Advanced Rhodium-Catalyzed Synthesis of Aminal Isoquinolinones for Commercial Pharmaceutical Manufacturing
Introduction to Next-Generation Isoquinolinone Synthesis
The landscape of organic synthesis methodology is continually evolving to meet the rigorous demands of the modern pharmaceutical industry, particularly in the construction of complex heterocyclic scaffolds. A significant breakthrough in this domain is documented in patent CN109053571B, which details a highly efficient preparation method for aminal isoquinolinone compounds. These structures serve as critical backbone motifs in numerous drug molecules and biologically active agents, making their accessible synthesis a priority for R&D teams globally. The disclosed technology leverages a sophisticated transition metal-catalyzed C-H activation strategy, specifically utilizing a rhodium catalyst system to drive the self-cyclization of N-vinyl aromatic amides. This approach represents a paradigm shift from classical condensation methods, offering a streamlined pathway that aligns perfectly with the needs of a reliable pharmaceutical intermediate supplier seeking to optimize production workflows. By integrating this methodology, manufacturers can achieve higher purity profiles and reduced operational complexity, directly addressing the pain points of cost and efficiency in fine chemical manufacturing.
The core innovation lies in the synergistic action of a rhodium catalyst, a silver oxidant, and a base under a protective nitrogen atmosphere. This combination facilitates the direct formation of the isoquinolinone core without the need for pre-functionalized coupling partners that are often expensive or unstable. For procurement managers and supply chain heads, this translates to a more resilient sourcing strategy, as the starting materials are commercially accessible and the reaction conditions are remarkably mild. The ability to convert simple substrates into high-value heterocycles in a single operational step significantly reduces the cumulative yield losses typically associated with multi-step syntheses. As we delve deeper into the technical specifics, it becomes evident that this patent offers not just a chemical reaction, but a comprehensive solution for cost reduction in pharmaceutical intermediates manufacturing through process intensification and simplification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the aminal isoquinolinone skeleton has relied on laborious and chemically demanding protocols that pose significant challenges for large-scale production. Traditional routes often involve the condensation of ketones or aldehydes with diamines, or the reaction of isocoumarins with amines, processes that frequently require harsh acidic or basic conditions and elevated temperatures. These legacy methods suffer from poor atom economy and generate substantial amounts of chemical waste, complicating downstream purification and environmental compliance. Furthermore, alternative transition metal-catalyzed approaches, such as palladium-catalyzed cyclization of N-allenyl benzamides, often necessitate specialized coupling reagents that are difficult to source and handle safely on a metric ton scale. The cumulative effect of these limitations is a prolonged lead time for high-purity pharmaceutical intermediates and inflated production costs, creating bottlenecks in the supply chain for active pharmaceutical ingredients (APIs). Additionally, the sensitivity of some intermediates to harsh reaction conditions can lead to decomposition or the formation of difficult-to-remove impurities, compromising the overall quality of the final product.
The Novel Approach
In stark contrast, the methodology described in patent CN109053571B introduces a transformative one-step cyclization strategy that bypasses the inefficiencies of conventional synthesis. By employing N-vinyl arylamides as the sole organic substrate, the process achieves direct C-C and C-N bond formation through a rhodium-catalyzed C-H activation mechanism. This eliminates the need for external coupling agents and reduces the number of unit operations required to reach the target molecule. The reaction proceeds under mild thermal conditions, typically around 50°C, which preserves the integrity of sensitive functional groups and minimizes energy consumption. 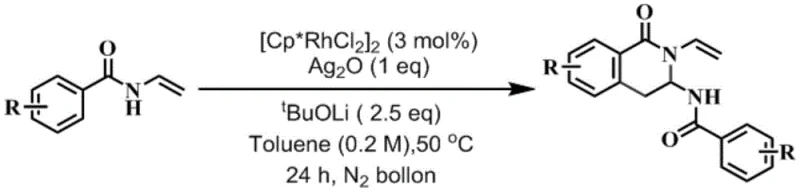
The operational simplicity is further enhanced by the use of common organic solvents like toluene and a straightforward workup procedure involving filtration and extraction. This novel approach not only accelerates the commercial scale-up of complex pharmaceutical intermediates but also ensures a cleaner reaction profile with fewer byproducts. For technical decision-makers, this represents a robust platform technology capable of generating diverse libraries of isoquinolinone derivatives with high consistency and reliability, thereby securing the supply chain against the volatility of raw material availability and processing failures.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation
The success of this synthesis hinges on the precise orchestration of organometallic steps mediated by the pentamethylcyclopentadienyl rhodium(III) complex, typically [Cp*RhCl2]2. The catalytic cycle initiates with the coordination of the rhodium species to the amide oxygen, directing the metal center to the proximal C-H bond on the aromatic ring. Through a concerted metalation-deprotonation (CMD) pathway facilitated by the base, a stable rhodacycle intermediate is formed. This key organometallic species then undergoes migratory insertion of the pendant vinyl group, effectively closing the six-membered ring structure characteristic of the isoquinolinone core. The presence of the silver oxidant, such as Ag2O, plays a dual role: it regenerates the active Rh(III) catalyst from the reduced Rh(I) species formed during the reductive elimination step, and it acts as a halide scavenger to maintain catalyst activity. This redox-neutral cycle ensures high turnover numbers and sustained catalytic efficiency throughout the reaction duration.
