Scalable Synthesis of Polysubstituted 2-Aryl Indole Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks efficient pathways to construct privileged scaffolds that serve as the backbone for new therapeutic agents. Among these, the indole nucleus stands out as a critical structure found in numerous bioactive molecules, including the estrogen antagonist Zindoxifene. A recent technological breakthrough, detailed in patent CN111004164B, introduces a highly effective preparation method for polysubstituted 2-aryl indole derivatives. This innovation leverages a copper-catalyzed oxidative cyclization strategy that transforms readily available 2-ethynyl aniline compounds and ketone derivatives into valuable intermediates. By utilizing a synergistic system of copper salts, specific ligands, and nitroxide radical oxidants, this process achieves superior chemical and regioselectivity. For R&D directors and procurement specialists, this represents a significant shift away from reliance on expensive noble metals towards a more sustainable and cost-effective base metal catalysis platform.
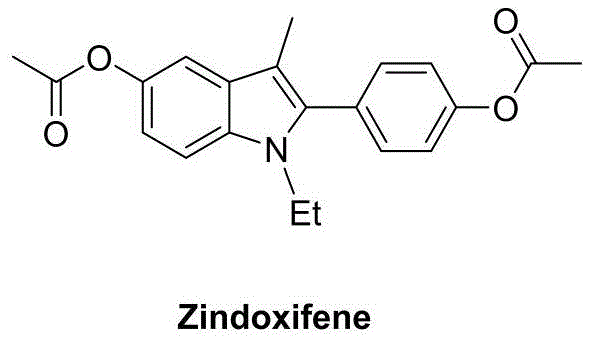
The historical landscape of synthesizing 2-aryl indoles has been fraught with significant economic and operational challenges that hinder large-scale adoption. Conventional methodologies often rely on the use of precious metal catalysts such as palladium or iridium, which not only inflate raw material costs but also introduce stringent requirements for heavy metal removal in the final API. For instance, earlier reports described iron and palladium co-catalyzed intramolecular hydroamination followed by Michael addition, a route plagued by the use of toxic methyl vinyl ketone and limited product structural diversity. Similarly, iridium-catalyzed approaches require specialized ligands that are difficult to prepare repeatedly, creating supply chain bottlenecks. Other copper-catalyzed attempts using boronic acids have suffered from low efficiencies and narrow substrate scopes, failing to meet the rigorous demands of modern process chemistry for diverse analog generation.
In stark contrast, the novel approach disclosed in the patent data offers a streamlined solution that directly addresses these legacy inefficiencies. The new methodology employs a robust copper catalytic system, utilizing accessible salts like copper acetate or cuprous chloride paired with common ligands such as 2,2'-bipyridine. The reaction proceeds through an oxidative cyclization mechanism using stable oxidants like 4-OH-TEMPO, eliminating the need for hazardous reagents. This transition allows for the direct coupling of 2-alkynylanilines with simple ketones under relatively mild thermal conditions, typically around 120 °C. The result is a versatile synthetic route that maintains high yields while drastically simplifying the operational workflow, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.

Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific targets. The reaction operates through a copper-mediated activation of the alkyne moiety in the 2-ethynyl aniline substrate, facilitating a nucleophilic attack by the amine nitrogen. The presence of the oxidant, specifically the TEMPO radical species, plays a pivotal role in the rearomatization step, ensuring the formation of the stable indole core without over-oxidation or degradation of sensitive functional groups. The choice of ligand, such as 1,10-phenanthroline or tetramethylethylenediamine, fine-tunes the electronic environment around the copper center, enhancing the turnover frequency and stabilizing the active catalytic species throughout the extended reaction times of 24 to 36 hours.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or acid-catalyzed alternatives. The high regioselectivity inherent in the copper-ligand complex ensures that the cyclization occurs exclusively at the desired position, minimizing the generation of regioisomers that are notoriously difficult to separate. Furthermore, the mild oxidative conditions prevent the polymerization of the alkyne starting material, a common side reaction in harsher environments. This clean reaction profile translates directly to reduced purification burdens, as the crude reaction mixtures contain fewer byproducts, allowing for more efficient isolation of the target polysubstituted 2-aryl indole derivatives via standard chromatographic techniques.
