Advanced Composite Catalytic Synthesis of Methyl 2-cyano-2-valproate for Commercial Scale-up
Advanced Composite Catalytic Synthesis of Methyl 2-cyano-2-valproate for Commercial Scale-up
The pharmaceutical industry continuously seeks robust, scalable, and cost-efficient pathways for producing critical antiepileptic intermediates. A pivotal advancement in this domain is detailed in patent CN114790151A, which discloses a novel composite catalytic preparation method for methyl 2-cyano-2-valproate. This chemical entity serves as a crucial precursor for valproic acid derivatives and valpromide, widely used in the management of epilepsy and bipolar disorders. The disclosed technology represents a significant departure from legacy methodologies by replacing hazardous strong bases and expensive alkylating agents with a safer, more economical composite catalytic system. By utilizing 1-chloropropane and potassium carbonate under phase transfer conditions, this innovation addresses long-standing challenges regarding raw material costs, operational safety, and environmental compliance. For global procurement teams and R&D directors, understanding the mechanistic nuances and commercial implications of this patent is essential for optimizing supply chains and reducing the cost of goods sold (COGS) in antiepileptic drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of valproic acid intermediates relied heavily on processes established in the early 1980s, such as the method described by Li Xinyuan et al. These conventional routes typically employed methyl cyanoacetate and 1-bromopropane as the alkylating agent, catalyzed by quaternary ammonium salts in the presence of solid potassium carbonate or strong bases like sodium methoxide. While effective, these legacy processes suffer from inherent economic and safety drawbacks. The reliance on 1-bromopropane introduces a significant cost burden due to the higher price of bromide precursors compared to their chloride counterparts. Furthermore, the use of strong alkoxides like sodium methoxide necessitates stringent handling protocols to mitigate fire hazards and moisture sensitivity. From a purity perspective, these older methods often generate complex impurity profiles, including O-alkylated by-products, which complicate downstream purification and reduce overall yield. The environmental footprint is also considerable, as the disposal of bromide-containing waste streams requires specialized treatment, adding to the operational overhead for manufacturers striving for green chemistry compliance.
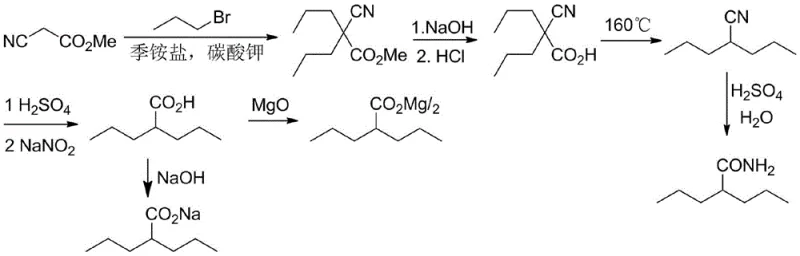
The Novel Approach
In stark contrast, the methodology outlined in patent CN114790151A introduces a transformative approach by utilizing 1-chloropropane, a far more abundant and inexpensive feedstock, in conjunction with a sophisticated composite catalyst system. This new route eliminates the need for hazardous strong bases like sodium methoxide or potassium tert-butoxide, opting instead for the mild and safe potassium carbonate. The core innovation lies in the dual-catalyst system comprising a quaternary ammonium salt (Catalyst A) and an alkali metal halide such as potassium iodide (Catalyst B). This synergy effectively activates the less reactive carbon-chlorine bond, facilitating high-efficiency dipropylation under moderate temperatures ranging from 60°C to 120°C. The result is a reaction profile that not only matches but often exceeds the yields of traditional methods while drastically simplifying the workup procedure. By avoiding the formation of ether by-products common in alkoxide-mediated reactions, this process ensures a cleaner crude product, thereby reducing the load on purification units and enhancing the overall throughput of the manufacturing facility.
