Scalable Copper-Catalyzed Synthesis of 5-Aryl-2-Arylseleno-1,3-Oxazole Compounds for Advanced Drug Discovery
Introduction to Patent CN108558785B and Technological Breakthrough
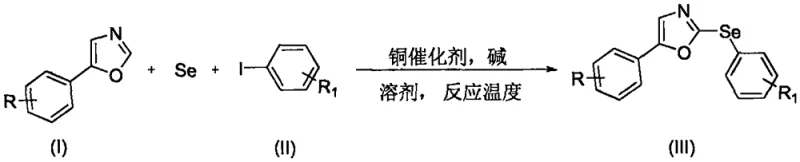
The pharmaceutical and agrochemical industries are constantly seeking efficient routes to access heterocyclic scaffolds containing selenium, given their profound biological activities ranging from anti-inflammatory to anticancer properties. Patent CN108558785B, published in mid-2021, introduces a transformative methodology for synthesizing 5-aryl-2-arylseleno-1,3-oxazole compounds, a class of molecules with significant potential as bioactive leads. This innovation addresses long-standing synthetic challenges by utilizing a copper-catalyzed tandem reaction that directly couples 5-aryl-1,3-oxazoles with aromatic iodides using elemental selenium as the selenium source. Unlike traditional methods that rely on pre-functionalized selenium reagents, this approach leverages abundant commodity chemicals, marking a significant leap forward in atom economy and process simplicity for fine chemical manufacturing.
The core breakthrough lies in the ability to perform a direct C-H bond arylselenylation under relatively mild thermal conditions without the need for exotic ligands or sensitive organometallic reagents. By employing a specific combination of copper chloride and cesium carbonate in dimethylformamide (DMF), the process achieves high yields and purity, making it highly attractive for the production of high-purity pharmaceutical intermediates. This patent not only expands the chemical space available for drug discovery but also provides a robust platform for the commercial scale-up of complex selenium-containing heterocycles, ensuring a reliable supply chain for downstream applications in medicine and crop protection.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
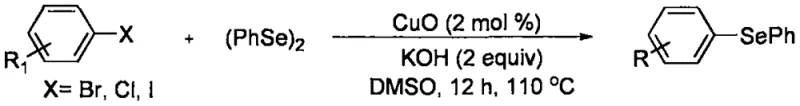
Historically, the synthesis of asymmetric diaryl selenides and related heterocyclic derivatives has been plagued by the reliance on expensive and difficult-to-handle starting materials. Conventional protocols often necessitate the prior preparation of diaryl diselenides or aryl boronic acids, which adds multiple synthetic steps, increases waste generation, and escalates the overall cost of goods. For instance, earlier literature describes copper oxide nanoparticle-catalyzed couplings that require strongly alkaline conditions and pre-synthesized diselenides, creating safety hazards and purification bottlenecks. Furthermore, methods utilizing aryl boronic acids demand the preparation of these moisture-sensitive reagents, which limits the functional group tolerance and complicates the logistics of large-scale manufacturing.
Another significant drawback of legacy technologies is the limited scope of substrates and the harsh reaction conditions often required to drive the transformation. Many existing routes suffer from poor regioselectivity or require stoichiometric amounts of toxic selenium reagents, leading to difficult downstream processing and environmental compliance issues. The inability to use simple, stable aryl halides directly with elemental selenium has been a persistent gap in the field, forcing process chemists to design convoluted linear syntheses that are inefficient and economically unviable for the mass production of specialty chemicals and active pharmaceutical ingredients.
The Novel Approach
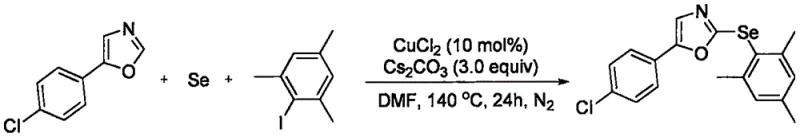
The methodology disclosed in CN108558785B fundamentally reshapes the synthetic landscape by enabling a direct, one-pot assembly of the target oxazole derivatives from three simple components: the oxazole substrate, an aromatic iodide, and elemental selenium powder. This tandem reaction strategy eliminates the need for pre-functionalized selenium reagents, thereby drastically simplifying the supply chain and reducing raw material costs. The use of elemental selenium, a stable and inexpensive solid, contrasts sharply with the volatile and malodorous nature of many traditional selenium sources, enhancing operator safety and facility hygiene during the manufacturing process.
Moreover, this novel approach demonstrates exceptional functional group compatibility, tolerating various substituents on both the oxazole and the aryl iodide rings, including halogens and electron-donating groups. The reaction proceeds efficiently in polar aprotic solvents like DMF at elevated temperatures, providing a practical and scalable solution for industrial applications. By streamlining the synthesis into a single operational step with high atom efficiency, this technology offers a compelling alternative for the cost reduction in pharmaceutical intermediate manufacturing, allowing producers to deliver high-quality materials with a significantly reduced environmental footprint.
Mechanistic Insights into Copper-Catalyzed Tandem Selenylation
The success of this transformation hinges on the synergistic interaction between the copper catalyst, the base, and the elemental selenium within the reaction medium. Mechanistically, the process is believed to initiate with the oxidative addition of the aromatic iodide to the copper center, generating an aryl-copper species that is crucial for the subsequent selenium insertion. The presence of cesium carbonate plays a pivotal role not only in neutralizing the hydrogen iodide byproduct but also in activating the elemental selenium, potentially forming a reactive copper-selenium intermediate that facilitates the transfer of the selenium moiety to the organic framework. This intricate dance of coordination and redox chemistry allows for the cleavage of the robust C-H bond on the oxazole ring, a feat that is typically challenging without directing groups.