From an impurity control perspective, the specificity of the C-H activation step is paramount. The steric and electronic properties of the Cp* ligand enforce a high degree of regioselectivity, ensuring that cyclization occurs exclusively at the desired position on the aromatic ring. This intrinsic selectivity minimizes the formation of regioisomers, which are often challenging to separate and can act as genotoxic impurities in final drug substances. Furthermore, the mild reaction temperature prevents thermal degradation of the N-vinyl moiety, a common side reaction in less controlled environments. 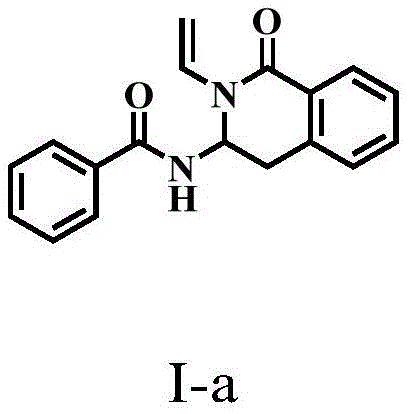
The result is a product stream with a simplified impurity profile, reducing the burden on downstream purification technologies like preparative HPLC or recrystallization. For R&D directors, understanding this mechanism provides confidence in the scalability of the process, as the fundamental chemical transformations are robust and less susceptible to minor fluctuations in reaction parameters, ensuring batch-to-batch consistency essential for regulatory compliance.
How to Synthesize Aminal Isoquinolinone Efficiently
Implementing this synthesis protocol requires careful attention to reagent stoichiometry and atmospheric control to maximize yield and purity. The process begins with the precise weighing of the N-vinyl arylamide substrate, the rhodium dimer catalyst, the silver oxide oxidant, and the lithium tert-butoxide base. These components are suspended in anhydrous toluene within a reaction vessel equipped for inert gas handling. The detailed standardized synthesis steps, including specific molar ratios, addition sequences, and purification parameters, are outlined in the guide below to ensure reproducibility across different laboratory and plant settings.
- Prepare the reaction mixture by combining N-vinyl arylamide substrate, [Cp*RhCl2]2 catalyst (3 mol%), Ag2O oxidant (1 equivalent), and tBuOLi base (2.5 equivalents) in toluene solvent.
- Ensure an inert atmosphere by purging the reaction vessel with nitrogen gas three times and maintaining a nitrogen balloon pressure throughout the process.
- Heat the mixture to 50°C and stir for 24 hours, followed by filtration, solvent removal, extraction with ethyl acetate, and purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this rhodium-catalyzed methodology offers profound strategic benefits for procurement and supply chain management, fundamentally altering the cost structure of producing aminal isoquinolinone derivatives. By collapsing a multi-step synthesis into a single pot operation, the process drastically reduces the consumption of solvents, reagents, and labor hours associated with intermediate isolation and purification. This consolidation of steps leads to substantial cost savings in manufacturing overheads, allowing companies to remain competitive in price-sensitive markets without compromising on quality. The elimination of expensive and hazardous coupling reagents further contributes to a leaner bill of materials, mitigating the financial risks associated with volatile raw material pricing.
- Cost Reduction in Manufacturing: The streamlined nature of this one-step cyclization significantly lowers the operational expenditure per kilogram of product. By avoiding the need for multiple reaction vessels and extensive workup procedures between steps, facilities can achieve higher throughput with existing infrastructure. The use of earth-abundant solvents like toluene and the recovery of silver byproducts add another layer of economic efficiency, ensuring that the overall cost of goods sold is minimized while maintaining high margins.
- Enhanced Supply Chain Reliability: The reliance on readily available N-vinyl arylamides as starting materials decouples production from the supply constraints of exotic or proprietary reagents. This accessibility ensures a continuous flow of raw materials, reducing the risk of production stoppages due to sourcing delays. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in utility supply or environmental conditions, enhancing the overall resilience of the manufacturing supply chain against external disruptions.
- Scalability and Environmental Compliance: The mild thermal requirements and reduced waste generation align perfectly with green chemistry principles and stringent environmental regulations. Scaling this process from gram to metric ton quantities does not require specialized high-pressure or cryogenic equipment, simplifying the engineering requirements for plant expansion. The reduced solvent usage and simpler waste streams lower the environmental footprint, facilitating easier permitting and compliance with increasingly strict global sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating the feasibility of adoption. Understanding these nuances is critical for making informed decisions about process integration and resource allocation.
Q: What are the primary advantages of this Rhodium-catalyzed method over traditional synthesis routes?
A: This method eliminates the need for cumbersome multi-step sequences and harsh reaction conditions often associated with traditional condensation reactions. It utilizes readily available N-vinyl arylamide substrates to achieve direct cyclization in a single step, significantly streamlining the manufacturing workflow.
Q: Is this synthesis protocol scalable for industrial production of pharmaceutical intermediates?
A: Yes, the protocol operates under mild thermal conditions (50°C) and uses standard organic solvents like toluene, which are compatible with large-scale reactor systems. The simplicity of the workup procedure, involving standard filtration and extraction, supports robust commercial scale-up.
Q: What is the substrate scope for this aminal isoquinolinone preparation method?
A: The method demonstrates broad substrate tolerance, accommodating various substituents on the aromatic ring including halogens (fluorine, chlorine, bromine), alkyl groups, and alkoxy groups. This versatility allows for the generation of diverse chemical libraries for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminal Isoquinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in reshaping the pharmaceutical intermediate landscape. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our capability to adapt complex synthetic routes like the rhodium-catalyzed cyclization described here allows us to offer customized solutions that meet the exacting standards of global pharmaceutical clients.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into the economic benefits tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to validate the compatibility of this technology with your current development pipeline. Let us partner with you to engineer a more efficient and sustainable future for your chemical manufacturing needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