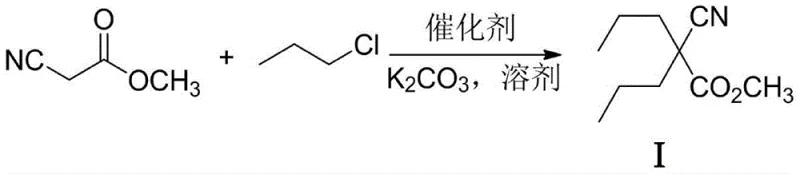
Mechanistic Insights into Composite Catalytic Dipropylation
The success of this novel synthesis hinges on the intricate interplay between the phase transfer catalyst and the halide exchange mediator. In this composite system, the quaternary ammonium salt functions to transport the carbonate anion or the enolate of methyl cyanoacetate into the organic phase where the alkylation occurs. Simultaneously, the addition of a catalytic amount of potassium iodide (KI) or potassium bromide (KBr) facilitates an in-situ halogen exchange, transiently generating the more reactive 1-iodopropane or 1-bromopropane species. This reactive intermediate undergoes rapid nucleophilic substitution with the cyanoacetate anion, regenerating the iodide ion to continue the cycle. This mechanism effectively bypasses the kinetic barrier associated with direct chloropropane alkylation, allowing the reaction to proceed to completion with high selectivity. The use of potassium carbonate as the base is particularly advantageous as it minimizes the risk of ester hydrolysis or decarboxylation during the alkylation step, preserving the integrity of the methyl ester functionality until the dedicated hydrolysis stage. This controlled reactivity is paramount for maintaining high purity specifications required for pharmaceutical-grade intermediates.
Furthermore, the downstream conversion of the intermediate methyl 2-cyano-2-valproate to valpromide demonstrates the robustness of the generated scaffold. As illustrated in the patent data, the intermediate can be seamlessly hydrolyzed to the corresponding acid and subsequently converted to the amide via thermal decarboxylation and acid-catalyzed hydration. The high purity of the initial alkylation product ensures that these subsequent steps proceed without the accumulation of difficult-to-remove impurities. The ability to recover and recycle the inorganic salts, specifically converting waste KCl and KHCO3 into industrial-grade potassium chloride, underscores the circular economy potential of this process. This level of integration between synthesis and waste valorization is increasingly critical for manufacturers aiming to meet stringent environmental regulations while maintaining cost competitiveness in the global market for antiepileptic medications.
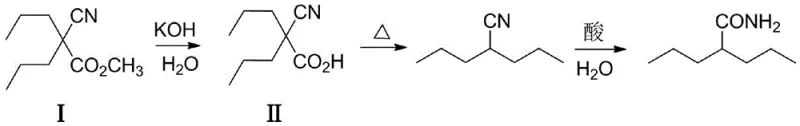
How to Synthesize Methyl 2-cyano-2-valproate Efficiently
Implementing this advanced synthetic route requires precise control over reaction parameters to maximize the benefits of the composite catalytic system. The process begins with the careful selection of solvents such as DMF, DMSO, or ethylene glycol dimethyl ether, which provide the necessary polarity to support the phase transfer mechanism while remaining stable under the reaction conditions. Operators must maintain the reaction temperature within the optimal window of 60°C to 120°C to ensure complete conversion without degrading the sensitive cyano or ester groups. Following the alkylation, the separation of inorganic salts is straightforward, allowing for the direct recovery of the solvent via rotary evaporation. The subsequent hydrolysis and decarboxylation steps are equally critical, requiring controlled pH adjustments and thermal management to ensure high yields of the final valpromide product. For a detailed, step-by-step standard operating procedure tailored to your specific reactor configuration, please refer to the technical guide below.
- Perform composite catalytic dipropylation of methyl cyanoacetate with 1-chloropropane using a quaternary ammonium salt and potassium iodide catalyst system in the presence of potassium carbonate.
- Hydrolyze the resulting methyl 2-cyano-2-valproate ester using aqueous potassium hydroxide to obtain 2-cyano-2-valproic acid.
