Scalable Silver-Catalyzed Synthesis of Cyanoalkyl Quinones for Advanced Pharmaceutical Intermediates
Scalable Silver-Catalyzed Synthesis of Cyanoalkyl Quinones for Advanced Pharmaceutical Intermediates
The landscape of organic synthesis for high-value intermediates is constantly evolving, driven by the need for more sustainable and scalable methodologies. Patent CN111138315B introduces a significant breakthrough in the field of quinone functionalization, specifically detailing a robust method for synthesizing cyanoalkyl-substituted quinone compounds. This technology leverages a silver-catalyzed radical system to achieve carbon-carbon bond formation under remarkably mild conditions, offering a compelling alternative to traditional photocatalytic routes. The core innovation lies in the utilization of an Ag+/S2O8 2- oxidation system, which effectively generates cyanoalkyl radicals from cyclobutanone oxime precursors. This approach not only broadens the substrate scope to include various substituted benzoquinones and naphthoquinones but also addresses critical industrial pain points regarding cost and scalability. For R&D teams and procurement strategists, this patent represents a pivotal shift towards more economically viable manufacturing processes for complex organic scaffolds used in drug discovery and material science.
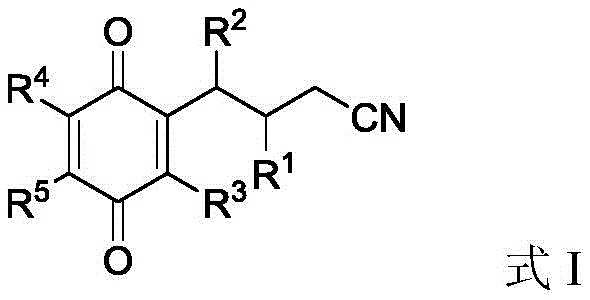
The versatility of this synthetic route is evident in the general structure of the resulting products, as depicted in the patent documentation. The ability to introduce cyanoalkyl chains onto the quinone core opens up new avenues for downstream derivatization, as the nitrile group serves as a versatile handle for further chemical transformations such as hydrolysis to carboxylic acids or reduction to amines. This structural flexibility is paramount for medicinal chemists designing libraries of bioactive molecules. Furthermore, the method operates without the need for specialized photoreactors or high-energy light sources, simplifying the equipment requirements for production facilities. By transitioning from light-driven catalysis to a thermal silver-catalyzed process, the technology aligns perfectly with the industry's demand for reliable pharma intermediates supplier capabilities that can guarantee consistent quality and supply continuity without the bottlenecks associated with photochemical scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of quinone compounds with alkyl groups has relied heavily on transition metal catalysis or, more recently, visible-light photocatalysis. While methods utilizing noble metals like palladium or photocatalysts such as fac-Ir(ppy)3 have demonstrated efficacy in academic settings, they present substantial hurdles for industrial application. The primary limitation of photocatalytic systems is the difficulty in scaling up reactions due to the Beer-Lambert law, where light penetration decreases exponentially with path length, leading to inconsistent reaction rates and poor reproducibility in large vessels. Additionally, the cost of iridium and ruthenium-based photocatalysts is prohibitively high for large-scale manufacturing, significantly impacting the overall cost reduction in electronic chemical manufacturing or pharmaceutical production. These factors combined create a barrier to entry for commercializing molecules that rely on these sophisticated but impractical synthetic routes, often forcing companies to seek alternative, less efficient pathways or abandon promising candidates entirely due to manufacturing complexity.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a cost-effective silver salt catalyst coupled with a persulfate oxidant to drive the cyanoalkylation reaction. This system operates efficiently at temperatures ranging from 0°C to 80°C, with optimal results observed at ambient conditions around 20°C to 30°C. The replacement of expensive photocatalysts with inexpensive silver nitrate drastically lowers the raw material costs, while the use of thermal energy instead of light energy removes the engineering constraints of reactor design. This methodology enables the commercial scale-up of complex polymer additives or pharmaceutical intermediates with unprecedented ease. The reaction tolerates a wide range of functional groups on both the quinone and the oxime components, demonstrating excellent chemoselectivity. By solving the amplification problems inherent to photocatalysis, this technology provides a direct pathway to high-purity OLED material or API intermediates, ensuring that the synthesis remains robust and predictable regardless of the batch size, from gram-scale laboratory experiments to multi-ton commercial production runs.
