Revolutionizing Phthalide Synthesis: A Green POMs-Catalyzed Route for High-Purity Pharmaceutical Intermediates
Revolutionizing Phthalide Synthesis: A Green POMs-Catalyzed Route for High-Purity Pharmaceutical Intermediates
The landscape of organic synthesis for bioactive heterocycles is constantly evolving, driven by the urgent need for greener, more efficient, and cost-effective manufacturing processes. A pivotal advancement in this domain is detailed in patent CN113004236B, which discloses a novel method for synthesizing 3-methylenebenzofuran-1(3H)-one compounds. These phthalide derivatives are not merely academic curiosities; they serve as critical scaffolds in medicinal chemistry, exhibiting potent anti-inflammatory, anti-diabetic, and anti-coagulant activities found in natural products like Angelica sinensis. The core innovation lies in the utilization of polyoxometalates (POMs), specifically phosphomolybdic acid (H3PMo12O40), as a highly efficient, heterogeneous-like catalyst. This approach replaces traditional, often toxic transition metal systems with a robust, environmentally benign alternative that operates under mild conditions, producing water as the sole byproduct. For R&D directors and procurement specialists alike, this represents a paradigm shift towards sustainable API intermediate manufacturing that does not compromise on yield or purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3-methyleneisobenzofuran-1(3H)-one skeleton has relied on methodologies that are increasingly untenable for modern green chemistry standards. Traditional intermolecular cyclization strategies, such as modified Perkin or Julia reactions involving phthalic anhydride, often suffer from harsh reaction conditions requiring high temperatures and strong bases. Furthermore, transition metal-catalyzed cyclizations, while effective, frequently necessitate the use of expensive noble metals like palladium or rhodium, coupled with sophisticated ligand systems that drive up raw material costs significantly. A critical bottleneck in these legacy processes is the generation of toxic byproducts and the subsequent difficulty in removing trace heavy metal residues to meet stringent pharmaceutical purity specifications. These factors collectively contribute to extended lead times, complex waste treatment protocols, and inflated production costs, creating substantial friction in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
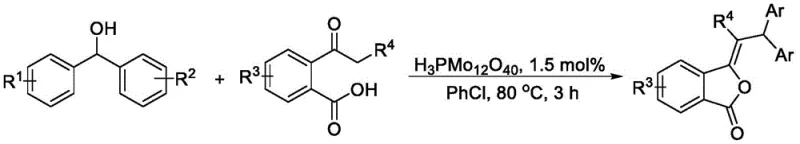
In stark contrast, the methodology outlined in CN113004236B introduces a streamlined dehydration coupling reaction between diphenylmethanol derivatives and 2-acylbenzoic acid derivatives. By leveraging the unique acidic and redox properties of the POMs catalyst, this route achieves high conversion rates at moderate temperatures ranging from 70°C to 100°C. The reaction is remarkably clean, with water identified as the only stoichiometric byproduct, thereby eliminating the need for complex downstream purification to remove hazardous waste streams. This novel approach not only simplifies the operational workflow but also enhances the overall atom economy of the synthesis. The versatility of the method is further demonstrated by its tolerance to various substituents, allowing for the efficient generation of diverse libraries of phthalide derivatives essential for drug discovery and development programs.
Mechanistic Insights into POMs-Catalyzed Dehydration Coupling
To fully appreciate the robustness of this synthetic route, one must delve into the intricate catalytic cycle facilitated by the phosphomolybdic acid cluster. The mechanism initiates with the protonation of the 2-acylbenzoic acid substrate by the strong Brønsted acidity of the POMs catalyst, generating a highly reactive oxocarbenium intermediate (INT1). This activated species undergoes an intramolecular nucleophilic attack by the adjacent carboxyl group, leading to the formation of a cyclic intermediate (INT2). Subsequent dehydration steps drive the equilibrium forward, eventually yielding the exocyclic double bond characteristic of the 3-methylenebenzofuran-1(3H)-one core. Crucially, the catalyst also activates the diphenylmethanol partner via protonation and dehydration to form a stable benzylic carbocation (INT6). This electrophilic species then engages in a Friedel-Crafts-type alkylation with the enol ether intermediate, forging the critical carbon-carbon bond that links the two aromatic systems. The elegance of this mechanism lies in its ability to orchestrate multiple bond-forming events in a single pot without the need for external activating agents.
