Scalable Metal-Free Alkenylation of Methyl-Substituted Nitrogen Heterocycles for Commercial Production
Scalable Metal-Free Alkenylation of Methyl-Substituted Nitrogen Heterocycles for Commercial Production
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for greener, more cost-effective, and scalable synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN114507180A, which details a novel method for the self-dehydroalkenylation of methyl-substituted nitrogen heterocyclic compounds via C(sp3)-H bond activation. This technology addresses a critical bottleneck in the synthesis of bis-heteroaryl ethylenes, which are privileged scaffolds found in numerous bioactive molecules, including treatments for leishmaniasis and HIV-1. By leveraging a metal-free catalytic system comprising iodine and organic acid in a specific solvent environment, this process offers a robust alternative to traditional transition-metal catalyzed couplings. For global procurement teams and R&D directors, this represents a tangible opportunity to streamline supply chains for high-purity pharmaceutical intermediates while adhering to stringent environmental and safety standards.
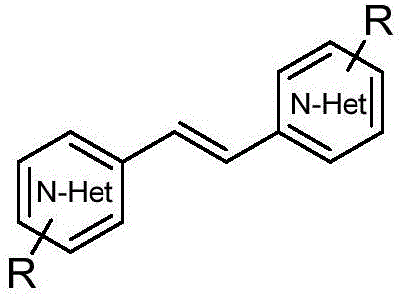
The core innovation lies in the precise control of reaction selectivity. Traditional methods often struggle with over-oxidation or unwanted side reactions when attempting to form carbon-carbon double bonds directly from methyl groups on heterocycles. The patented approach utilizes molecular iodine as an inexpensive oxidant and catalyst, paired with trifluoroacetic acid (TFA) and benzylamine as a crucial additive. This combination, operating under an oxygen atmosphere in dimethyl sulfoxide (DMSO), facilitates the efficient construction of C=C bonds with yields reaching up to 90%. Such high efficiency is paramount for reducing the cost of goods sold (COGS) in the production of complex fine chemicals. Furthermore, the avoidance of precious metals simplifies the downstream purification process, a factor that significantly impacts the lead time for delivering high-purity active pharmaceutical ingredients (APIs) to the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this development, the synthesis of these valuable alkene-bridged heterocycles relied heavily on harsh conditions or expensive catalysts. Literature precedents often employed selenium dioxide (SeO2) or strong base systems like potassium tert-butoxide (KOtBu) with crown ethers. While these methods could achieve the transformation, they presented substantial drawbacks for industrial application. The use of SeO2 introduces toxicity concerns and generates hazardous waste streams that require specialized disposal protocols, increasing the environmental footprint of the manufacturing process. Alternatively, the KOtBu/18-Crown-6 system, while effective, operates under strongly basic conditions that may not be compatible with sensitive functional groups present on advanced intermediates. Moreover, these conventional routes often suffer from moderate atom economy and can produce complex mixtures of byproducts, necessitating rigorous and costly purification steps to meet the purity specifications required by regulatory bodies.
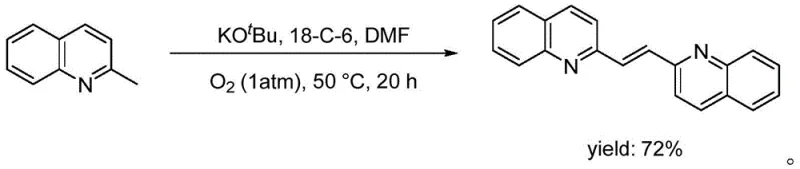
The Novel Approach
The methodology described in CN114507180A fundamentally shifts the paradigm by introducing a mild, metal-free oxidative coupling strategy. By utilizing iodine and TFA in DMSO, the reaction proceeds under relatively温和 conditions (60-100°C) compared to the extreme temperatures or pressures sometimes required by older technologies. The key differentiator is the role of the solvent and additives in directing the reaction pathway. Unlike previous methods that might rely on stoichiometric oxidants generating large amounts of waste, this catalytic cycle uses molecular oxygen as the terminal oxidant, producing water as the primary byproduct. This aligns perfectly with the principles of green chemistry, offering a sustainable route for the commercial scale-up of complex pharmaceutical intermediates. The simplicity of the reagent setup—using commodity chemicals like iodine and benzylamine—also enhances supply chain reliability, as these materials are readily available globally without the geopolitical risks associated with rare earth metals.
Mechanistic Insights into Iodine-Catalyzed Oxidative Coupling
Understanding the mechanistic underpinnings of this transformation is vital for R&D directors aiming to adapt this chemistry to novel substrates. The reaction likely proceeds through an iodine-mediated radical or cationic pathway where the methyl C-H bond is activated. The presence of benzylamine is not merely incidental; it acts as a transient mediator or hydrogen atom transfer agent that facilitates the initial oxidation step. However, the most critical mechanistic feature is the solvent effect. In many oxidative aminations, the amine additive would typically couple with the substrate to form a styryl-amine derivative (cross-deaminoalkenylation). However, in this specific system, the polar aprotic nature of DMSO stabilizes the intermediate species in a way that favors homocoupling (self-dehydroalkenylation) over cross-coupling. This selectivity is a masterstroke of process chemistry, ensuring that the desired bis-heteroaryl ethylene is the major product rather than a mixture of mono-substituted impurities.