Furthermore, the specific choice of copper(II) chloride as the catalyst precursor appears to be critical for maintaining the catalytic cycle's efficiency. Experimental data within the patent reveals that other copper sources, such as copper powder or copper triflate, result in negligible conversion, suggesting that the specific oxidation state and ligand environment provided by the chloride salt are optimal for this tandem sequence. The reaction likely proceeds through a radical or organometallic pathway where the selenium atom is inserted between the two aryl systems, followed by a reductive elimination step that releases the final 5-aryl-2-arylseleno-1,3-oxazole product and regenerates the active copper catalyst for further turnover.
In terms of impurity control, the high selectivity of this copper-catalyzed system minimizes the formation of homocoupling byproducts or over-selenylated species, which are common pitfalls in selenium chemistry. The use of a stoichiometric excess of the aryl iodide and selenium helps drive the reaction to completion while the robust nature of the oxazole core ensures stability under the thermal conditions employed. This mechanistic robustness translates directly to easier purification profiles, as the crude reaction mixtures contain fewer closely related impurities, allowing for straightforward isolation of the target compound via standard chromatographic techniques or crystallization, which is essential for meeting the stringent purity specifications required in the life sciences sector.
How to Synthesize 5-Aryl-2-Arylseleno-1,3-Oxazole Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and environmental controls to maximize yield and reproducibility. The process begins with the careful weighing of 5-aryl-1,3-oxazole, the chosen aromatic iodide, and elemental selenium, typically in a molar ratio favoring the iodide and selenium to ensure complete consumption of the valuable oxazole substrate. These solids are combined with the copper chloride catalyst and cesium carbonate base in a reaction vessel equipped with a nitrogen inlet to maintain an inert atmosphere, which prevents the oxidation of sensitive intermediates and ensures the longevity of the catalyst system throughout the extended heating period.
- Combine 5-aryl-1,3-oxazole, aromatic iodide, elemental selenium, copper chloride catalyst, and cesium carbonate base in an organic solvent such as DMF under nitrogen.
- Heat the reaction mixture to 140°C and stir for approximately 24 hours to facilitate the tandem C-H bond arylselenylation.
- Upon completion, cool the mixture, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio; by replacing specialized, high-cost selenium reagents with commodity-grade elemental selenium and widely available aryl iodides, companies can significantly mitigate supply risk and reduce exposure to price volatility in the fine chemical market. This shift eliminates the dependency on niche suppliers of diselenides, whose production capacity may be limited, thereby enhancing the overall resilience of the supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of multi-step pre-synthesis sequences for selenium reagents. By utilizing elemental selenium directly, manufacturers avoid the labor, energy, and solvent costs associated with preparing diaryl diselenides or boronic acids. Furthermore, the high catalytic efficiency reduces the loading of expensive transition metals, and the simplified workup procedure lowers utility consumption for purification, collectively driving down the cost of goods sold without compromising on the quality of the final active ingredient.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key inputs—elemental selenium, copper salts, and aryl iodides—are produced on a massive global scale for various industries. This abundance ensures that production schedules are not disrupted by shortages of exotic reagents. Additionally, the robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-validation, providing flexibility in sourcing and production planning for global enterprise clients seeking a reliable pharmaceutical intermediate supplier.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior as it avoids the use of malodorous and toxic liquid selenium compounds. The solid nature of elemental selenium simplifies handling and containment, reducing the risk of workplace exposure. Moreover, the high atom economy and reduced waste generation align with green chemistry principles, facilitating easier regulatory approval and lowering the costs associated with waste disposal and environmental remediation, which is increasingly critical for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed selenylation technology. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, aiming to clarify the operational parameters and advantages for potential licensees and manufacturing partners.
Q: Why is elemental selenium preferred over diaryl diselenides in this synthesis?
A: Elemental selenium is significantly cheaper and more readily available as a commodity chemical compared to pre-synthesized diaryl diselenides, which require additional preparation steps and storage precautions, thereby reducing overall raw material costs and supply chain complexity.
Q: What is the critical role of the base in this copper-catalyzed reaction?
A: The patent data indicates that cesium carbonate is uniquely effective; other common bases like potassium carbonate or organic amines resulted in no reaction, suggesting that the specific basicity and solubility profile of cesium carbonate are essential for activating the selenium species and facilitating the C-H activation cycle.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the method utilizes robust, non-sensitive reagents and standard solvents like DMF, avoiding cryogenic conditions or highly air-sensitive catalysts, which makes the transition from laboratory scale to multi-kilogram commercial production feasible with standard stainless steel reactor infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Aryl-2-Arylseleno-1,3-Oxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN108558785B for the development of next-generation therapeutics and agrochemicals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and atmospheric requirements of this copper-catalyzed process, guaranteeing consistent batch-to-batch quality and adherence to stringent purity specifications through our rigorous QC labs.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this advanced chemistry for your pipeline projects. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this streamlined synthesis can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data for related selenium-heterocycles and comprehensive route feasibility assessments, securing a competitive edge in the fast-paced landscape of fine chemical intermediates.
