Advanced Metal-Free Synthesis of Isoquinoline Substituted Flavones for Commercial Pharmaceutical Applications
Advanced Metal-Free Synthesis of Isoquinoline Substituted Flavones for Commercial Pharmaceutical Applications
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more cost-effective synthetic methodologies. A pivotal advancement in this domain is detailed in Chinese Patent CN110790752B, which discloses a novel, metal-free approach for synthesizing (iso)quinoline and quinoxaline substituted flavone and quinolone derivatives. This technology represents a paradigm shift from traditional transition metal-catalyzed cross-coupling reactions to a highly efficient, acid-catalyzed one-pot cyclization strategy. For R&D directors and procurement specialists alike, this innovation offers a compelling solution to the persistent challenges of catalyst toxicity, high raw material costs, and complex purification protocols associated with legacy methods. By leveraging readily available alkynone compounds and nitrogen oxide heterocycles, this process delivers high-purity scaffolds essential for anticancer and antibacterial drug development without the environmental burden of heavy metal waste.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of complex heterocyclic systems like isoquinoline-substituted flavones has relied heavily on palladium-catalyzed cross-coupling reactions, such as Stille or Suzuki-Miyaura couplings. As illustrated in the prior art, these conventional pathways often necessitate the use of stoichiometric amounts of organotin reagents or boronic acids alongside expensive palladium catalysts. 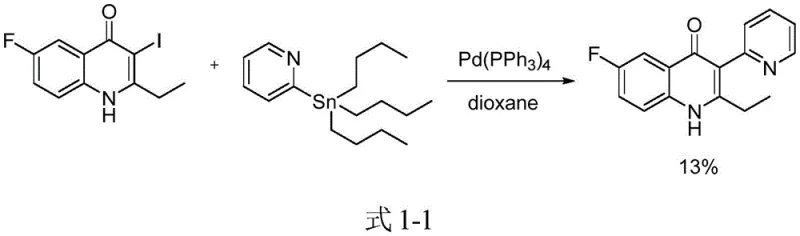 These traditional methods suffer from inherent drawbacks, including low overall yields, harsh reaction conditions, and the generation of hazardous heavy metal byproducts that are difficult to remove to pharmaceutical grade standards. Furthermore, the synthesis of the requisite halogenated starting materials adds additional steps and cost to the supply chain, creating bottlenecks in production scalability. The reliance on precious metals also introduces significant volatility in manufacturing costs, making long-term supply planning difficult for procurement managers seeking stability.
These traditional methods suffer from inherent drawbacks, including low overall yields, harsh reaction conditions, and the generation of hazardous heavy metal byproducts that are difficult to remove to pharmaceutical grade standards. Furthermore, the synthesis of the requisite halogenated starting materials adds additional steps and cost to the supply chain, creating bottlenecks in production scalability. The reliance on precious metals also introduces significant volatility in manufacturing costs, making long-term supply planning difficult for procurement managers seeking stability.
The Novel Approach
In stark contrast, the methodology described in patent CN110790752B utilizes a direct, metal-free cyclization strategy that dramatically simplifies the synthetic workflow. The core innovation involves the reaction of alkynone compounds with isoquinoline, quinoline, or quinoxaline N-oxides under acidic conditions to form the target flavone or quinolone skeletons in a single pot. 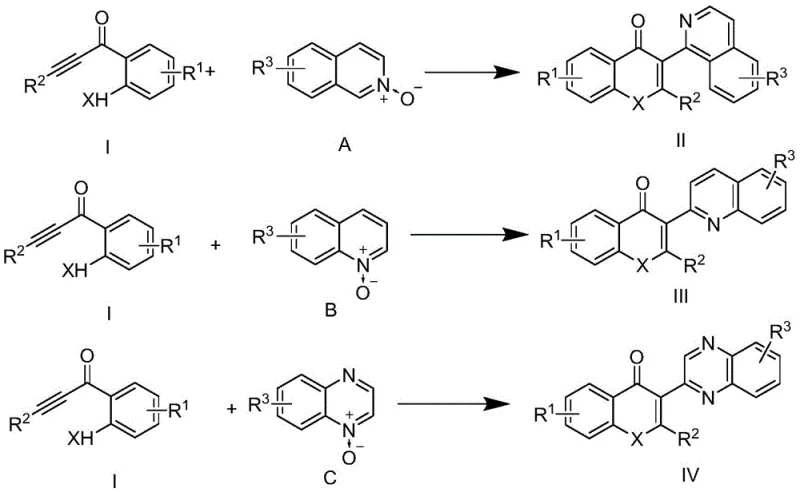 This approach completely bypasses the need for transition metal catalysts, thereby eliminating the risk of metal contamination in the final active pharmaceutical ingredient (API). The reaction conditions are remarkably mild yet effective, typically employing common organic solvents like DMF and concentrated hydrochloric acid at temperatures ranging from 100 °C to 140 °C. This not only reduces the complexity of the operation but also enhances the safety profile of the manufacturing process, aligning perfectly with modern green chemistry principles and regulatory expectations for impurity control.
This approach completely bypasses the need for transition metal catalysts, thereby eliminating the risk of metal contamination in the final active pharmaceutical ingredient (API). The reaction conditions are remarkably mild yet effective, typically employing common organic solvents like DMF and concentrated hydrochloric acid at temperatures ranging from 100 °C to 140 °C. This not only reduces the complexity of the operation but also enhances the safety profile of the manufacturing process, aligning perfectly with modern green chemistry principles and regulatory expectations for impurity control.
