Advanced Photocatalytic Synthesis of Homoallyltrifluoromethyl Compounds for Commercial Scale-up
The landscape of fluorine chemistry has evolved dramatically, driven by the critical role trifluoromethyl groups play in enhancing the metabolic stability and lipophilicity of bioactive molecules. A groundbreaking advancement in this field is detailed in Chinese Patent CN109776324B, which discloses a highly efficient preparation method for homoallyltrifluoromethyl compounds. This technology represents a paradigm shift from traditional thermal catalysis to visible-light-driven photocatalysis, offering a greener and more economically viable pathway for constructing benzyl α-trifluoromethyl tertiary or quaternary carbon centers. For R&D directors and procurement strategists in the pharmaceutical and agrochemical sectors, this innovation addresses long-standing challenges regarding reagent cost, reaction severity, and substrate availability. By leveraging a radical cation mechanism driven by single-electron oxidation, this process enables the direct fluoroallylation of electron-rich gem-difluoroolefins without the need for cumbersome pre-functionalization, positioning it as a superior choice for the reliable homoallyltrifluoromethyl compound supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
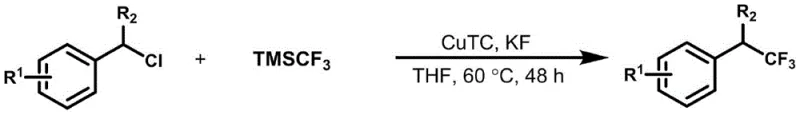
The Limitations of Conventional Methods
Historically, the construction of α-trifluoromethylbenzyl structures has been fraught with synthetic hurdles that impede cost reduction in pharmaceutical intermediate manufacturing. Traditional strategies often rely on the use of expensive and hazardous trifluoromethylating agents or require complex multi-step sequences to install the trifluoromethyl group prior to coupling. As illustrated in prior art strategies, such as those utilizing pre-functionalized α-trifluoromethylbenzyl substrates for coupling reactions, the necessity for specific activating groups on the aromatic ring severely limits the substrate scope. Furthermore, alternative palladium-catalyzed nucleophilic attacks often demand additional electron-withdrawing structures to stabilize intermediate carbanions, rendering electron-rich substrates unreactive. These conventional pathways not only inflate raw material costs but also generate significant waste streams due to low atom economy and the requirement for stoichiometric amounts of heavy metal catalysts, creating substantial burdens for supply chain sustainability and environmental compliance.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN109776324B introduces a streamlined photocatalytic fluoroallylation strategy that fundamentally simplifies the synthetic architecture. This novel approach utilizes inexpensive and commercially available gem-difluoroolefins and allyl sulfones as starting materials, reacting them under mild room temperature conditions driven solely by visible light energy. The elimination of pre-functionalization steps drastically shortens the synthetic route, thereby reducing both time and resource consumption. By employing a photocatalyst with an extremely low loading of merely 0.2 mol%, the process achieves high efficiency while minimizing the presence of residual metal impurities in the final product. This method effectively overcomes the electronic limitations of previous techniques, allowing for the successful transformation of electron-rich substrates that were previously inaccessible, thus expanding the chemical space available for drug discovery and development.
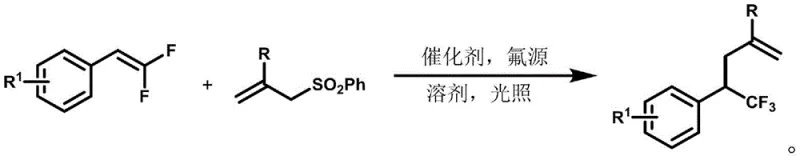
Mechanistic Insights into Photocatalytic Fluoroallylation
The mechanistic elegance of this transformation lies in its unique radical cation pathway, which diverges significantly from standard anionic or neutral mechanisms. Upon irradiation with blue LED light, the photocatalyst enters an excited state capable of performing a single-electron oxidation on the electron-rich gem-difluoroolefin substrate. This oxidation event generates a radical cation intermediate, which significantly enhances the electrophilicity of the olefinic bond and facilitates the subsequent nucleophilic attack by fluoride ions derived from the fluorine source. This step is critical as it generates a benzyl α-trifluoromethyl radical species in situ, a highly reactive intermediate that would be difficult to access via traditional ionic pathways. The resulting radical then engages in a rapid coupling reaction with the allyl sulfone fragment, driven by the thermodynamic stability of the newly formed carbon-carbon bond and the expulsion of the sulfonyl leaving group. This cascade ensures high regioselectivity and chemoselectivity, preventing side reactions that typically plague radical chemistry.
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity homoallyltrifluoromethyl compounds. The mild reaction conditions prevent thermal degradation of sensitive functional groups often present in complex pharmaceutical intermediates. Moreover, the use of triethylamine trihydrofluoride as the fluorine source provides a controlled release of fluoride ions, mitigating the risk of uncontrolled polymerization or decomposition that can occur with stronger, more corrosive fluoride sources. The radical nature of the reaction also means that the process is less sensitive to trace moisture compared to strictly anionic organometallic methods, simplifying the operational requirements for maintaining anhydrous conditions. Consequently, the resulting crude product profile is cleaner, requiring less intensive purification downstream, which directly translates to higher overall yields and reduced solvent usage in the isolation phase.
