Advanced Copper-Catalyzed Synthesis of Benzoquinoline Esters for Scalable Pharmaceutical Production
Advanced Copper-Catalyzed Synthesis of Benzoquinoline Esters for Scalable Pharmaceutical Production
The landscape of organic synthesis for nitrogen-containing polycyclic aromatic hydrocarbons is undergoing a significant transformation, driven by the urgent need for greener and more cost-effective manufacturing processes. A pivotal development in this field is detailed in patent CN110054589B, which discloses a novel synthetic method for producing benzoquinoline ester derivatives through a heteroatom-oriented mode. This technology represents a substantial leap forward from traditional methodologies that rely heavily on scarce and expensive noble metals. By leveraging a copper-catalyzed system operating under ambient air conditions, this invention addresses critical pain points in the production of bioactive compounds, including anti-tumor and anti-inflammatory agents. For R&D directors and procurement specialists alike, understanding the mechanistic nuances and commercial implications of this patent is essential for optimizing supply chains and reducing the overall cost of goods sold in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzoquinoline ester compounds has been fraught with challenges that hinder efficient large-scale production. Prior art methods, such as those reported by Sanford in 2007 or the Eiichi group in 2008, predominantly relied on palladium acetate or rhodium catalysts to achieve cross-dehydrocoupling or arylation. These noble metal catalysts are not only prohibitively expensive due to their scarcity but also introduce significant downstream processing burdens, specifically the rigorous requirement to remove trace heavy metal residues to meet stringent pharmaceutical purity standards. Furthermore, many of these legacy processes necessitate the use of highly toxic substrates or strictly anhydrous and anaerobic conditions, which escalate operational costs and safety risks. The reliance on complex synthesis steps often leads to lower overall yields and increased generation of hazardous waste, making these conventional routes economically unviable for the mass production of high-purity pharmaceutical intermediates required by the global market.
The Novel Approach
In stark contrast, the methodology outlined in CN110054589B introduces a paradigm shift by utilizing inexpensive copper catalysts, such as cuprous oxide or copper acetate, in conjunction with a silver salt oxidant. This approach effectively bypasses the need for precious metals like palladium or ruthenium, thereby drastically reducing the raw material expenditure. The reaction proceeds smoothly under an air atmosphere, eliminating the capital and operational expenses associated with inert gas systems and specialized pressure equipment. As illustrated in the reaction scheme below, the process facilitates the direct esterification of quinoline derivatives with organic carboxylic acids, achieving high conversion rates with minimal side reactions. This streamlined workflow not only enhances the atom economy of the transformation but also simplifies the purification protocol, making it an exceptionally attractive route for the commercial scale-up of complex pharmaceutical intermediates.
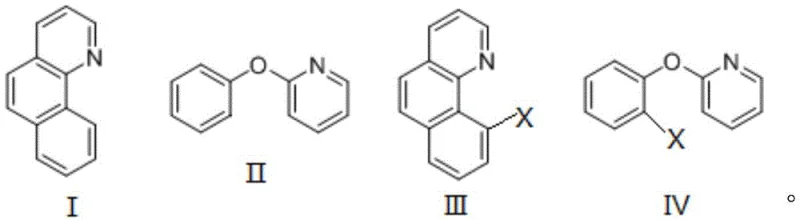
Mechanistic Insights into Heteroatom-Directed C-H Esterification
The core innovation of this technology lies in its exploitation of heteroatom-directed C-H bond activation. The nitrogen atom within the benzo[f]quinoline skeleton (Formula I) or the oxygen atom in 2-phenoxypyridine (Formula II) acts as an intrinsic directing group. This heteroatom coordinates with the copper catalyst, forming a transient cyclic intermediate that brings the metal center into close proximity with the specific C-H bond targeted for functionalization. This chelation-assisted mechanism ensures high regioselectivity, directing the esterification exclusively to the desired position on the aromatic ring without the need for pre-functionalized halides or protecting groups. The silver salt, typically silver carbonate or silver acetate, serves a dual role as both an oxidant to regenerate the active copper species and a promoter for the cleavage of the carboxylic acid O-H bond. This synergistic interaction between the copper catalyst and the silver oxidant drives the oxidative coupling forward efficiently, even in the presence of oxygen from the air, which acts as a benign terminal oxidant in some variations of such mechanisms.
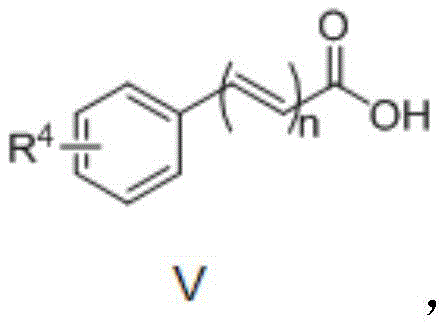
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity intermediates. Because the reaction relies on the specific coordination geometry of the heteroatom, non-specific background reactions are minimized, leading to a cleaner crude reaction profile. The use of copper salts, which are less prone to forming stable organometallic byproducts compared to palladium, further reduces the complexity of the impurity spectrum. The patent data indicates that a wide range of organic carboxylic acids can be employed, including substituted aromatic acids where R4 can be hydrogen, methyl, chloro, or methoxy groups, as well as heteroaromatic and aliphatic acids. This substrate flexibility, depicted in the structural formula below, allows medicinal chemists to rapidly generate diverse libraries of benzoquinoline derivatives for structure-activity relationship (SAR) studies without altering the fundamental robustness of the synthetic platform.
