Advanced Catalytic Synthesis of C2-Phosphonomethylene Indole Compounds for Pharmaceutical Applications
Introduction to Novel Phosphonoindole Technology
The landscape of organic synthesis is continually evolving to meet the rigorous demands of the pharmaceutical and fine chemical industries, particularly for complex heterocyclic scaffolds. Patent CN111187298A introduces a groundbreaking methodology for the efficient construction of C2-phosphonomethylene indole compounds, a class of molecules that serves as critical building blocks in medicinal chemistry and ligand design. These multifunctional compounds, characterized by an indole ring substituted with a phosphono group at the C2 position, offer unique electronic and steric properties that are highly valued in the development of bioactive agents and catalytic ligands. The disclosed technology represents a significant leap forward by enabling the direct formation of the C(sp3)-P bond through a streamlined catalytic process that operates under remarkably mild conditions. Unlike traditional multi-step sequences that often require stringent exclusion of moisture or oxygen, this innovative approach allows reactions to proceed directly in an air atmosphere, drastically simplifying operational protocols for process chemists. The versatility of this method is underscored by its compatibility with a broad spectrum of substrates, ensuring that diverse chemical libraries can be accessed with high efficiency and minimal waste generation.
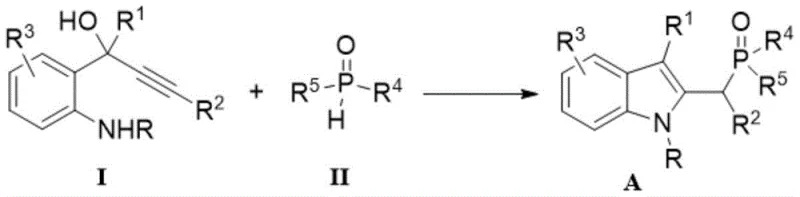
Furthermore, the strategic importance of these phosphorus-containing indoles extends beyond their immediate utility as intermediates; they act as precursors for further functionalization, enabling the rapid assembly of complex molecular architectures required for modern drug discovery programs. The ability to introduce a phosphono group specifically at the methylene position adjacent to the indole core opens new avenues for modulating the physicochemical properties of lead compounds, such as solubility and metabolic stability. By leveraging this patented technology, manufacturers can secure a reliable supply of high-purity pharmaceutical intermediates that are essential for the next generation of therapeutic agents. The robustness of the reaction conditions, combined with the use of commercially available starting materials, positions this synthesis route as a cornerstone for scalable production in the fine chemical sector. As the industry shifts towards more sustainable and cost-effective manufacturing practices, the adoption of such atom-economical processes becomes not just a technical preference but a commercial imperative for maintaining competitiveness in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
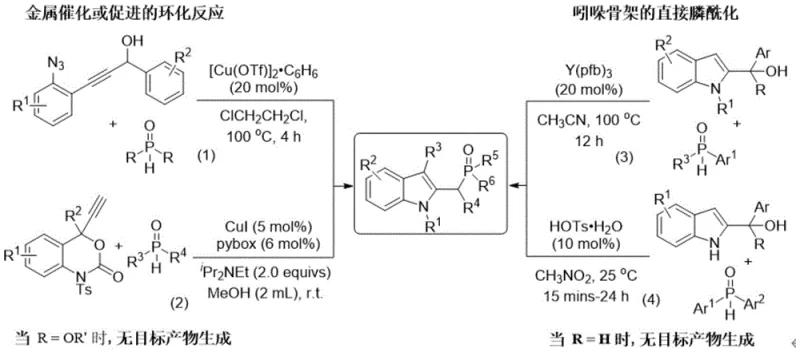
Historically, the construction of C2-phosphonomethylidene indole backbones has been fraught with significant technical challenges that hindered their widespread adoption in large-scale manufacturing. Prior art methods, as illustrated in the comparative analysis of existing technologies, often relied on metal-catalyzed cyclization reactions or direct phosphonylation strategies that suffered from severe limitations in substrate scope and operational complexity. Many conventional protocols necessitated the use of expensive and sensitive transition metal catalysts in high loadings, which not only inflated production costs but also introduced difficult-to-remove metal impurities into the final product, posing risks for pharmaceutical applications. Additionally, these traditional routes frequently required harsh reaction conditions, including elevated temperatures and the use of hazardous solvents like methanol, which complicated safety management and environmental compliance. The reliance on specialized starting materials with rigid structural requirements further restricted the diversity of accessible compounds, limiting the ability of R&D teams to explore comprehensive structure-activity relationships. Moreover, the generation of stoichiometric amounts of toxic byproducts in some older methods resulted in poor atom economy, creating substantial waste disposal burdens that are increasingly unacceptable in modern green chemistry frameworks.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent data offers a transformative solution that addresses nearly every bottleneck associated with conventional synthesis. This method utilizes a catalytic system that promotes the dehydration and subsequent cyclization of readily available propargyl alcohol derivatives with phosphine oxides, achieving high conversion rates under exceptionally mild conditions. The reaction is capable of proceeding at temperatures ranging from 0°C to 100°C, allowing for precise thermal control that minimizes energy consumption and prevents the degradation of sensitive functional groups. A defining feature of this technology is its remarkable tolerance for air and moisture, eliminating the need for costly inert gas protection systems and specialized glovebox