- Convert the acid to valpromide through thermal decarboxylation followed by acid-catalyzed hydration of the nitrile group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN114790151A offers compelling strategic advantages that extend beyond mere technical feasibility. The primary driver for adoption is the substantial reduction in raw material costs achieved by substituting 1-bromopropane with 1-chloropropane. Given that alkylating agents often constitute a significant portion of the variable costs in fine chemical synthesis, this switch translates directly to improved margins and a more resilient cost structure. Moreover, the elimination of strong bases like sodium methoxide reduces the dependency on hazardous chemical suppliers and lowers the costs associated with special storage and handling infrastructure. This shift not only optimizes the bill of materials but also simplifies the logistics of raw material intake, allowing for more flexible sourcing strategies and reduced inventory holding costs for dangerous goods.
- Cost Reduction in Manufacturing: The transition to a chloride-based alkylation strategy fundamentally alters the economic model of producing valproic acid intermediates. By leveraging the significantly lower market price of 1-chloropropane compared to its bromide analog, manufacturers can achieve drastic reductions in direct material expenses. Additionally, the use of potassium carbonate as a benign base eliminates the need for expensive quenching procedures and specialized corrosion-resistant equipment often required for handling aggressive alkoxides. The high atom economy of the reaction, coupled with the ability to recover and reuse solvents and catalysts, further compounds these savings, resulting in a leaner, more cost-effective production process that enhances overall profitability without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain stability is paramount in the pharmaceutical sector, and this novel process mitigates several key risks. 1-chloropropane is a commodity chemical with a robust global supply network, reducing the vulnerability to shortages that can plague specialized reagents like 1-bromopropane. The simplified reaction conditions also mean that the process is less sensitive to minor fluctuations in reagent quality, ensuring consistent batch-to-batch performance. Furthermore, the ability to recover high-purity potassium chloride from the waste stream creates an additional revenue stream or cost offset, turning a waste disposal liability into a value-generating asset. This circular approach strengthens the supply chain by reducing dependence on external waste management vendors and insulating the operation from fluctuating disposal fees.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, particularly regarding heat management and waste treatment. This composite catalytic method is inherently scalable due to its mild exothermic profile and the use of heterogeneous bases that are easy to filter. The environmental benefits are equally significant; the process avoids the generation of bromide-heavy wastewater, which is notoriously difficult and expensive to treat. Instead, the primary inorganic by-product is potassium chloride, which can be purified to industrial standards and sold or reused. Solvent recovery rates are high, minimizing volatile organic compound (VOC) emissions. These factors collectively streamline the regulatory approval process for new facilities and ensure long-term compliance with increasingly strict environmental protection laws, safeguarding the company's social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this composite catalytic technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for procurement officers negotiating supply contracts. The answers highlight the specific advantages of the chloride-based route and the operational efficiencies gained through the dual-catalyst system.
Q: Why is 1-chloropropane preferred over 1-bromopropane in this synthesis?
A: 1-chloropropane is significantly more abundant and cost-effective than 1-bromopropane. The novel composite catalytic system activates the less reactive chloride sufficiently, eliminating the need for expensive bromide reagents while maintaining high yields.
Q: How does the composite catalyst system improve reaction efficiency?
A: The combination of a quaternary ammonium salt (Phase Transfer Catalyst) and an alkali metal halide (like KI) creates a synergistic effect. This enhances the nucleophilicity of the cyanoacetate anion in the organic phase, allowing the reaction to proceed rapidly under milder conditions with potassium carbonate instead of hazardous strong bases.
Q: What are the waste management benefits of this process?
A: The process generates inorganic salts like KCl and KHCO3 which can be recovered and purified to industrial grade standards. Additionally, the solvent can be recycled via rotary evaporation, drastically reducing chemical waste and environmental impact compared to traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 2-cyano-2-valproate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team has extensively analyzed the methodology described in CN114790151A and possesses the requisite experience to scale diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that achieving the theoretical benefits of this process demands rigorous process control and state-of-the-art equipment. Our facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of methyl 2-cyano-2-valproate meets the exacting standards required for pharmaceutical applications. We are committed to delivering high-purity intermediates that enable our clients to produce safe and effective antiepileptic medications.
We invite you to collaborate with us to optimize your supply chain for valproic acid derivatives. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to our proprietary process optimizations and reliable supply networks. We encourage you to contact us today to request specific COA data and route feasibility assessments. Let us help you leverage this innovative technology to drive down costs and secure your position in the global market for neurological therapeutics.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