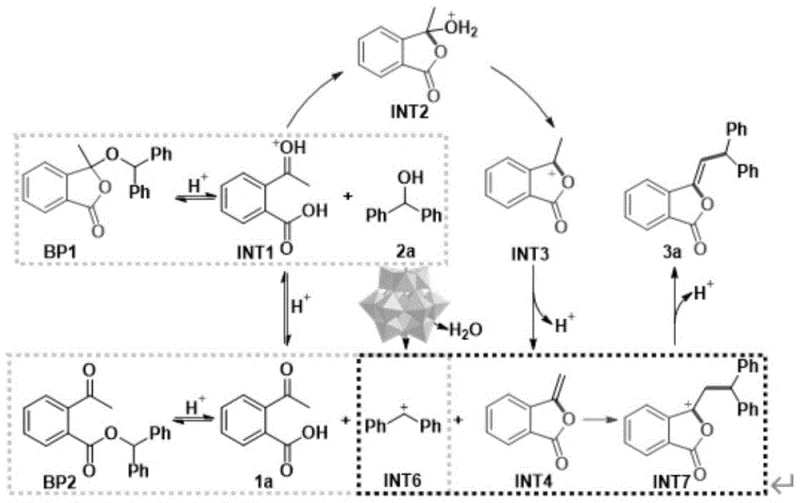
Beyond the primary reaction pathway, a deep understanding of the impurity profile is vital for ensuring the quality of the final API intermediate. The patent elucidates potential side reactions, such as the formation of byproduct BP1 via direct nucleophilic attack of the alcohol on the activated acid, or ester BP2 via interaction with the carbocation. However, the reversible nature of these side reactions under the acidic conditions provided by the POMs catalyst acts as a self-correcting mechanism. For instance, BP1 can revert to the starting materials, re-entering the productive catalytic cycle. This dynamic equilibrium effectively suppresses the accumulation of persistent impurities, resulting in a cleaner crude reaction mixture. Such intrinsic impurity control is a hallmark of a mature chemical process, significantly reducing the burden on purification teams and ensuring that the final product meets the rigorous quality standards demanded by regulatory bodies for pharmaceutical applications.
How to Synthesize 3-Methylenebenzofuran-1(3H)-ones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility. The process is designed to be operationally simple, avoiding the need for inert atmospheres or specialized high-pressure equipment. Typically, the reaction is conducted in chlorobenzene, which has been identified as the optimal solvent for balancing solubility and reaction rate, although other polar aprotic solvents can also be employed. The catalyst loading is remarkably low, often around 1.5 mol%, which underscores the high turnover efficiency of the polyoxometalate system. Reaction times are generally short, typically completing within 3 hours at 80°C, allowing for rapid throughput in process development campaigns. For a comprehensive breakdown of the standardized operating procedures, please refer to the technical guide below.
- Charge a reaction vessel with diphenylmethanol derivatives, 2-acylbenzoic acid derivatives, and the H3PMo12O40 catalyst (1.5 mol%).
- Add chlorobenzene as the solvent and stir the mixture at 80°C for approximately 3 hours to facilitate the dehydration coupling.
