Advanced Copper-Catalyzed Synthesis of Arylethynyl Formamides for Pharmaceutical Intermediates
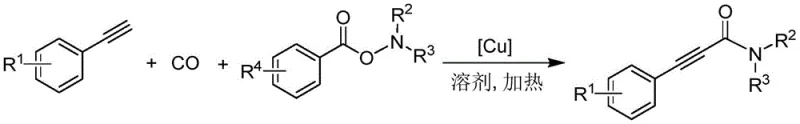
The chemical industry is constantly seeking more efficient pathways to construct complex molecular architectures, particularly those containing amide functionalities which are ubiquitous in bioactive molecules. Patent CN114213272B, published in late 2022, introduces a groundbreaking synthetic methodology for arylethynyl formamide compounds that addresses many of the inefficiencies plaguing traditional amide synthesis. This innovation leverages a monovalent copper-catalyzed three-component coupling reaction involving aryl acetylenes, carbon monoxide, and O-aryl formyl hydroxylamines. For R&D directors and procurement specialists alike, this patent represents a significant shift towards more atom-economical and cost-effective manufacturing processes. The ability to construct the amide bond directly from simple precursors under relatively mild conditions opens new avenues for the rapid assembly of pharmaceutical intermediates and agrochemical building blocks. By utilizing earth-abundant copper instead of precious metals, the process not only lowers the barrier to entry for synthesis but also aligns with modern green chemistry principles, reducing the environmental footprint associated with heavy metal waste disposal.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of amide bonds has relied on several established yet problematic methodologies that often hinder efficient large-scale production. Traditional routes frequently involve the reaction of aldehydes with amines, which necessitates the use of stoichiometric amounts of peroxides or additives that generate significant chemical waste and pose safety hazards. Alternatively, the rearrangement of oximes to amides typically demands harsh reaction conditions, including high temperatures and strong acids, which can degrade sensitive functional groups and require specialized corrosion-resistant equipment. Another common approach involves the direct coupling of carboxylic acids and amines, but this often requires high thermal energy and transition metal catalysts that leave behind toxic residues difficult to remove from the final product. Furthermore, methods utilizing alcohols or nitriles often depend on equivalent amounts of noble metal catalysts like palladium, driving up the raw material costs substantially and creating supply chain vulnerabilities due to the fluctuating market prices of precious metals. These conventional limitations collectively result in higher production costs, complex purification workflows, and increased environmental compliance burdens for manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a streamlined copper-catalyzed carbonylation strategy that dramatically simplifies the synthetic landscape. By employing a monovalent copper salt as the catalyst, the reaction proceeds efficiently at atmospheric pressure of carbon monoxide, eliminating the need for high-pressure equipment often required for carbonylation reactions. The use of O-aryl formyl hydroxylamines as electrophilic partners allows for the direct formation of the formamide moiety without the need for pre-activation of carboxylic acids or the use of hazardous coupling reagents. This method demonstrates exceptional substrate universality, tolerating a wide range of substituents on the aryl rings, which is crucial for the diversification of drug candidates during the lead optimization phase. The operational simplicity is further highlighted by the moderate temperature range of 80-100°C and reaction times of approximately 8 to 12 hours, which facilitates easier energy management in a plant setting. Ultimately, this approach transforms a multi-step, resource-intensive process into a concise, one-pot transformation that maximizes yield while minimizing waste.
Mechanistic Insights into Copper-Catalyzed Carbonylative Coupling
The mechanistic pathway of this transformation offers profound insights into why this method achieves such high efficiency and selectivity compared to traditional alternatives. The catalytic cycle initiates with the activation of the terminal alkyne by the monovalent copper species, forming a reactive copper-acetylide intermediate. This species is pivotal as it serves as the nucleophilic driver for the subsequent steps. Following this activation, carbon monoxide inserts into the copper-carbon bond, generating an acyl-copper intermediate. This insertion step is critical and is facilitated by the specific electronic properties of the copper catalyst, which stabilizes the acyl species sufficiently to prevent premature decomposition. The final step involves the nucleophilic attack of this acyl-copper species onto the electron-deficient amino group of the O-aryl formyl hydroxylamine. This attack displaces the aryloxy leaving group and forms the desired amide bond, regenerating the copper catalyst to continue the cycle. Understanding this mechanism allows chemists to fine-tune ligand environments and solvent systems to further enhance reaction rates and suppress potential side reactions such as homocoupling of the alkynes.
From an impurity control perspective, the mild nature of this catalytic system plays a vital role in ensuring high product purity, a key metric for pharmaceutical intermediates. Harsh acidic or basic conditions found in older methods often lead to the hydrolysis of sensitive functional groups or the formation of polymeric byproducts that are difficult to separate. In this copper-catalyzed system, the neutral to slightly basic conditions inherent to the amine substrates help maintain the integrity of the molecular scaffold. Furthermore, the use of carbon monoxide as a C1 source is inherently clean, producing no stoichiometric byproducts other than the phenol derivative from the hydroxylamine leaving group, which is easily removed during the aqueous workup. The high selectivity of the copper catalyst minimizes the formation of regioisomers or over-carbonylated species, resulting in a cleaner crude reaction profile. This reduction in impurity load directly translates to simplified downstream processing, requiring fewer chromatography columns or recrystallization steps to meet stringent quality specifications required by regulatory bodies.