Mechanistic Insights into Ag+-Catalyzed Radical Cyanoalkylation
The mechanistic pathway of this transformation is a fascinating example of radical chemistry orchestrated by a simple metal salt. The process initiates with the interaction between the silver(I) catalyst and the persulfate oxidant, generating a highly reactive silver(II) species in situ. This high-valent silver complex acts as a single-electron oxidant towards the cyclobutanone oxime carboxylic acid precursor. Upon oxidation, the oxime derivative undergoes a rapid decarboxylation and fragmentation sequence, cleaving the strained cyclobutane ring to release a cyanoalkyl radical along with nitrogen gas and acetone byproducts. This radical generation step is crucial as it avoids the use of hazardous cyanide sources, instead deriving the cyano group from a stable, safe oxime ether scaffold. The generated cyanoalkyl radical is then free to diffuse and attack the electron-deficient double bond of the quinone substrate. This addition step forms a new carbon-carbon bond and a transient radical intermediate on the quinone ring, which is subsequently oxidized and deprotonated to restore aromaticity or quinoid character, yielding the final cyanoalkyl-substituted product. This elegant mechanism highlights the power of redox-active metal catalysis in constructing complex molecular architectures from simple building blocks.
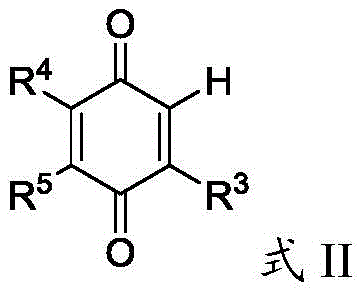
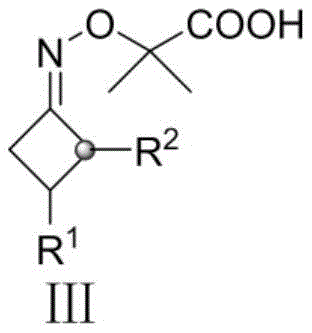
Understanding the impurity profile is critical for any GMP-compliant manufacturing process, and this silver-catalyzed system offers distinct advantages in杂质 control. Since the reaction proceeds via a radical mechanism rather than ionic pathways often prone to rearrangement, the side reactions are minimized. The primary byproducts are gaseous nitrogen and volatile acetone, which are easily removed during the workup procedure, simplifying the purification process. The use of a biphasic solvent system comprising 1,2-dichloroethane and water further aids in partitioning inorganic silver salts and persulfate residues into the aqueous phase, leaving the organic product in the organic layer. This inherent separation capability reduces the burden on downstream purification steps like column chromatography or recrystallization. For quality control teams, this means a cleaner crude product profile and higher overall yields, directly translating to reduced waste generation and lower solvent consumption. The robustness of the radical generation ensures that even with slight variations in reagent stoichiometry, the reaction maintains high selectivity for the desired mono-alkylated product, preventing over-alkylation or polymerization issues that often plague radical reactions.
How to Synthesize Cyanoalkyl Quinones Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory research and pilot plant operations. The procedure involves dissolving the selected quinone compound and the cyclobutanone oxime derivative in a mixture of 1,2-dichloroethane and water under an inert atmosphere to prevent unwanted oxidation of sensitive intermediates. Silver nitrate is added as the catalyst, followed by the slow addition of sodium persulfate as the terminal oxidant. The reaction mixture is then stirred at room temperature for approximately 12 hours, allowing sufficient time for the radical cascade to complete. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during implementation.
- Dissolve the quinone compound and cyclobutanone oxime derivative in a mixed solvent of 1,2-dichloroethane and water under inert gas protection.
- Add silver nitrate catalyst and sodium persulfate oxidant to the mixture and stir at 20-30°C for approximately 12 hours.