- Upon completion, isolate the target 3-methylenebenzofuran-1(3H)-one product via column chromatography separation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this POMs-catalyzed technology offers transformative benefits for procurement managers and supply chain heads tasked with optimizing the cost of goods sold (COGS) and ensuring supply continuity. The shift away from precious transition metals to earth-abundant polyoxometalates represents a direct and significant reduction in raw material expenditure. Moreover, the elimination of toxic heavy metals from the process flow removes the necessity for expensive and time-consuming metal scavenging steps, which are often required to meet residual metal limits in active pharmaceutical ingredients. This simplification of the downstream processing train translates directly into reduced manufacturing cycles and lower utility consumption. Additionally, the generation of water as the only byproduct drastically simplifies waste management protocols, aligning with increasingly strict environmental regulations and reducing the overhead associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily driven by the substitution of costly catalysts and the simplification of purification workflows. By utilizing a cheap, non-toxic solid acid catalyst, manufacturers can avoid the volatility associated with the pricing of noble metals like palladium or platinum. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, thereby improving the overall mass balance and yield. The absence of complex ligand systems further reduces the bill of materials, while the mild reaction conditions lower energy consumption related to heating and cooling. Collectively, these factors contribute to a substantially leaner cost structure for the production of complex phthalide intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials. Diphenylmethanol derivatives and 2-acylbenzoic acids are commodity chemicals with robust global supply networks, reducing the risk of bottlenecks associated with specialized reagents. The stability of the POMs catalyst ensures long shelf-life and consistent performance batch-to-batch, mitigating the risk of production delays due to catalyst degradation. Furthermore, the scalability of the process from gram to kilogram scales without significant re-optimization allows for seamless technology transfer from R&D to commercial manufacturing. This reliability ensures that downstream customers receive their critical intermediates on schedule, supporting uninterrupted drug production timelines.
- Scalability and Environmental Compliance: As the pharmaceutical industry moves towards greener manufacturing paradigms, this process stands out for its exceptional environmental profile. The use of a non-corrosive, solid catalyst minimizes equipment wear and tear, extending the lifespan of reactor vessels and reducing capital expenditure on maintenance. The benign nature of the byproducts facilitates easier regulatory approval for new drug filings, as the impurity profile is well-defined and free from genotoxic heavy metal concerns. The process is inherently safe, operating at atmospheric pressure and moderate temperatures, which lowers the barrier for scale-up in existing multipurpose facilities. This combination of safety, scalability, and sustainability makes it an ideal candidate for the commercial production of high-value fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is crucial for evaluating the feasibility of integrating this technology into existing production portfolios. We encourage technical teams to review these points carefully to assess the alignment with their specific project requirements.
Q: What are the primary advantages of using POMs catalysts over traditional transition metals?
A: Polyoxometalates (POMs) like H3PMo12O40 offer significant advantages including lower cost, non-toxicity, and ease of handling compared to expensive transition metals. Furthermore, they eliminate the need for complex heavy metal removal steps, streamlining the purification process and reducing environmental impact.
Q: How does the substituent effect influence the reaction yield in this synthesis?
A: The patent data indicates that electron-withdrawing groups (such as fluorine, chlorine, or bromine) on the diphenylmethanol substrate significantly enhance the reaction yield, reaching up to 97%. Conversely, electron-donating groups may result in slightly lower but still robust yields above 85%.
Q: Is this method scalable for industrial production of API intermediates?
A: Yes, the method utilizes mild reaction conditions (70-100°C) and generates water as the only byproduct, which simplifies waste treatment. The use of a solid, stable catalyst and common organic solvents like chlorobenzene makes the process highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methylenebenzofuran-1(3H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative synthetic routes that balance efficiency with sustainability. Our team of expert chemists has extensively evaluated the POMs-catalyzed pathway described in CN113004236B and validated its potential for large-scale application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-methylenebenzofuran-1(3H)-one delivered meets the highest international standards. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that enhance your competitive advantage.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this green catalytic system. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of this intermediate with your downstream processes. Let us collaborate to drive innovation and efficiency in your pharmaceutical supply chain, ensuring a reliable source of high-quality intermediates for your critical drug development programs.
