Advanced Pyridine-Amide-Oxazoline Ligands for High-Efficiency Asymmetric Hydroboration
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the urgent need for sustainable, cost-effective, and highly selective chemical processes. Patent CN109593085B introduces a groundbreaking class of pyridine-amide-oxazoline ligands that address these critical industrial challenges. These novel compounds, represented by Formula (1), are designed to function not only as standalone reagents but, more importantly, as robust scaffolds for forming stable metal complexes with a diverse array of transition metals. The structural versatility allows for coordination with earth-abundant metals such as Iron, Cobalt, and Copper, offering a viable alternative to expensive noble metal catalysts traditionally used in fine chemical manufacturing.
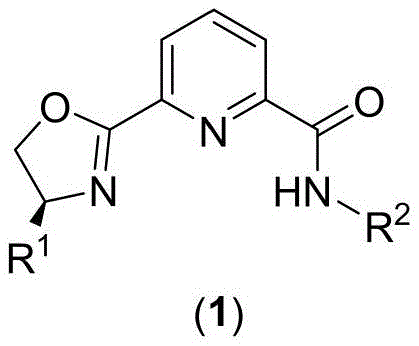
From a commercial perspective, the significance of this technology lies in its dual capability to facilitate both chiral and achiral organic synthesis with exceptional efficiency. The ligand framework is engineered to provide a rigid yet tunable coordination environment, which is essential for inducing high enantioselectivity in challenging transformations like the asymmetric hydroboration of olefins. For R&D directors and process chemists, this represents a powerful tool for accessing chiral building blocks that are pivotal in the synthesis of active pharmaceutical ingredients (APIs) and agrochemical intermediates. The patent details a streamlined synthetic pathway that achieves a total yield of over 80 percent, demonstrating immediate potential for scalable production without the bottlenecks often associated with complex ligand synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral organoboron compounds, which are indispensable intermediates in modern organic synthesis, has relied heavily on catalysts based on precious metals like Rhodium, Iridium, or Palladium. While effective, these systems suffer from severe economic and supply chain vulnerabilities, including high raw material costs, price volatility, and the stringent requirement for removing trace metal residues from final pharmaceutical products. Furthermore, many conventional oxazoline-based ligands lack the necessary structural rigidity or electronic tuning to maintain high catalytic activity when paired with cheaper, earth-abundant metals, often leading to poor conversion rates or low stereoselectivity. Traditional synthetic routes for similar ligands often involve multi-step sequences with harsh conditions, resulting in lower overall yields and generating significant chemical waste, which complicates environmental compliance and increases the cost of goods sold.
The Novel Approach
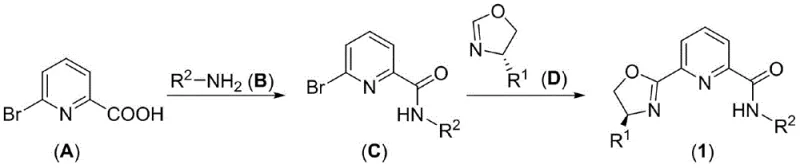
The innovative strategy outlined in the patent data circumvents these issues by introducing a pyridine-amide-oxazoline architecture that stabilizes base metal centers effectively. This approach utilizes a concise two-step synthesis starting from readily available commodity chemicals like 6-bromo-2-picolinic acid. The first step involves the formation of an amide bond, followed by a palladium-catalyzed coupling with a chiral oxazoline derivative. This route is not only operationally simple, proceeding under relatively mild conditions, but also highly efficient, as evidenced by the high yields reported in the examples. By enabling the use of iron, cobalt, and copper salts, this technology drastically reduces the dependency on critical raw materials, thereby enhancing supply chain security and reducing the overall environmental footprint of the catalytic process.
Mechanistic Insights into Earth-Abundant Metal Catalysis
The core innovation of this technology resides in the unique tridentate coordination mode of the pyridine-amide-oxazoline ligand. As illustrated in Formula (2), the ligand binds to the metal center (M) through the pyridine nitrogen, the amide nitrogen, and the oxazoline nitrogen, creating a highly stable chelate ring system. This specific geometry is crucial for maintaining the integrity of the catalyst during the rigorous conditions of hydroboration reactions. For iron and cobalt complexes, this coordination environment facilitates the activation of the boron-hydrogen bond while simultaneously controlling the approach of the olefin substrate, ensuring high regio- and enantioselectivity. The ability to fine-tune the steric bulk via the R1 and R2 substituents allows chemists to optimize the catalyst for specific substrates, minimizing side reactions and impurity formation.
