Advanced Synthesis of Planar Chiral Cyclophane Phosphines for High-Performance Asymmetric Catalysis
Advanced Synthesis of Planar Chiral Cyclophane Phosphines for High-Performance Asymmetric Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for highly selective and stable chiral ligands that can streamline the production of complex pharmaceutical intermediates. Patent CN101003549B introduces a significant breakthrough in this domain by disclosing a novel class of planar chiral [2.2]paracyclophane phosphine compounds and a robust synthetic methodology for their preparation. Unlike traditional chiral phosphines that often rely on central chirality and can be prone to oxidation, these compounds derive their stereochemical integrity from the rigid planar chirality of the cyclophane framework. This structural feature imparts exceptional stability and unique steric environments, making them ideal candidates for demanding catalytic transformations such as allylic amination and asymmetric hydrogenation. For R&D directors seeking reliable solutions for enantioselective synthesis, this technology offers a compelling alternative to existing ligand systems.
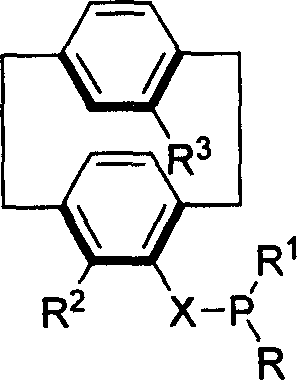
At the heart of this innovation is a versatile molecular scaffold where the phosphorus functionality is directly attached to the [2.2]paracyclophane backbone. The general structure allows for extensive tunability, with substituents R and R1 ranging from unsubstituted phenyl groups to various alkyl, aryloxy, and alkoxy groups, enabling fine-tuning of electronic and steric properties. The presence of the planar chiral element ensures that the ligand maintains its configuration under reaction conditions, a critical factor for maintaining high enantiomeric excess (ee) in the final product. By leveraging this patented architecture, manufacturers can access a new generation of catalysts that combine the reactivity of monophosphines with the durability required for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of [2.2]paracyclophane derivatives has been fraught with challenges, primarily due to the difficulty in introducing specific substituents without disrupting the delicate strained ring system. Conventional methods for preparing chiral phosphines often involve multi-step sequences with low overall yields or require expensive chiral pool starting materials that limit scalability. Furthermore, many existing planar chiral ligands suffer from poor stability upon exposure to air or moisture, necessitating rigorous handling protocols that increase operational costs and complexity in a manufacturing setting. The derivatization of cyclophanes is notoriously difficult, and achieving high optical purity typically requires cumbersome resolution processes that generate significant waste. These limitations have hindered the widespread adoption of cyclophane-based ligands in large-scale industrial applications, creating a gap in the market for more accessible and robust chiral auxiliaries.
The Novel Approach
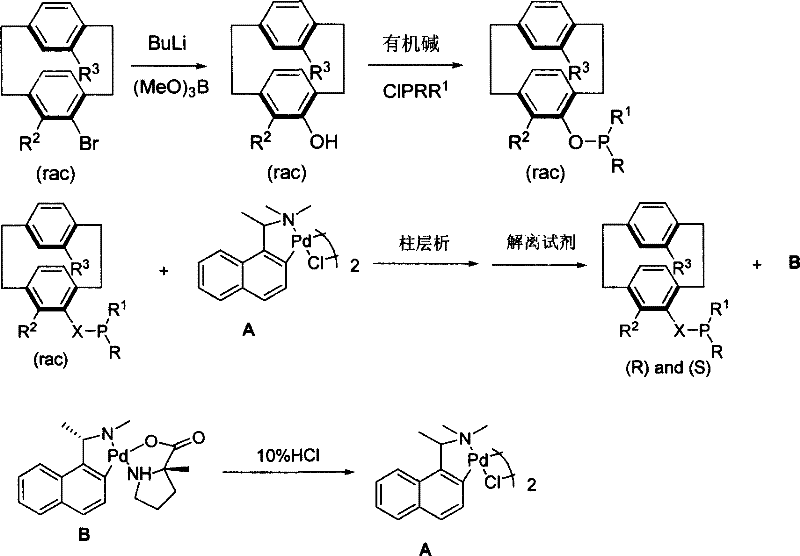
The methodology outlined in CN101003549B overcomes these hurdles through a streamlined two-stage process involving direct lithiation-phosphination followed by an elegant chiral resolution strategy. The synthesis begins with the conversion of readily available 4-bromo[2.2]paracyclophane derivatives into racemic phosphines via a controlled reaction with butyllithium and chlorophosphines. This step is highly efficient, avoiding the need for complex protecting group strategies. Subsequently, the racemic mixture is subjected to resolution using a specialized chiral cyclopalladium complex (Compound A). This approach transforms the difficult separation of enantiomers into a more manageable separation of diastereomers, which can be easily isolated via standard silica gel column chromatography. The result is a scalable route to high-purity planar chiral phosphines that maintains structural integrity throughout the process.