How to Synthesize Benzoquinoline Ester Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The general protocol involves mixing the quinoline derivative substrate with the chosen organic carboxylic acid in a molar ratio ranging from 1:1 to 1:1.8. To this mixture, a catalytic amount of copper source (0.1 to 0.2 equivalents) and a stoichiometric amount of silver salt oxidant (2.0 to 3.0 equivalents) are added. The reaction is conducted in a high-boiling organic solvent such as chlorobenzene, N,N-dimethylformamide, or xylene. The mixture is then heated to a temperature between 130°C and 140°C and stirred for a duration of 18 to 24 hours. Unlike sensitive organometallic reactions, this process does not require rigorous exclusion of moisture or oxygen, significantly lowering the barrier to entry for execution. Following the reaction, the workup involves standard extraction with ethyl acetate, drying, and concentration, followed by purification via silica gel column chromatography using a petroleum ether and ethyl acetate solvent system. Detailed standardized synthesis steps see the guide below.
- Mix quinoline derivatives (benzo[f]quinoline or 2-phenoxypyridine) with organic carboxylic acids, a copper catalyst (e.g., Cu2O), and a silver salt oxidant in an organic solvent like chlorobenzene.
- Heat the reaction mixture to 130°C - 140°C under air atmosphere for 18 to 24 hours to facilitate the C-H esterification.
- Extract the crude product with ethyl acetate, dry over anhydrous sodium sulfate, concentrate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from noble metal catalysis to this copper-based system presents a compelling value proposition centered on cost stability and operational resilience. The elimination of palladium and rhodium removes exposure to the volatile pricing markets associated with these precious metals, ensuring more predictable long-term budgeting for raw materials. Furthermore, the ability to run the reaction under air atmosphere simplifies the engineering controls required for the reactor setup, reducing both capital expenditure for new equipment and maintenance costs for existing infrastructure. The simplified downstream processing, characterized by fewer purification steps and the absence of complex metal scavenging procedures, translates directly into reduced cycle times and higher throughput capacity. These factors collectively contribute to a more agile and cost-efficient supply chain capable of responding rapidly to market demands for key pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with abundant copper salts results in a drastic reduction in catalyst costs per kilogram of product. Since copper is orders of magnitude cheaper than palladium or rhodium, the direct material cost savings are substantial. Additionally, the use of silver carbonate as an oxidant, while a cost factor, is balanced by the high yields achieved (often exceeding 85-90%), which minimizes waste of the valuable starting materials. The overall process economics are further improved by the elimination of specialized ligands often required for palladium catalysis, which are frequently proprietary and costly. This comprehensive reduction in input costs allows for a significantly lower cost of goods sold, enhancing the competitiveness of the final API in the global marketplace.
- Enhanced Supply Chain Reliability: Relying on copper and silver salts mitigates the supply risk associated with geopolitically constrained precious metals. Copper is widely available globally, ensuring a stable and continuous supply of critical catalysts even during market disruptions. The robustness of the reaction conditions, specifically the tolerance to air and moisture, reduces the likelihood of batch failures due to environmental excursions, thereby improving overall production reliability. This stability is crucial for maintaining consistent delivery schedules to downstream pharmaceutical clients who depend on just-in-time inventory models. By securing a synthesis route that is less susceptible to raw material shortages, manufacturers can guarantee supply continuity for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing common solvents and standard heating equipment that are readily available in multi-purpose chemical plants. The absence of highly toxic substrates and the use of less hazardous reagents align with green chemistry principles, simplifying regulatory compliance and waste disposal procedures. Reduced generation of heavy metal waste lowers the environmental footprint of the manufacturing process, which is increasingly a key criterion for supplier selection by major multinational corporations. The straightforward purification via column chromatography or recrystallization facilitates easy scale-up from gram to ton scale without requiring complex process re-engineering, ensuring a smooth transition from R&D to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this heteroatom-directed synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their specific product portfolios. The answers reflect the practical realities of running copper-catalyzed C-H activation processes in an industrial setting.
Q: What are the primary advantages of this copper-catalyzed method over traditional palladium routes?
A: The primary advantage is cost reduction and environmental safety. By replacing expensive noble metal catalysts like palladium or rhodium with abundant copper salts, the process significantly lowers raw material costs. Additionally, operating under air atmosphere eliminates the need for inert gas protection, simplifying operational complexity.
Q: What types of carboxylic acids are compatible with this synthesis protocol?
A: The method demonstrates broad substrate tolerance. It is effective with aromatic acids (such as benzoic acid and its derivatives like 4-chlorobenzoic acid), heteroaromatic acids (like thiophene-2-carboxylic acid), aliphatic acids (pivalic acid), and cinnamic acid derivatives, allowing for diverse functionalization of the benzoquinoline core.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable. The use of cheap catalysts, operation in air, and simple purification steps (extraction and column chromatography) make it ideal for commercial scale-up. The high yields reported (up to 91%) further support its viability for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoquinoline Ester Supplier
The technological advancements described in CN110054589B underscore the potential for producing high-value nitrogen heterocycles with unprecedented efficiency and economic viability. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that the consistency of intermediates like benzoquinoline esters is critical for the efficacy and safety of the final drug product, and our manufacturing protocols are designed to deliver exactly that level of reliability.
We invite potential partners to engage with our technical team to explore how this copper-catalyzed route can be optimized for your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume needs. We encourage you to contact our technical procurement team to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us collaborate to drive down costs and accelerate the development of your next-generation pharmaceutical products through superior chemical manufacturing solutions.