equipment, thereby democratizing access to these valuable intermediates for a wider range of laboratories. The catalyst loading is significantly reduced compared to prior art, often requiring only catalytic amounts of inexpensive metals like copper or iron, or even organic acids, which translates to direct cost savings and simplified downstream purification. Furthermore, the exclusive formation of water as a byproduct ensures high atom economy, aligning perfectly with the principles of green chemistry and reducing the environmental footprint of the manufacturing process. This combination of operational simplicity, cost efficiency, and environmental sustainability makes the novel approach the superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Catalytic Cyclization and Phosphonylation
Understanding the underlying mechanistic pathway is crucial for R&D directors aiming to optimize this process for specific target molecules. The reaction mechanism involves a sophisticated cascade initiated by the activation of the hydroxyl group in the propargyl alcohol substrate by a Lewis or Brønsted acid catalyst. This activation facilitates the elimination of a water molecule, generating a reactive propargylic cation intermediate that is stabilized by the adjacent aromatic system. Subsequently, the phosphine oxide acts as a nucleophile, attacking the electrophilic center to form a new carbon-phosphorus bond. This nucleophilic substitution is followed by a concerted 5-exo-trig cyclization, where the nitrogen atom of the aniline moiety attacks the alkyne triple bond, closing the indole ring and restoring aromaticity. The elegance of this mechanism lies in its step-economy, combining bond formation and ring closure into a single operational sequence without the need for isolating unstable intermediates. The choice of catalyst plays a pivotal role in modulating the electrophilicity of the intermediate and the nucleophilicity of the phosphine oxide, with transition metals like copper and scandium offering distinct electronic environments that can be tuned for specific substrate classes. This mechanistic clarity allows chemists to predict reactivity patterns and troubleshoot potential issues related to steric hindrance or electronic deactivation in complex substrates.
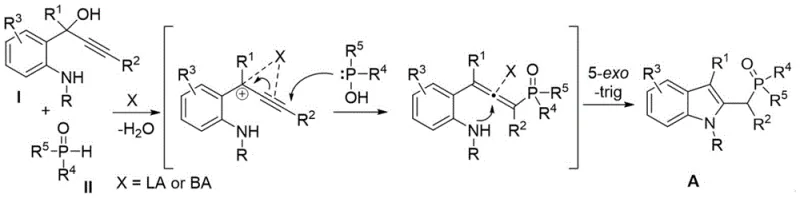
From an impurity control perspective, the mechanism offers inherent advantages that simplify the purification profile of the final product. Since the only stoichiometric byproduct is water, the reaction mixture does not accumulate salt wastes or organic side products that typically complicate chromatographic separation. The high selectivity of the 5-exo-trig cyclization ensures that the indole core is formed regioselectively, minimizing the formation of isomeric byproducts that could compromise the purity specifications required for API intermediates. The mild acidic conditions employed prevent the polymerization of the alkyne moiety or the decomposition of acid-sensitive protecting groups, which is a common issue in harsher cyclization protocols. Furthermore, the ability to operate under air atmosphere suggests that the catalytic cycle is robust against oxidative deactivation, ensuring consistent performance over extended reaction times. For process development teams, this mechanistic robustness translates to a wider operating window, reducing the risk of batch failures and ensuring reproducible quality across different production scales. The detailed understanding of this pathway empowers manufacturers to implement rigorous quality control measures that guarantee the structural integrity and purity of the C2-phosphonomethylene indole compounds.
How to Synthesize C2-Phosphonomethylene Indole Efficiently
The practical implementation of this synthesis route is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment. The general procedure involves mixing the propargyl alcohol substrate, the phosphine oxide reagent, the selected catalyst, and an appropriate solvent in a reaction vessel. The mixture is then heated to the specified temperature, which can vary depending on the specific catalyst-substrate combination, typically ranging from ambient temperature to 100°C. Reaction progress is conveniently monitored using thin-layer chromatography (TLC), allowing for real-time assessment of conversion and ensuring that the reaction is stopped at the optimal point to maximize yield and minimize side reactions. Upon completion, the workup procedure is remarkably simple, often involving the removal of the solvent under reduced pressure followed by direct purification via column chromatography. This streamlined workflow eliminates the need for complex aqueous workups or extensive extraction procedures, saving both time and solvent resources. The detailed standardized synthesis steps for specific embodiments are provided below to guide technical teams in replicating these results with high fidelity.
- Mix compound I (propargyl alcohol derivative), compound II (phosphine oxide), catalyst, and solvent in a reaction vessel.
