Scalable Synthesis of High-Purity Synthetic Cannabinoid Hapten Intermediates for Immunoassay Development
Scalable Synthesis of High-Purity Synthetic Cannabinoid Hapten Intermediates for Immunoassay Development
The rapid expansion of the diagnostic market for novel psychoactive substances has created an urgent demand for high-quality immunogens capable of detecting trace levels of synthetic cannabinoids in complex biological matrices. Patent CN115850146A addresses this critical gap by disclosing a robust methodology for synthesizing specialized hapten compounds that serve as the foundational building blocks for antibody generation. These haptens are engineered to mimic the structural epitopes of synthetic cannabinoids while incorporating a functional linker arm that facilitates stable conjugation to carrier proteins. The innovation lies not merely in the molecular structure but in the strategic placement of the carboxylic acid functionality, which ensures that the antigenic determinants of the parent drug molecule remain exposed to the immune system. For R&D directors and procurement specialists in the diagnostic sector, this patent represents a significant leap forward in the reliability of immunoassay kits, offering a pathway to produce reagents with superior sensitivity and specificity compared to traditional random conjugation methods.
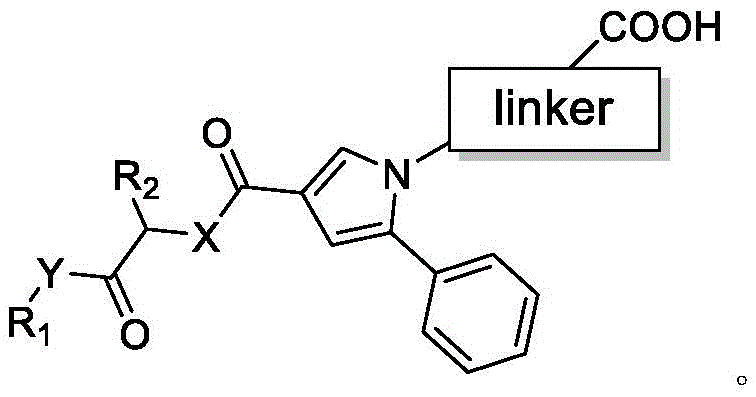
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of haptens for synthetic cannabinoid detection has been plagued by inefficient synthetic routes that often compromise the structural integrity of the target molecule. Traditional methods frequently rely on non-specific functionalization, where reactive groups are introduced at random positions on the cannabinoid scaffold, leading to heterogeneous populations of antigens that confuse the immune response. Furthermore, many legacy processes utilize harsh reaction conditions, such as strong acidic or basic environments at elevated temperatures, which can degrade sensitive heterocyclic cores like pyrroles or indoles commonly found in these drugs. This degradation results in low overall yields and necessitates extensive purification steps, such as preparative HPLC, which drastically increases the cost of goods and extends the timeline for reagent development. Additionally, the lack of a standardized linker strategy in older patents often means that each new analog requires a completely bespoke synthesis route, preventing the economies of scale that are essential for a reliable synthetic cannabinoid hapten supplier to maintain competitive pricing.
The Novel Approach
The methodology outlined in the patent data introduces a modular and highly controlled synthetic strategy that overcomes these historical bottlenecks through a rational design of the linker arm and reaction sequence. By utilizing a 5-phenylpyrrole-3-carboxylic acid core, the process establishes a stable scaffold that closely mimics the pharmacophore of many synthetic cannabinoids, ensuring high cross-reactivity and detection accuracy. The novelty is further enhanced by the use of a bromo-ester intermediate for alkylation, which allows for the precise installation of a carbon chain linker under mild conditions, preserving the delicate electronic properties of the heterocycle. This approach effectively decouples the complexity of the core synthesis from the functionalization step, allowing manufacturers to produce a library of related haptens using a common set of intermediates. Consequently, this streamlines the workflow for cost reduction in API intermediate manufacturing, as the same reaction vessels and purification protocols can be adapted for multiple targets with minimal retooling or process validation overhead.
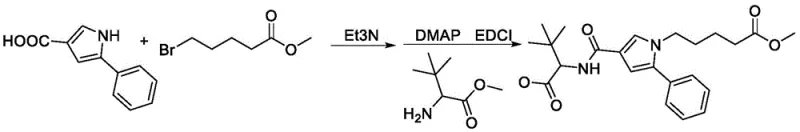
Mechanistic Insights into Peptide Coupling and N-Alkylation
The core of this synthetic success relies on a sophisticated understanding of nucleophilic substitution and carbodiimide-mediated amide bond formation, executed with precision to minimize impurity profiles. The initial alkylation step involves the deprotonation of the pyrrole nitrogen or the carboxylic acid oxygen, followed by a nucleophilic attack on the electrophilic carbon of methyl 5-bromovalerate. This SN2-type reaction is carefully managed using triethylamine as a base, which scavenges the generated hydrogen bromide without promoting elimination side reactions that could lead to alkene impurities. Following this, the introduction of the amino acid moiety is achieved through a classic EDCI/DMAP coupling mechanism. Here, EDCI activates the carboxylic acid to form an O-acylisourea intermediate, which is highly susceptible to nucleophilic attack by the amine of tert-leucine methyl ester. The presence of DMAP acts as a nucleophilic catalyst, accelerating the formation of the active ester and suppressing the formation of unreactive N-acylurea byproducts, which are common pitfalls in peptide synthesis.