How to Synthesize Homoallyltrifluoromethyl Compound Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The procedure involves the sequential addition of the gem-difluoroolefin, allyl sulfone, fluorine source, and photocatalyst into a suitable solvent such as acetonitrile or dichloromethane under an inert nitrogen or argon atmosphere. The reaction mixture is then subjected to irradiation from a standard 8W blue LED light source at a wavelength of 450nm, maintaining the system at ambient temperature for a duration ranging from 24 to 96 hours depending on the specific substrate reactivity. This straightforward setup eliminates the need for specialized high-pressure or high-temperature equipment, making it accessible for widespread adoption. For detailed standardized synthesis steps and specific molar ratios optimized for different substrates, please refer to the guide below.
- Prepare the reaction mixture by sequentially adding gem-difluoroolefin, allyl sulfone, a fluorine source (triethylamine trihydrofluoride), and a photocatalyst into a solvent like acetonitrile under an inert atmosphere.
- Maintain the molar ratio of reactants at approximately 1: 3:2:0.002 (substrate:allyl sulfone:fluorine source:catalyst) to ensure optimal conversion efficiency.
- Irradiate the mixture with blue LED light (450nm) at room temperature for 24 to 96 hours, then isolate the target homoallyltrifluoromethyl compound from the reaction product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift towards light-driven chemistry aligns perfectly with global trends towards sustainable manufacturing, reducing the carbon footprint associated with heating large-scale reactors. By replacing expensive transition metal catalysts and complex pre-functionalized building blocks with commodity chemicals, manufacturers can achieve substantial cost savings in raw material procurement. The operational simplicity of running reactions at room temperature further reduces energy consumption and infrastructure wear-and-tear, contributing to a lower total cost of ownership for the production facility. Additionally, the robustness of the method against varying electronic properties of substrates ensures a consistent supply of diverse intermediates, mitigating the risk of production bottlenecks caused by difficult-to-synthesize precursors.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic reduction in catalyst loading and the use of low-cost reagents. Traditional methods often require stoichiometric amounts of precious metals or expensive fluorinating agents, whereas this method utilizes a photocatalyst at only 0.2 mol% and triethylamine trihydrofluoride, which is inexpensive and non-corrosive to glassware. This significant decrease in reagent costs, combined with the elimination of pre-functionalization steps, streamlines the entire production workflow. Furthermore, the avoidance of harsh conditions reduces the need for specialized corrosion-resistant equipment, lowering capital expenditure requirements for new production lines dedicated to fluorine chemistry.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on readily available starting materials such as gem-difluoroolefins and allyl sulfones, which are easier to source in bulk quantities compared to specialized trifluoromethylated building blocks. The mild reaction conditions also imply a safer working environment, reducing the regulatory hurdles and insurance costs associated with handling hazardous high-energy reagents. This stability allows for more predictable production scheduling and shorter lead times for high-purity homoallyltrifluoromethyl compounds, ensuring that downstream API synthesis is not delayed by intermediate shortages. The versatility of the method also means that a single production line can be easily adapted to synthesize a wide variety of analogues, providing flexibility in responding to changing market demands.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but the use of efficient LED arrays and flow chemistry compatibility makes this process highly scalable from gram to tonne levels. The green nature of the process, characterized by the use of light as a traceless reagent and the generation of minimal waste, simplifies environmental compliance and waste disposal procedures. The absence of heavy metal residues in the final product reduces the burden on purification teams to meet stringent regulatory limits for metal impurities in pharmaceutical ingredients. This alignment with green chemistry principles not only enhances the corporate sustainability profile but also future-proofs the manufacturing process against increasingly strict environmental regulations globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic fluoroallylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, aiming to clarify the practical implications for industrial application. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this method into existing manufacturing portfolios. The answers highlight the balance between innovative chemistry and practical engineering considerations necessary for successful commercialization.
Q: What are the key advantages of this photocatalytic method over traditional trifluoromethylation?
A: Unlike traditional methods that require expensive trifluoromethylating agents or difficult pre-functionalization steps, this novel approach utilizes readily available gem-difluoroolefins and allyl sulfones. It operates under mild room temperature conditions using visible light, eliminating the need for harsh thermal conditions or expensive transition metal catalysts often found in palladium-catalyzed routes.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is highly amenable to scale-up. The use of low-cost raw materials, a minimal catalyst loading of only 0.2 mol%, and energy-efficient LED light sources significantly reduces operational costs. Furthermore, the reaction proceeds at room temperature, simplifying thermal management requirements for large reactors compared to high-temperature processes.
Q: What types of substrates are compatible with this fluoroallylation strategy?
A: The method demonstrates broad substrate scope, tolerating various functional groups on the aromatic ring of the gem-difluoroolefin, including electron-donating groups like methoxy and alkyl, as well as electron-withdrawing groups like halogens and esters. It also accommodates complex structures such as naphthalene, morpholine, and tocopherol derivatives, making it versatile for diverse pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homoallyltrifluoromethyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photocatalytic synthesis method described in CN109776324B for the next generation of fluorinated pharmaceuticals and agrochemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity homoallyltrifluoromethyl compounds that adhere to the highest quality standards, supporting your R&D efforts with reliable and consistent supply.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this novel route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate the development of life-saving medicines and innovative agricultural solutions through superior chemical manufacturing.
