Advanced Base-Catalyzed Synthesis of Dimethyl 4-Methoxymethylene-2-Glutaconate for Pharma
The landscape of pharmaceutical and agrochemical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is detailed in patent CN109293507B, which introduces a novel method for synthesizing dimethyl 4-methoxymethylene-2-glutaconate. This compound is not merely a standalone chemical entity but serves as a pivotal building block for nicotinic acetylcholine receptor agonists and, crucially, as a reference standard for monitoring impurities in the production of the insecticide flonicamid. The patented technology shifts the paradigm from traditional, resource-intensive extraction methods to a streamlined base-catalyzed self-condensation reaction. By leveraging methyl 3-methoxyacrylate as a starting material, this approach addresses long-standing challenges related to yield, reaction time, and impurity profiles, offering a robust solution for reliable pharmaceutical intermediate supplier networks seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dimethyl 4-methoxymethylene-2-glutaconate has relied heavily on the ring-opening of coumaric acid derivatives, a process fraught with inefficiencies and economic drawbacks. As illustrated in prior art, traditional methods often involve reacting coumaric acid with methanol under acidic conditions or using acetyl chloride as a catalyst. These legacy pathways suffer from prolonged reaction times, typically ranging from 10 to 48 hours, which severely bottlenecks production throughput. Furthermore, the reliance on coumaric acid, often derived from plant extracts, introduces variability in raw material costs and availability. A critical technical failure of these acidic routes is the generation of stubborn impurities; for instance, acid-catalyzed ring-opening frequently yields isomeric byproducts and addition impurities that are chemically similar to the target molecule, making purification arduous and reducing overall yield to as low as 48% in some cases. The use of large excesses of reagents like trimethyl orthoformate further complicates downstream processing and raises environmental concerns due to waste generation.
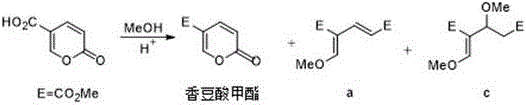
The Novel Approach
In stark contrast, the methodology disclosed in CN109293507B revolutionizes the synthesis by employing a base-catalyzed self-condensation of methyl 3-methoxyacrylate. This innovative route bypasses the need for expensive natural precursors and harsh acidic environments entirely. The reaction proceeds rapidly, achieving completion within just 3 hours under mild thermal conditions (35-40°C), representing a drastic improvement in operational efficiency. By utilizing industrial-grade methyl 3-methoxyacrylate, the process ensures a consistent and cost-effective supply of starting materials. Most importantly, this alkaline condensation pathway exhibits superior selectivity, effectively suppressing the formation of the isomeric and addition impurities that plague conventional methods. The result is a cleaner reaction profile with yields reaching up to 65.5%, significantly enhancing the economic viability of producing this high-purity pharmaceutical intermediate for commercial applications.
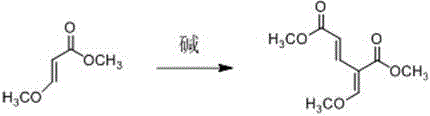
Mechanistic Insights into Base-Catalyzed Condensation
The core of this technological advancement lies in the precise manipulation of reaction kinetics through alkali catalysis. The mechanism involves the deprotonation of the alpha-position of methyl 3-methoxyacrylate by strong bases such as sodium methoxide or potassium methoxide, generating a reactive enolate species. This nucleophile then attacks the beta-carbon of another molecule of the starting ester in a Michael-type addition fashion, followed by elimination steps to establish the conjugated diene system characteristic of the glutaconate structure. The choice of solvent plays a pivotal role in stabilizing these ionic intermediates; polar aprotic solvents like DMF (N,N-Dimethylformamide) or DMAC (N,N-Dimethylacetamide) are preferred as they enhance the solubility of the ionic catalyst and facilitate the collision frequency of reactants without participating in side reactions. This careful orchestration of base strength and solvent polarity ensures that the condensation proceeds selectively towards the desired 4-methoxymethylene configuration rather than polymerizing or forming cyclic byproducts.
From an impurity control perspective, the alkaline environment is inherently superior to the acidic conditions of the past. Acidic catalysis often promotes thermodynamic equilibration that leads to mixtures of E/Z isomers and unwanted addition products where methanol adds across the double bond. The base-catalyzed route, however, operates under kinetic control that favors the formation of the specific conjugated system required for downstream applications. This mechanistic clarity translates directly to product quality; analytical data confirms the absence of detectable isomers and addition impurities in the final product. For R&D teams focused on cost reduction in pharmaceutical intermediate manufacturing, this means fewer purification cycles, less solvent consumption for recrystallization, and a final product that meets stringent specifications for use as a reference standard in HPLC analysis of agrochemicals like flonicamid.