- Extract the reaction mixture with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this silver-catalyzed technology offers profound benefits for supply chain resilience and cost management. The shift away from proprietary or scarce photocatalysts to commodity chemicals like silver nitrate and sodium persulfate mitigates the risk of supply disruptions caused by geopolitical factors or limited manufacturer capacity. This stability is crucial for maintaining continuous production schedules for high-purity pharmaceutical intermediates. Furthermore, the elimination of specialized lighting equipment reduces capital expenditure (CAPEX) for new production lines, allowing existing standard stainless steel reactors to be utilized without modification. This flexibility accelerates the time-to-market for new products relying on this chemistry, providing a competitive edge in fast-moving therapeutic areas. The simplified process flow also reduces the operational complexity, requiring less specialized training for plant operators compared to handling sensitive photochemical setups.
- Cost Reduction in Manufacturing: The economic impact of replacing iridium-based photocatalysts with silver salts cannot be overstated. Precious metal photocatalysts often cost thousands of dollars per gram, whereas silver nitrate is available at a fraction of that price, leading to substantial cost savings in raw material procurement. Additionally, the removal of high-energy LED arrays or mercury lamps reduces electricity consumption and maintenance costs associated with bulb replacement and cooling systems. The mild reaction conditions also minimize the need for extreme heating or cooling, further optimizing utility usage. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), enabling more competitive pricing strategies for the final active ingredients or fine chemical products derived from this pathway.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk commodity reagents ensures a stable supply chain that is less susceptible to market volatility. Silver salts and persulfates are produced by multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in environmental parameters, ensuring consistent batch-to-batch quality. This reliability is essential for long-term supply agreements with major pharmaceutical clients who require guaranteed delivery timelines. By simplifying the logistics of raw material sourcing and storage, procurement teams can optimize inventory levels and reduce working capital tied up in expensive or hazardous specialty reagents.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant is often fraught with challenges, particularly for photochemical reactions where light penetration becomes a limiting factor. This thermal silver-catalyzed process scales linearly, meaning that the reaction kinetics observed in the lab are directly translatable to large-scale reactors. This predictability reduces the risk of failed batches during tech transfer. Moreover, the generation of benign byproducts like nitrogen gas and acetone simplifies waste treatment protocols, aiding in compliance with increasingly stringent environmental regulations. The reduced solvent usage and easier purification steps contribute to a greener manufacturing footprint, aligning with corporate sustainability goals and enhancing the company's reputation as a responsible chemical manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyanoalkylation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is vital for project managers evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the Ag+/S2O8 2- system preferred over photocatalysis for quinone functionalization?
A: The Ag+/S2O8 2- system eliminates the need for expensive noble metal photocatalysts like Iridium or Ruthenium complexes and avoids the engineering challenges of scaling up light-dependent reactions, such as poor light penetration in large reactors.
Q: What are the typical reaction conditions for this cyanoalkylation process?
A: The reaction proceeds under mild thermal conditions, typically between 20°C and 30°C, using a biphasic solvent system of 1,2-dichloroethane and water, which facilitates easy workup and product isolation.
Q: Can this method accommodate diverse quinone substrates?
A: Yes, the method demonstrates excellent substrate adaptability, successfully functionalizing benzoquinones, naphthoquinones, and substituted quinones with various electron-donating or withdrawing groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyanoalkyl Quinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the silver-catalyzed cyanoalkylation technology described in CN111138315B. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry to the market. Our state-of-the-art facilities are equipped to handle silver-catalyzed reactions with stringent purity specifications, ensuring that every batch meets the rigorous quality standards demanded by the global pharmaceutical industry. Our dedicated QC labs employ advanced analytical techniques to verify the identity and purity of cyanoalkyl quinone derivatives, guaranteeing that our clients receive materials that are ready for immediate use in downstream synthesis.
We invite you to collaborate with us to leverage this cost-effective and scalable synthetic route for your next project. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this technology can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Together, we can accelerate the development of life-saving medicines and advanced materials through superior chemical manufacturing.