Impurity control is inherently built into this mechanism. The patent data explicitly highlights that switching the solvent to toluene, acetonitrile, or methanol drastically alters the product distribution, leading to significant formation of cross-coupled byproducts like 4-styrylquinoline. By strictly adhering to the DMSO protocol, manufacturers can minimize the formation of these structurally similar impurities, which are often difficult to separate via standard crystallization. This inherent selectivity reduces the burden on analytical QC labs and lowers the risk of batch rejection due to out-of-specification impurity profiles. For a reliable pharmaceutical intermediate supplier, this level of process robustness translates directly into consistent quality and reduced operational risk during multi-ton production campaigns.
How to Synthesize Bis-heteroaryl Ethylenes Efficiently
Implementing this synthesis on a pilot or commercial scale requires adherence to specific operational parameters to maximize yield and safety. The process involves mixing the methyl-substituted nitrogen heterocycle with benzylamine, iodine, and trifluoroacetic acid in DMSO, followed by heating under an oxygen atmosphere. The detailed standardized synthesis steps below outline the critical control points identified in the patent examples, ensuring reproducibility from gram to kilogram scale.
- Combine the methyl-substituted nitrogen heterocycle substrate, benzylamine additive, molecular iodine, and trifluoroacetic acid in dimethyl sulfoxide (DMSO) solvent within a reactor.
- Establish an oxygen atmosphere and heat the reaction mixture to a temperature range of 60-100°C, maintaining stirring for 18 to 24 hours to ensure complete conversion.
- Quench the reaction with water, remove excess iodine using saturated sodium thiosulfate, extract with ethyl acetate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-catalyzed methodology offers distinct strategic advantages over traditional transition-metal catalyzed routes. The primary benefit stems from the elimination of expensive and supply-constrained precious metals. By replacing palladium or ruthenium catalysts with commodity iodine, the raw material costs are significantly reduced, and the vulnerability to metal price volatility is mitigated. Furthermore, the absence of heavy metals simplifies the regulatory compliance landscape, as there is no need for extensive testing and validation of residual metal levels in the final API, a process that can be both time-consuming and costly. This streamlined workflow allows for faster batch release and improved cash flow dynamics for manufacturing partners.
- Cost Reduction in Manufacturing: The economic impact of this metal-free process is profound. By utilizing inexpensive reagents like iodine and TFA instead of proprietary ligand-metal complexes, the direct material costs are drastically lowered. Additionally, the high atom economy and the use of oxygen as a green oxidant minimize waste disposal costs. The simplified workup procedure, which avoids complex metal scavenging steps, further reduces processing time and solvent consumption. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final therapeutic more accessible while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, non-strategic raw materials. Iodine and DMSO are produced at a massive global scale, ensuring that production schedules are not disrupted by the shortages that frequently plague the supply of specialized catalysts. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, reducing the rate of failed batches. This reliability ensures a continuous flow of high-purity pharmaceutical intermediates, allowing downstream drug manufacturers to maintain steady production schedules without the fear of raw material bottlenecks.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is highly favorable for scale-up. The reaction operates at moderate temperatures and atmospheric pressure, reducing the energy load and safety risks associated with high-pressure hydrogenation or high-temperature pyrolysis. The generation of water as the main byproduct aligns with increasingly strict environmental regulations regarding waste discharge. The ability to run this reaction safely in large reactors without the need for specialized containment for toxic metals makes it an ideal candidate for rapid capacity expansion to meet surging market demand for these critical building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this oxidative coupling technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process parameters and scope.
Q: Why is DMSO critical as the solvent in this alkenylation process?
A: According to patent CN114507180A, dimethyl sulfoxide (DMSO) is essential to suppress the competing cross-deaminoalkenylation reaction between the substrate and benzylamine. Using solvents like toluene or acetonitrile significantly reduces the yield of the desired self-coupled product and increases impurity formation.
Q: What is the maximum yield achieved for 4-methylquinoline derivatives?
A: The patent data indicates that when 4-methylquinoline is used as the substrate under optimized conditions (80°C, O2 atmosphere), the yield of the self-dehydroalkenylation product can reach up to 90%, demonstrating high efficiency for commercial scale-up.
Q: Does this method require expensive transition metal catalysts?
A: No, this methodology utilizes molecular iodine as the catalyst and trifluoroacetic acid as a promoter. This metal-free approach eliminates the need for costly palladium or ruthenium catalysts and avoids the complex removal of heavy metal residues from the final pharmaceutical intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-heteroaryl Ethylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free C-H activation technologies in modern drug synthesis. Our team of process chemists has extensively evaluated the methodology described in CN114507180A and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to pilot plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of nitrogen heterocycle intermediate we deliver meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cost-effective and sustainable synthetic route for your next project. Whether you require custom synthesis of specific quinoline derivatives or scale-up of existing intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with us can optimize your supply chain and accelerate your time to market.