How to Synthesize Dimethyl 4-Methoxymethylene-2-Glutaconate Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory scale-up and industrial production. The process begins with the preparation of a catalytic suspension, where the chosen alkali base is dispersed in the solvent prior to the addition of the substrate. This order of addition is critical to prevent localized overheating or premature polymerization. Once the methyl 3-methoxyacrylate is introduced, the reaction mixture is gently heated to the optimal range of 35-40°C. Monitoring the reaction progress is straightforward, as the conversion is typically complete within a short 3-hour window. Upon completion, the workup procedure is equally simple, involving a quench in ice water to neutralize the base, followed by standard liquid-liquid extraction. The crude product can be further purified via column chromatography or recrystallization using hexane/ethyl acetate systems to obtain the target compound as white solid particles. For detailed standardized operating procedures and safety protocols, please refer to the guide below.
- Prepare the reaction mixture by adding a catalytic amount of alkali base, such as sodium methoxide or potassium methoxide, into a polar aprotic solvent like DMF or DMAC under stirring.
- Introduce methyl 3-methoxyacrylate to the suspension and maintain the reaction temperature between 35°C and 40°C for approximately 3 hours to ensure complete conversion.
- Quench the reaction mixture in ice water, extract the organic phase with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical curiosity. The shift from plant-derived coumaric acid to synthetic methyl 3-methoxyacrylate fundamentally alters the cost structure of the supply chain. Methyl 3-methoxyacrylate is a commodity chemical produced on a massive industrial scale, ensuring price stability and abundant availability compared to the fluctuating markets of botanical extracts. This transition mitigates the risk of raw material shortages that could disrupt production schedules. Furthermore, the drastic reduction in reaction time from days to hours implies a significant increase in asset utilization rates for manufacturing facilities. Reactors that were previously tied up for 48-hour batches can now turnover multiple times in the same period, effectively expanding production capacity without the need for capital expenditure on new equipment. This efficiency gain is a powerful driver for cost reduction in agrochemical manufacturing sectors where margin pressure is constant.
- Cost Reduction in Manufacturing: The elimination of expensive reagents like trimethyl orthoformate and acetyl chloride directly lowers the bill of materials. Additionally, the simplified purification process reduces the consumption of solvents and energy associated with extensive distillation or chromatography. By avoiding the generation of complex impurity mixtures, the yield loss during purification is minimized, leading to substantial overall cost savings per kilogram of finished product. The use of common, recyclable solvents like DMF further enhances the economic profile of the process.
- Enhanced Supply Chain Reliability: Relying on widely available industrial feedstocks decouples the supply chain from agricultural variables such as harvest seasons or crop failures. The robustness of the synthetic route ensures consistent output quality, which is vital for maintaining long-term contracts with downstream pharmaceutical and agrochemical clients. The ability to produce the compound on demand with short lead times strengthens the reliability of the supply network, allowing partners to operate with leaner inventory levels.
- Scalability and Environmental Compliance: The process is inherently green by design, avoiding the use of heavy metal catalysts or corrosive acids that require specialized waste treatment. The reduction in reaction time and the absence of toxic byproducts simplify the environmental compliance burden. This scalability ensures that the transition from pilot plant to multi-ton commercial production is seamless, supporting the commercial scale-up of complex pharmaceutical intermediates with minimal regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of dimethyl 4-methoxymethylene-2-glutaconate. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy for decision-makers evaluating this technology for integration into their own processes.
Q: What are the primary advantages of the new condensation method over traditional coumaric acid routes?
A: The novel base-catalyzed condensation eliminates the need for expensive plant-extracted coumaric acid and harsh acidic conditions. It significantly reduces reaction time from over 10 hours to just 3 hours while avoiding the formation of difficult-to-remove isomeric impurities common in acid-catalyzed ring-opening processes.
Q: How does this intermediate assist in quality control for agrochemical production?
A: This compound serves as a critical reference standard for detecting and monitoring specific impurities during the synthesis of flonicamid. Its availability allows manufacturers to rigorously test intermediate streams, ensuring the final pesticide product meets stringent purity specifications without contamination from this specific byproduct.
Q: Is the synthesis process scalable for industrial manufacturing?
A: Yes, the process utilizes readily available industrial raw materials like methyl 3-methoxyacrylate and common solvents such as DMF. The operational simplicity, mild temperature requirements (35-40°C), and absence of complex purification steps make it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dimethyl 4-Methoxymethylene-2-Glutaconate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of global drug development and agrochemical production. Our technical team has thoroughly analyzed the potential of the base-catalyzed condensation route described in CN109293507B and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is smooth and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of dimethyl 4-methoxymethylene-2-glutaconate meets the exacting standards required for use as an analytical reference standard or a synthetic building block.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your supply chain. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive pricing quotes. Let us be your partner in driving innovation and efficiency in the fine chemical sector.