- Heat the mixture to a temperature between 0°C and 100°C depending on the catalyst system used.
- Monitor reaction by TLC, then purify the crude product via column chromatography to obtain the target indole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers compelling strategic advantages that extend far beyond mere technical feasibility. The primary value proposition lies in the significant reduction of manufacturing costs driven by the use of inexpensive and commercially available raw materials. The starting propargyl alcohols and phosphine oxides are commodity chemicals that can be sourced from multiple suppliers, mitigating the risk of supply chain disruptions and preventing vendor lock-in scenarios. The low catalyst loading requirements mean that the consumption of precious metals is minimized, and in many cases, base metals like copper or iron can be utilized, which are orders of magnitude cheaper than palladium or gold. This shift in catalyst selection directly impacts the bill of materials, resulting in substantial cost savings that improve the overall margin profile of the final intermediate. Additionally, the operational simplicity of running reactions under air atmosphere reduces the capital expenditure required for specialized reactor infrastructure, as there is no need for extensive nitrogen purging systems or explosion-proof facilities typically associated with pyrophoric reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are profound, primarily due to the elimination of expensive ligands and the reduction of catalyst loading to trace levels. By avoiding the use of hazardous solvents like methanol in favor of greener alternatives such as ethyl acetate or toluene, companies can also reduce waste disposal costs and regulatory compliance burdens. The high atom economy ensures that the majority of the mass of the starting materials ends up in the final product, minimizing raw material waste and maximizing resource efficiency. Furthermore, the short reaction times reported in the patent data imply higher throughput capabilities, allowing manufacturing facilities to produce more batches within the same timeframe, effectively lowering the fixed cost per unit. These cumulative factors create a highly competitive cost structure that enables suppliers to offer aggressive pricing while maintaining healthy profitability margins in the volatile fine chemical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of this synthetic method, which tolerates a wide variety of functional groups and substrate structures. This flexibility means that if a specific raw material becomes scarce, alternative analogs can often be substituted without requiring a complete re-optimization of the process, ensuring continuity of supply for downstream customers. The ability to store raw materials at normal temperature without strict special treatment simplifies inventory management and reduces the need for climate-controlled warehousing, further lowering logistical overheads. Moreover, the scalability of the reaction from milligram to kilogram scales has been demonstrated, providing confidence that the process can meet the demands of commercial production without unforeseen technical hurdles. This reliability is critical for long-term supply agreements with multinational pharmaceutical companies that demand consistent quality and uninterrupted delivery schedules.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this technology aligns perfectly with modern sustainability goals and regulatory requirements. The generation of water as the sole byproduct eliminates the need for complex effluent treatment systems to handle toxic organic waste streams, simplifying the permitting process for new manufacturing lines. The use of non-hazardous solvents and the absence of heavy metal contamination in the final product streamline the regulatory filing process for drug substances, accelerating time-to-market for new therapies. The process is inherently safe, operating at moderate temperatures and pressures, which reduces the risk of thermal runaways and enhances workplace safety for operators. These environmental and safety advantages not only protect the company's reputation but also future-proof the manufacturing asset against tightening global environmental regulations, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled a list of frequently asked questions based on the detailed patent specifications. These answers provide clarity on the scope, limitations, and practical considerations of implementing this method in a production environment. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The following information is derived directly from the experimental data and claims of the source intellectual property, ensuring accuracy and relevance for decision-makers.
Q: What catalysts are suitable for this C2-phosphonomethylene indole synthesis?
A: The patent discloses a wide range of catalysts including transition metal compounds like copper, iron, palladium, gold, platinum, mercury, and scandium salts, as well as organic acids like p-toluenesulfonic acid.
Q: What are the main advantages of this preparation method over conventional routes?
A: The method features mild reaction conditions, operation under air atmosphere, low catalyst loading, short reaction times, and high atom economy with water as the only byproduct.
Q: Can this method tolerate various functional groups on the substrate?
A: Yes, the method demonstrates good substrate tolerance, accommodating halogens, nitro groups, alkoxy groups, and various aryl or heteroaryl substituents without significant loss in yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C2-Phosphonomethylene Indole Supplier
As the global demand for high-performance pharmaceutical intermediates continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a strategic advantage in navigating the complexities of modern drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing without compromising on quality or timeline. We understand that the synthesis of complex heterocycles like C2-phosphonomethylene indoles requires precise control over reaction parameters and rigorous analytical testing. Therefore, our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch delivered meets the highest international standards for pharmaceutical grade materials. Our commitment to technical excellence ensures that we can handle the most challenging synthetic routes with efficiency and reliability.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing your supply chain with our advanced synthesis methods can drive down your overall COGS. We encourage potential partners to contact us directly to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than assumptions. Whether you require small quantities for preclinical studies or metric tons for commercial launch, NINGBO INNO PHARMCHEM is positioned to be your trusted partner in delivering high-purity C2-phosphonomethylene indole compounds that power the next generation of life-saving medicines.