How to Synthesize Polysubstituted 2-Aryl Indole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters identified in the patent examples to ensure reproducibility and high yield. The process begins with the precise weighing of the 2-alkynyl aniline and the ketone substrate, typically in a molar ratio ranging from 1:1 to 1:2, to drive the equilibrium towards product formation. These reagents are combined with the copper catalyst (5-40 mol%) and ligand (10-20 mol%) in a high-boiling solvent like 1,2-dichloroethane or chlorobenzene. The addition of the oxidant, usually 1.0 equivalent of 4-OH-TEMPO, is critical for the success of the oxidative cyclization. The following guide outlines the standardized operational procedure derived from the most successful experimental embodiments.
- Dissolve 2-alkynyl aniline compound, ketone compound, copper catalyst, ligand, and oxidant in a suitable solvent such as 1,2-dichloroethane.
- Heat the reaction mixture at a temperature between 60-150 °C, preferably around 120 °C, for a duration of 12 to 48 hours to ensure complete conversion.
- Upon completion, recover the solvent by concentration under reduced pressure and purify the crude product via column chromatography to isolate the target indole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed protocol offers substantial strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs associated with catalyst procurement. By replacing scarce and volatile noble metals like palladium and iridium with abundant copper salts, manufacturers can insulate their production budgets from the fluctuations of the precious metals market. Additionally, the use of commodity chemicals such as acetophenone derivatives and simple anilines ensures a stable and diversified supply base, reducing the risk of single-source dependency that often plagues specialized synthetic routes.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and complex, custom-synthesized ligands leads to significant savings in the bill of materials. Furthermore, the simplified workup procedure, which avoids the need for specialized scavengers to remove trace heavy metals, reduces the consumption of auxiliary processing materials. This streamlined approach lowers the overall cost of goods sold (COGS) for the final intermediate, providing a competitive edge in pricing negotiations with downstream API manufacturers.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial solvents and reagents enhances the resilience of the supply chain. Unlike methods requiring air-sensitive reagents or strictly anhydrous conditions, this process tolerates standard laboratory conditions, simplifying logistics and storage requirements. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by minor variations in raw material quality or environmental factors, ensuring consistent on-time delivery for clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in batch reactors with straightforward thermal control. From an environmental standpoint, the avoidance of toxic reagents like methyl vinyl ketone and the use of less hazardous copper salts align with green chemistry principles. This facilitates easier regulatory compliance and waste management, as the effluent streams are less contaminated with persistent organic pollutants or toxic heavy metals, thereby reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of the method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects.
Q: What are the primary advantages of this copper-catalyzed method over traditional palladium or iridium routes?
A: This method utilizes inexpensive copper salts instead of costly noble metals like palladium or iridium. It avoids toxic reagents such as methyl vinyl ketone and eliminates the need for complex, repeatedly prepared ligands, significantly lowering raw material costs and operational complexity.
Q: What is the typical substrate scope for this oxidative cyclization reaction?
A: The process demonstrates high chemical and regioselectivity across a broad range of substrates. It accommodates various substituted 2-alkynyl anilines (with groups like fluoro, chloro, methoxy) and diverse ketone compounds, allowing for the synthesis of complex polysubstituted indole frameworks essential for drug discovery.
Q: How does this process impact impurity profiles and purification efforts?
A: The high regioselectivity of the copper-catalyzed system minimizes the formation of structural isomers and side products. This results in a cleaner reaction profile, which simplifies downstream purification steps like column chromatography and reduces the overall burden on quality control laboratories.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 2-Aryl Indole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex copper-catalyzed transformations allows us to offer reliable supply solutions for even the most challenging indole-based scaffolds.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall project costs.