How to Synthesize Arylethynyl Formamide Efficiently
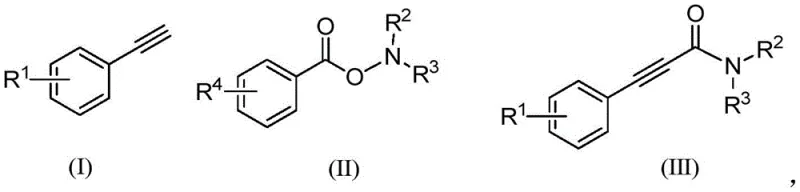
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the handling of carbon monoxide and the selection of appropriate solvents to maximize yield. The patent outlines a robust protocol where aryl acetylene and O-aryl formyl hydroxylamine are combined with a catalytic amount of copper salt, such as cuprous acetate, in a polar aprotic solvent like DMF. The reaction vessel must be thoroughly degassed to remove oxygen, which can oxidize the copper catalyst and inhibit the reaction, before being pressurized with 1 atmosphere of CO. Heating the mixture to 80-100°C drives the reaction to completion within 8 to 12 hours, after which standard workup procedures involving filtration and washing are sufficient to isolate the product. For detailed operational parameters and safety guidelines regarding the specific molar ratios and purification techniques, please refer to the standardized synthesis steps provided below.
- Charge a reaction vessel with aryl acetylene, O-aryl formyl hydroxylamine, and a monovalent copper salt catalyst such as cuprous acetate.
- Evacuate the system to remove air and introduce carbon monoxide (CO) gas at 1 atmosphere pressure, followed by the addition of an organic solvent like DMF.
- Heat the reaction mixture to 80-100°C for 8-12 hours, then filter, wash, dry, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology offers tangible strategic advantages that extend beyond mere technical feasibility. The most immediate benefit is the drastic reduction in raw material costs achieved by replacing expensive noble metal catalysts like palladium with inexpensive and abundant copper salts. This substitution not only lowers the direct cost of goods sold but also mitigates the risk associated with the volatile pricing of precious metals, ensuring more stable long-term budget forecasting. Additionally, the elimination of stoichiometric oxidants and harsh reagents reduces the cost of waste treatment and disposal, contributing to a leaner and more sustainable manufacturing operation. The simplicity of the workup procedure, which avoids complex extraction or distillation steps, further enhances throughput and reduces labor costs associated with production. These factors combine to create a highly competitive cost structure that allows suppliers to offer better pricing to downstream pharmaceutical clients without compromising on quality.
- Cost Reduction in Manufacturing: The shift from palladium to copper catalysts represents a fundamental change in the cost dynamics of amide synthesis, removing the dependency on critical raw materials that are subject to geopolitical supply constraints. By utilizing earth-abundant metals, manufacturers can achieve significant savings on catalyst procurement while simultaneously reducing the burden of heavy metal residue testing in the final API. The avoidance of expensive coupling reagents and oxidants further strips away unnecessary cost layers from the bill of materials. This economic efficiency is compounded by the high yields reported in the patent examples, which minimize the loss of valuable starting materials and maximize the output per batch. Consequently, the overall cost of production is substantially lowered, enabling more aggressive pricing strategies in the competitive intermediates market.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as aryl acetylenes and simple hydroxylamines ensures a robust and resilient supply chain that is less prone to disruptions. Unlike specialized reagents that may have single-source suppliers or long lead times, the substrates for this reaction are commodity chemicals produced by multiple vendors globally. This diversity in sourcing options provides procurement teams with greater flexibility and bargaining power, reducing the risk of production stoppages due to material shortages. Furthermore, the mild reaction conditions reduce the wear and tear on reactor equipment, extending asset life and minimizing unplanned maintenance downtime. The combination of reliable raw material availability and durable processing conditions creates a supply chain framework that can consistently meet delivery schedules even during periods of market volatility.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the use of standard organic solvents and atmospheric pressure conditions, which do not require specialized high-pressure reactors or exotic engineering controls. The reduced generation of hazardous waste streams simplifies environmental compliance and lowers the costs associated with effluent treatment and regulatory reporting. The atom economy of the reaction is superior to many traditional methods, as the majority of the reactant mass is incorporated into the final product rather than being lost as byproducts. This alignment with green chemistry principles not only satisfies increasingly strict environmental regulations but also enhances the corporate sustainability profile of the manufacturer. As global pharmaceutical companies prioritize green suppliers, this environmentally friendly process becomes a key differentiator in securing long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and comparative data. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers. We encourage stakeholders to review these points carefully to fully appreciate the transformative potential of this copper-catalyzed approach.
Q: What are the primary advantages of this copper-catalyzed method over traditional palladium-catalyzed routes?
A: This method eliminates the need for expensive noble metal palladium catalysts and toxic oxidants, significantly reducing raw material costs and environmental impact while maintaining high yields up to 91%.
Q: What are the typical reaction conditions required for this synthesis?
A: The reaction operates under mild conditions, typically requiring temperatures between 80-100°C and atmospheric pressure of carbon monoxide (1 atm) in solvents like DMF for 8-12 hours.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process utilizes cheap and readily available raw materials with a simple workup procedure involving filtration and chromatography, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylethynyl Formamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN114213272B to reshape the landscape of fine chemical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that for pharmaceutical and agrochemical clients, consistency and reliability are paramount, and our infrastructure is designed to deliver high-purity arylethynyl formamide intermediates that meet the most demanding global standards. By leveraging our expertise in copper catalysis and carbonylation chemistry, we can help you optimize your supply chain and reduce time-to-market for your critical drug candidates.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this copper-catalyzed method for your specific molecule. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver this complex intermediate at the scale and quality you require. Let us collaborate to build a more efficient and sustainable supply chain for your next-generation therapeutics, leveraging our technical prowess to drive your success in the global market.
