substrate with two equivalents of sodium acetate and two equivalents of potassium persulfate in excess DMSO. The mixture is heated to 120°C and stirred until TLC analysis indicates complete consumption of the starting material. Following the reaction, the excess DMSO is recovered via distillation, allowing for solvent recycling which further enhances the economic viability of the process. The crude product is then purified using standard column chromatography techniques to isolate the target α-unsaturated ketone.
- Mix arylethanone derivative, sodium acetate catalyst, and potassium persulfate oxidant in dimethyl sulfoxide solvent.
- Heat the reaction mixture to 110-130°C and maintain stirring for 6 to 12 hours to ensure complete conversion.
- Recover excess dimethyl sulfoxide via distillation and purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMSO-based methylenation technology offers transformative benefits regarding cost structure and logistical stability. The elimination of transition metal catalysts removes the necessity for expensive metal scavenging resins and complex filtration steps, directly translating to reduced manufacturing overheads. Furthermore, the reliance on commodity chemicals like DMSO and sodium acetate ensures a stable supply chain unaffected by the volatility often seen with specialized organometallic reagents.
- Cost Reduction in Manufacturing: The process utilizes dimethyl sulfoxide, a low-cost bulk solvent, as the primary reagent, effectively removing the need for expensive methylating agents like paraformaldehyde derivatives or fluorinated amine salts. By operating without transition metals, the downstream purification process is significantly simplified, avoiding the capital expenditure associated with heavy metal removal technologies. This streamlined workflow reduces both raw material costs and waste disposal fees, resulting in substantial overall cost savings for large-volume production runs.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, specifically potassium persulfate and sodium acetate, are globally available commodity chemicals with robust supply networks. This contrasts sharply with proprietary catalysts that may have single-source suppliers and long lead times. The ability to source all key inputs from multiple vendors mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The one-pot nature of the reaction minimizes unit operations, making it inherently easier to scale from kilogram to multi-ton batches without significant re-engineering. Moreover, the absence of toxic heavy metals and fluorinated byproducts simplifies environmental compliance and wastewater treatment. The process generates fewer hazardous wastes, aligning with increasingly stringent global environmental regulations and supporting sustainable manufacturing goals without compromising on yield or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for technical teams evaluating this route for potential adoption in their manufacturing pipelines.
Q: What are the primary advantages of using DMSO over traditional methylating agents?
A: Unlike toxic formaldehyde sources or expensive amine salts, DMSO serves as a dual-purpose low-cost solvent and methylene source, eliminating the need for transition metal catalysts like iron chloride and simplifying downstream purification.
Q: Does this reaction mechanism involve free radicals?
A: Experimental data indicates the reaction proceeds via an ionic pathway rather than a free radical mechanism, as evidenced by the lack of inhibition when radical scavengers like BHT or TEMPO are added to the system.
Q: What is the substrate scope for this methylenation protocol?
A: The method demonstrates broad compatibility with various substituted acetophenones, including electron-donating groups like methoxy and ethoxy, as well as electron-withdrawing groups such as halogens and cyano groups, yielding products in up to 90% efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Unsaturated Aryl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes like the DMSO-mediated methylenation described in CN107759458B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of α-unsaturated aryl ketone delivered meets the highest standards required for pharmaceutical and electronic material applications.
We invite you to collaborate with our technical team to evaluate the feasibility of this cost-effective route for your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior chemical manufacturing solutions.
