Advanced Chiral Ferrocene Ligands for High-Efficiency Asymmetric Hydrogenation Catalysis
Advanced Chiral Ferrocene Ligands for High-Efficiency Asymmetric Hydrogenation Catalysis
The landscape of asymmetric synthesis is continuously evolving, driven by the relentless demand for enantiomerically pure compounds in the pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is documented in Chinese Patent CN103012498A, which discloses a novel class of chiral ferrocene tridentate ligands possessing both planar and central chirality. These ligands represent a significant leap forward in catalyst design, specifically tailored for the asymmetric hydrogenation of pro-chiral ketones. By integrating the robust structural motif of ferrocene with the versatile stereochemistry of Ugi's amine derivatives, this technology addresses critical limitations found in earlier generations of catalysts. For R&D directors and process chemists, understanding the nuances of this patent is essential for developing next-generation synthetic routes that prioritize both efficiency and stereoselectivity. The following analysis dissects the technical merits and commercial viability of this innovation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on established ligand families such as DIOP, BINAP, and DuPHOS to facilitate asymmetric hydrogenation reactions. While these ligands have served as workhorses for decades, they are not without their drawbacks when applied to complex modern synthesis challenges. Traditional bidentate phosphine ligands often struggle to maintain high turnover numbers (TON) and enantioselectivity simultaneously, particularly when dealing with sterically hindered or electronically diverse ketone substrates. Furthermore, the synthesis of some high-performance ligands like SpiroPAP can involve intricate multi-step sequences that are difficult to scale cost-effectively. The background art cited in the patent highlights that while TON values reaching one million are the gold standard, catalysts achieving this benchmark remain scarce. This scarcity creates a bottleneck for procurement managers seeking reliable sources for high-volume chiral intermediates, as low catalyst efficiency directly translates to higher metal loading costs and more complex downstream purification processes.
The Novel Approach
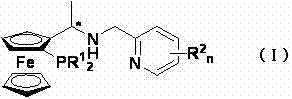
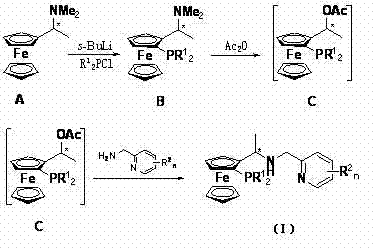
The methodology outlined in CN103012498A offers a compelling alternative by leveraging a modular synthesis strategy centered on chiral Ugi's amine. The novelty lies in the sequential functionalization of the ferrocene core, which allows for precise control over both the planar chirality of the metallocene and the central chirality of the side chain. As illustrated in the reaction scheme, the process begins with the lithiation of the starting amine, followed by coupling with chlorophosphines and subsequent esterification. This route avoids the need for resolving racemic mixtures at late stages, as the chirality is inherited directly from the commercially available or easily prepared Ugi's amine precursor. The introduction of the nitrogen donor arm via nucleophilic substitution with aminomethylpyridine derivatives completes the tridentate architecture. This structural rigidity is crucial for creating a well-defined chiral pocket around the iridium center, thereby enhancing the catalyst's ability to discriminate between enantiotopic faces of the substrate during hydrogenation.
Mechanistic Insights into Ir-Catalyzed Asymmetric Hydrogenation
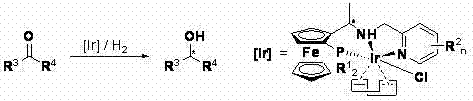
The efficacy of these ferrocene ligands stems from their ability to form stable, highly active iridium complexes that operate through a well-defined catalytic cycle. When coordinated with an iridium precursor such as [Ir(COD)Cl]2, the tridentate ligand creates a cationic or neutral active species capable of activating molecular hydrogen under relatively mild conditions. The mechanism involves the oxidative addition of hydrogen to the metal center, followed by the coordination of the pro-chiral ketone substrate. The unique spatial arrangement of the phosphine and nitrogen donors, dictated by the ferrocene backbone, forces the substrate into a specific orientation that favors the formation of one enantiomer over the other. This stereochemical control is further reinforced by the steric bulk of the substituents on the phosphorus atom (R1 groups) and the pyridine ring (R2 groups), which can be tuned to match the specific steric demands of the target ketone. Such tunability is a powerful tool for process optimization, allowing chemists to maximize enantiomeric excess (ee) without changing the core catalytic machinery.