Impurity control is paramount in the production of high-purity OLED material or pharmaceutical intermediates, and this mechanism offers inherent advantages in that regard. The use of tert-leucine, with its bulky tert-butyl side chain, provides significant steric hindrance that prevents racemization at the alpha-carbon during the coupling process, a frequent issue with other amino acids. Furthermore, the final hydrolysis step utilizes lithium hydroxide under mild conditions to cleave the methyl ester without affecting the newly formed amide bond or the ether linkage in the chain. This chemoselectivity ensures that the final product contains the free carboxylic acid required for protein conjugation while maintaining the integrity of the rest of the molecule. For quality control teams, this translates to a cleaner crude product that requires less aggressive purification, thereby improving the overall mass balance and reducing the environmental footprint associated with solvent-intensive chromatography.
How to Synthesize Synthetic Cannabinoid Hapten Efficiently
The operational execution of this synthesis is designed to be compatible with standard multipurpose reactor setups found in most fine chemical facilities, requiring no specialized high-pressure or cryogenic equipment beyond standard cooling capabilities. The process begins with the dissolution of the pyrrole acid in anhydrous dichloromethane, followed by the controlled addition of the alkylating agent and base, a sequence that manages the exotherm effectively to prevent thermal runaway. Subsequent steps involve straightforward aqueous workups and silica gel chromatography, techniques that are universally understood by process chemists and easily transferable from gram to kilogram scales. The detailed标准化 synthesis steps see the guide below for specific stoichiometric ratios and temperature profiles that ensure reproducible results across different batches.
- Perform nucleophilic substitution by reacting 5-phenylpyrrole-3-carboxylic acid with methyl 5-bromovalerate in the presence of triethylamine to form the alkylated intermediate.
- Execute amide condensation using EDCI and DMAP catalysts to couple the intermediate with tert-leucine methyl ester, forming the protected hapten precursor.
- Conduct mild basic hydrolysis using lithium hydroxide to remove the methyl ester protecting group, yielding the final carboxylic acid functionalized hapten.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible benefits that extend far beyond the laboratory bench, directly impacting the bottom line and operational resilience. The primary advantage lies in the simplification of the raw material portfolio; by relying on commodity chemicals like methyl 5-bromovalerate and protected amino acids, the process mitigates the risk of supply disruptions often associated with custom-synthesized building blocks. This accessibility ensures that production schedules can be maintained even during periods of global raw material volatility, providing a level of supply chain reliability that is crucial for meeting the strict delivery deadlines of diagnostic kit manufacturers. Moreover, the high atom economy of the coupling reactions reduces the volume of waste generated per kilogram of product, aligning with increasingly stringent environmental regulations and lowering waste disposal costs significantly.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts, which are often required for cross-coupling reactions in alternative routes, results in substantial cost savings and removes the need for costly heavy metal scavenging steps. By avoiding precious metals like palladium or platinum, the process not only lowers the direct material cost but also simplifies the regulatory burden associated with residual metal limits in pharmaceutical and diagnostic products. The use of standard organic solvents and reagents further drives down the operational expenditure, as these materials are available in bulk quantities at competitive market rates without the need for specialized storage or handling protocols.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the decoupling of intermediate production from final assembly, enabling a more flexible inventory management strategy where key precursors can be stockpiled without risking degradation. This flexibility is vital for reducing lead time for high-purity synthetic cannabinoid haptens, as it allows manufacturers to respond rapidly to surges in demand for specific diagnostic tests without waiting for long lead-time custom synthesis campaigns. Additionally, the robustness of the chemical bonds formed ensures that the intermediates have excellent shelf-life stability, reducing the risk of inventory write-offs due to expiration.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, moving seamlessly from benchtop flasks to multi-ton reactors without the need for complex engineering modifications or safety overrides. The absence of hazardous reagents like azides or diazo compounds minimizes the safety risks associated with commercial scale-up of complex pharmaceutical intermediates, facilitating faster regulatory approval for manufacturing sites. Furthermore, the aqueous workup procedures generate waste streams that are easier to treat and neutralize compared to those containing heavy metals or persistent organic pollutants, supporting the company's sustainability goals and corporate social responsibility initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized hapten compounds, derived directly from the patent specifications and practical manufacturing experience. Understanding these details is essential for partners looking to integrate these intermediates into their own diagnostic workflows or research pipelines. The answers provided reflect the consensus on best practices for handling, storage, and conjugation to ensure optimal performance of the final immunoassay products.
Q: What is the primary advantage of using tert-leucine in this hapten structure?
A: The incorporation of tert-leucine introduces significant steric bulk and metabolic stability to the hapten molecule, which enhances the specificity of the resulting antibodies during immunoassay development while preventing rapid degradation in biological matrices.
Q: How does this synthesis route improve supply chain reliability for diagnostic manufacturers?
A: By utilizing readily available starting materials such as 5-phenylpyrrole-3-carboxylic acid and standard amino acid derivatives, the process eliminates reliance on exotic or controlled reagents, thereby ensuring consistent availability and reducing lead times for large-scale production.
Q: Is the final carboxylic acid group suitable for protein conjugation?
A: Yes, the final hydrolysis step specifically exposes a terminal carboxylic acid group designed for efficient activation and subsequent amidation with carrier proteins like BSA or OVA, which is critical for generating high-titer antibodies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Synthetic Cannabinoid Hapten Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the hapten is the single most critical factor determining the success of an immunoassay kit, which is why we have invested heavily in mastering the complex chemistry described in patent CN115850146A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global diagnostic leaders without compromising on quality. We operate state-of-the-art rigorous QC labs equipped with advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of synthetic cannabinoid hapten we deliver meets the exacting standards required for clinical and forensic applications.
We invite you to collaborate with us to optimize your supply chain and accelerate your product development timelines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your next generation of high-performance diagnostic solutions.