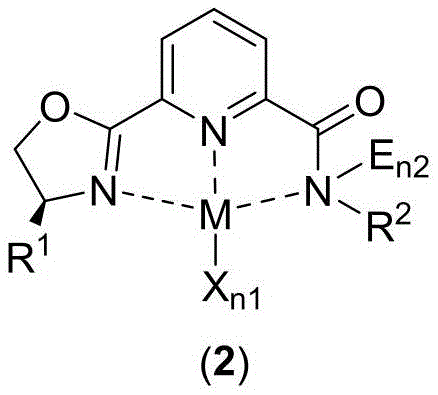
Impurity control is a paramount concern in the manufacture of high-purity pharmaceutical intermediates. The robust nature of these metal complexes ensures that the catalytic cycle proceeds with minimal decomposition, preventing the release of free metal ions that could contaminate the product stream. In the context of asymmetric hydroboration, the chiral information encoded in the oxazoline moiety is efficiently transferred to the product, as demonstrated by the high enantiomeric excess (ee) values observed in the patent examples. For instance, the iron-catalyzed hydroboration of disubstituted olefins achieved yields up to 97 percent with 98 percent ee, showcasing the superior performance of this ligand system compared to uncatalyzed or poorly ligated controls. This level of precision reduces the need for downstream purification steps like recrystallization or chiral chromatography, further driving down manufacturing costs.
How to Synthesize Pyridine-Amide-Oxazoline Ligand Efficiently
The synthesis of these high-value ligands is designed for scalability and operational simplicity, making it ideal for contract development and manufacturing organizations (CDMOs). The process begins with the activation of 6-bromo-2-picolinic acid using oxalyl chloride, followed by amidation with a substituted aniline to generate the key bromo-intermediate. This intermediate is then subjected to a cross-coupling reaction with a chiral oxazoline in the presence of a palladium catalyst and a strong base. The detailed standardized synthetic steps below outline the precise conditions required to achieve the high yields and purity specifications necessary for commercial applications.
- React 6-bromo-2-picolinic acid with oxalyl chloride and subsequently with an amine to form the 6-bromo-2-pyridineamide intermediate.
- Perform a coupling reaction between the bromo-intermediate and a chiral oxazoline using lithium tert-butoxide and a palladium catalyst.
- Purify the final ligand via column chromatography to achieve yields exceeding 80 percent with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this pyridine-amide-oxazoline ligand technology offers substantial strategic advantages beyond mere technical performance. The shift from precious metal catalysts to earth-abundant alternatives like iron and cobalt directly addresses the volatility of raw material pricing and the geopolitical risks associated with the supply of rare metals. By utilizing base metals, manufacturers can secure a more stable and predictable cost structure, insulating their operations from market fluctuations that frequently impact the bottom line of fine chemical production. Additionally, the simplified two-step synthesis of the ligand itself ensures a reliable supply of the catalyst precursor, reducing lead times and inventory holding costs.
- Cost Reduction in Manufacturing: The elimination of expensive noble metals such as rhodium or iridium from the catalytic cycle results in significant direct cost savings on raw materials. Furthermore, the high catalytic activity allows for lower catalyst loading, meaning less ligand and metal salt are required per kilogram of product. The robust nature of the catalyst also minimizes batch failures and reprocessing needs, leading to a more efficient utilization of reactor time and labor resources. Qualitatively, this translates to a drastically simplified cost model for producing chiral boron intermediates, making previously uneconomical routes viable for large-scale commercialization.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including 6-bromo-2-picolinic acid and various chiral amino alcohol derivatives, are commodity chemicals available from multiple global suppliers. This diversity in sourcing prevents single-point failures in the supply chain. The synthetic route does not rely on exotic reagents or cryogenic conditions, allowing for production in standard multipurpose chemical plants without the need for specialized infrastructure. This flexibility ensures consistent delivery schedules and the ability to rapidly scale up production volumes to meet surging market demand for complex pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, such as room temperature operations and the use of common solvents like THF and toluene, are inherently safer and easier to manage on a multi-ton scale. The high selectivity of the catalyst reduces the generation of hazardous byproducts and simplifies waste treatment protocols. By avoiding heavy metal contamination issues associated with traditional catalysts, the downstream purification process becomes less intensive, reducing solvent consumption and energy usage. This alignment with green chemistry principles supports corporate sustainability goals and facilitates regulatory approval for new drug substances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand technology in industrial settings. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance for process development teams.
Q: What metals are compatible with this pyridine-amide-oxazoline ligand system?
A: The ligand forms stable complexes with a wide range of transition metals including earth-abundant options like Iron (Fe), Cobalt (Co), Copper (Cu), Manganese (Mn), and Zinc (Zn), as well as noble metals such as Ruthenium, Rhodium, and Palladium.
Q: What is the typical yield for the ligand synthesis?
A: The patented two-step synthetic route is highly efficient, consistently delivering a total yield of over 80 percent, with individual steps often exceeding 90 percent yield under optimized conditions.
Q: Can this catalyst system be used for alkyne hydroboration?
A: Yes, specifically the copper complexes derived from this ligand demonstrate high activity and stereoselectivity in the hydroboration of alkynes to produce alkenyl boron esters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine-Amide-Oxazoline Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this pyridine-amide-oxazoline ligand technology in advancing asymmetric synthesis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand or metal complex we deliver performs consistently in your catalytic processes. We understand that reliability and quality are non-negotiable in the pharmaceutical supply chain, and our dedicated technical team is committed to supporting your long-term production needs.
We invite you to leverage our expertise to optimize your catalytic workflows and achieve superior cost efficiencies. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you evaluate the integration of these advanced ligands into your manufacturing portfolio. Let us collaborate to drive innovation and efficiency in your next generation of chiral synthesis projects.