Mechanistic Insights into Acid-Catalyzed Cyclization
The mechanistic pathway of this transformation is rooted in the unique reactivity of heterocyclic N-oxides under acidic activation. In the presence of a proton source like HCl, the N-oxide moiety becomes highly electrophilic, facilitating a nucleophilic attack by the electron-rich alkyne system of the alkynone substrate. This initial interaction triggers a cascade of intramolecular cyclization events that construct the fused ring system characteristic of flavones and quinolones. The absence of metal coordination complexes means the reaction trajectory is governed purely by electronic and steric factors, allowing for a broader tolerance of functional groups on the aromatic rings. This mechanistic simplicity is crucial for R&D teams, as it minimizes the formation of side products often associated with metal-mediated radical pathways, resulting in cleaner reaction profiles and higher crude purity.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for regulatory compliance. Traditional palladium-catalyzed routes often leave trace residues of heavy metals that require extensive downstream processing, such as scavenging or recrystallization, to meet ICH Q3D guidelines. By avoiding these catalysts entirely, the new process inherently produces an impurity profile that is easier to characterize and control. The primary byproducts are typically organic salts or unreacted starting materials that can be removed through standard aqueous workups or silica gel chromatography. This streamlined purification capability ensures that the final high-purity pharmaceutical intermediates meet stringent quality specifications with minimal processing effort, directly translating to improved batch consistency and reduced production lead times.
How to Synthesize Isoquinoline Substituted Flavones Efficiently
Implementing this synthetic route in a laboratory or pilot plant setting is straightforward due to its one-pot nature and the use of commodity chemicals. The general procedure involves dissolving the alkynone and the heterocyclic N-oxide in a polar aprotic solvent, followed by the careful addition of concentrated hydrochloric acid. The mixture is then heated to the optimal temperature, typically around 140 °C, and monitored via TLC until completion. For instance, in a representative embodiment, reacting 1-(2-hydroxyphenyl)-3-phenylprop-2-yn-1-one with isoquinoline N-oxide in DMF at 140 °C for 2 hours yielded the target flavone derivative with an impressive 88% isolated yield. 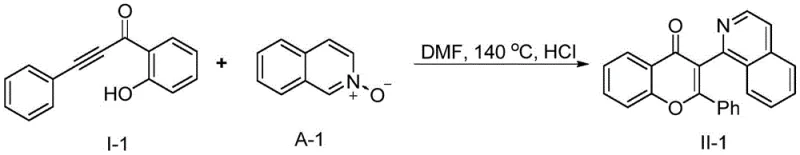 Following the reaction, the solvent is recovered via distillation, and the crude product is purified using standard silica gel column chromatography. Detailed standardized synthesis steps are provided in the guide below.
Following the reaction, the solvent is recovered via distillation, and the crude product is purified using standard silica gel column chromatography. Detailed standardized synthesis steps are provided in the guide below.
- Mix alkynone compounds and isoquinoline nitrogen oxides in an organic solvent such as DMF.
- Add concentrated hydrochloric acid to the reaction mixture to initiate catalysis.
- Heat the reaction to 100-140 °C for 2-6 hours, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology presents a multitude of strategic benefits that extend far beyond simple chemical efficiency. The elimination of precious metal catalysts fundamentally alters the cost structure of manufacturing these valuable intermediates. By removing the dependency on palladium and toxic tin reagents, companies can achieve substantial cost savings in raw material procurement while simultaneously reducing the capital expenditure associated with specialized waste treatment facilities. This shift not only lowers the direct cost of goods sold but also mitigates the financial risks associated with the volatile pricing of precious metals in the global market.
- Cost Reduction in Manufacturing: The most immediate impact of this technology is the drastic reduction in manufacturing costs driven by the exclusion of expensive transition metal catalysts. Traditional methods require significant investment in palladium sources and ligand systems, which constitute a major portion of the variable costs in fine chemical synthesis. Furthermore, the downstream costs associated with removing trace metal impurities to meet pharmaceutical standards are completely eradicated. This allows for a leaner production model where resources can be reallocated to capacity expansion or quality assurance initiatives, ultimately enhancing the overall profitability of the product line without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of readily available starting materials and simplified logistics. Alkynone compounds and heterocyclic N-oxides are commercially accessible bulk chemicals, unlike specialized organometallic reagents which may have limited suppliers and long lead times. The robustness of the reaction conditions also means that production is less susceptible to disruptions caused by the scarcity of specific catalysts or ligands. This reliability ensures a consistent flow of high-purity pharmaceutical intermediates to downstream customers, minimizing the risk of stockouts and enabling more accurate demand forecasting and inventory management strategies.
- Scalability and Environmental Compliance: Scaling this process from benchtop to commercial production is inherently safer and more environmentally compliant. The one-pot design reduces the number of unit operations, minimizing solvent usage and energy consumption per kilogram of product. Additionally, the absence of heavy metal waste simplifies effluent treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This green chemistry profile not only reduces the environmental footprint of the manufacturing site but also enhances the corporate sustainability narrative, which is becoming a critical factor in vendor selection for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this innovative synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of adopting this technology for your specific application needs.
Q: What are the primary advantages of this metal-free synthesis over traditional coupling methods?
A: This method eliminates the need for expensive and toxic transition metal catalysts like palladium and tin reagents, significantly reducing raw material costs and simplifying the purification process by removing heavy metal residues.
Q: What are the typical reaction conditions required for this cyclization?
A: The reaction typically proceeds in polar aprotic solvents like DMF at temperatures between 100 °C and 140 °C, using concentrated hydrochloric acid as the catalyst for a duration of 2 to 6 hours.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the one-pot nature of the reaction, combined with readily available starting materials and the absence of sensitive metal catalysts, makes it highly scalable and robust for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinoline Substituted Flavones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route for the next generation of therapeutic agents. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of isoquinoline substituted flavones delivered meets the highest industry standards for safety and efficacy.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your time to market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the competitive pharmaceutical landscape.