From an impurity control perspective, the robustness of the ferrocene-iridium bond ensures that the catalyst remains intact throughout the reaction, minimizing the leaching of free metal ions into the product stream. This is a critical consideration for pharmaceutical manufacturing, where residual heavy metals must be kept below strict regulatory thresholds (e.g., ICH Q3D guidelines). The patent data indicates that these catalysts can operate at molar ratios of substrate to ligand ranging from 1000:1 to 1,000,000:1, demonstrating exceptional turnover capabilities. High TON values not only reduce the amount of expensive iridium required per batch but also simplify the workup procedure, as less catalyst residue needs to be removed. This mechanistic efficiency directly supports the goal of producing high-purity pharmaceutical intermediates with minimal environmental footprint and reduced operational complexity.
How to Synthesize Chiral Ferrocene Tridentate Ligands Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing these high-value ligands with consistent quality. The process is designed to be operationally simple, utilizing standard laboratory equipment such as Schlenk tubes and autoclaves, which facilitates easy translation to pilot and commercial scales. The key to success lies in maintaining strict anhydrous conditions during the lithiation step and carefully controlling the temperature profiles to prevent side reactions. Below is a summary of the critical operational parameters derived from the experimental examples, which serve as a foundation for process development teams aiming to implement this technology.
- Lithiation of chiral Ugi's amine using sec-butyllithium in methyl tert-butyl ether at controlled temperatures.
- Coupling with chlorophosphines followed by esterification with acetic anhydride to form the acetate intermediate.
- Final nucleophilic substitution with aminomethylpyridine derivatives in methanol/THF to yield the target tridentate ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ligand technology presents several strategic advantages that align with goals of cost reduction and supply security. The synthesis route relies on readily available starting materials, such as Ugi's amine and common chlorophosphines, which mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Furthermore, the reaction conditions employ standard organic solvents like methyl tert-butyl ether, methanol, and tetrahydrofuran, eliminating the need for exotic or hazardous reagents that require special handling and disposal protocols. This compatibility with existing infrastructure means that contract manufacturing organizations (CMOs) can integrate this process without significant capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The high catalytic activity and turnover numbers associated with these iridium complexes translate directly into lower catalyst loading requirements. By reducing the amount of precious metal needed per kilogram of product, manufacturers can achieve substantial cost savings in the bill of materials. Additionally, the simplified synthesis of the ligand itself, which avoids complex resolution steps, contributes to a more favorable cost profile compared to legacy ligand systems. The elimination of extensive purification steps for the final alcohol product, due to high selectivity, further drives down processing costs and improves overall yield.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply to downstream drug substance manufacturers. The use of stable intermediates and the ability to store the ligand precursors allows for flexible production scheduling, enabling suppliers to respond quickly to fluctuating market demands. Moreover, the versatility of the ligand structure, which can be modified by varying the R1 and R2 substituents, provides a buffer against supply disruptions for specific reagents, as alternative analogs can often be substituted with minimal impact on performance.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-gram quantities in the patent examples with straightforward workup procedures like filtration and crystallization. The absence of toxic heavy metal byproducts and the use of recyclable solvents align with green chemistry principles, helping companies meet increasingly stringent environmental regulations. This compliance reduces the burden of waste treatment and disposal, resulting in indirect cost savings and a stronger sustainability profile for the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ferrocene ligand technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners and licensees.
Q: What distinguishes this ferrocene ligand from traditional BINAP or DuPHOS systems?
A: This ligand uniquely combines both planar chirality from the ferrocene backbone and central carbon chirality from the Ugi's amine scaffold, offering superior stereocontrol in asymmetric hydrogenation compared to purely axial chiral ligands.
Q: What are the optimal reaction conditions for the asymmetric hydrogenation of ketones using this catalyst?
A: The patent specifies reaction temperatures between 0°C and 60°C, hydrogen pressures ranging from 1 to 150 atm, and the use of alcoholic solvents like methanol, achieving high turnover numbers (TON) suitable for industrial application.
Q: Is the synthesis route scalable for commercial production of pharmaceutical intermediates?
A: Yes, the synthesis utilizes robust reagents like sec-butyllithium and standard solvents such as methyl tert-butyl ether and acetic anhydride, avoiding exotic conditions and facilitating scale-up from laboratory to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ferrocene Ligand Supplier
The technological potential of the chiral ferrocene tridentate ligands described in CN103012498A is immense, offering a pathway to more efficient and sustainable asymmetric synthesis. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to bring this advanced chemistry from the laboratory bench to full-scale commercial production. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of ligand or catalyst meets the highest industry standards, providing you with the confidence needed for critical drug development programs.
We invite you to collaborate with us to explore how this technology can optimize your specific synthetic routes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