![Synthetic route showing conversion of 4-bromo[2.2]paracyclophane to racemic phosphine and subsequent resolution](/insights/img/planar-chiral-cyclophane-phosphine-ligands-pharma-supplier-20260309002945-03.png)
Mechanistic Insights into Cyclopalladium-Mediated Chiral Resolution
The core innovation of this patent lies in the sophisticated use of a cyclopalladium complex to achieve chiral resolution, a mechanism that warrants detailed examination for its implications on process efficiency. The resolution agent, designated as Compound A, acts as a chiral selector that coordinates with the racemic phosphine ligand to form distinct diastereomeric complexes. Due to the rigid geometry of the [2.2]paracyclophane framework, the interaction between the phosphine and the palladium center creates significant steric differentiation between the (R) and (S) enantiomers. This differentiation manifests in distinct physical properties, such as solubility and polarity, allowing for their separation using conventional chromatographic techniques. The formation of these diastereomers is rapid, typically occurring within 0.5 to 3 hours at temperatures ranging from 0°C to room temperature, ensuring high throughput in a production environment.
Following the separation of the diastereomeric complexes, the optically pure phosphine ligand is released through a dissociation step using specific reagents such as sodium amino acids or diamine compounds. Crucially, this step is designed to be reversible and non-destructive to the chiral auxiliary. The spent palladium complex (Compound B) generated after dissociation can be treated with 10% hydrochloric acid to regenerate the original chiral cyclopalladium Compound A with high recovery rates, reportedly up to 98% in experimental examples. This recyclability is a major technical advantage, as it minimizes the consumption of expensive palladium resources and reduces the environmental footprint associated with heavy metal waste. For procurement teams, this closed-loop system translates to significant long-term cost stability and supply chain reliability.
How to Synthesize Planar Chiral Cyclophane Phosphines Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing these valuable ligands with high fidelity. The process starts with the lithiation of the bromo-precursor in an ether solvent at low temperatures, followed by quenching with the appropriate chlorophosphine to generate the racemic intermediate. This crude material is then purified and subjected to the resolution sequence involving the cyclopalladium complex. The entire workflow is designed to be compatible with standard laboratory and pilot plant equipment, utilizing common solvents like toluene, tetrahydrofuran, and methanol. Detailed standardized synthesis steps are provided below to guide process development teams in replicating these results.
- Perform lithiation of 4-bromo[2.2]paracyclophane derivatives with butyllithium followed by reaction with chlorophosphines to obtain racemic monophosphines.
- React the racemic phosphine with a chiral cyclopalladium compound (Compound A) to form separable diastereomeric complexes.
- Separate diastereomers via column chromatography and treat with a dissociation reagent to release the optically pure (R) or (S) phosphine ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits beyond mere technical performance. The ability to recover and reuse the chiral cyclopalladium resolving agent significantly reduces the dependency on scarce and costly palladium resources, leading to substantial cost savings in raw material procurement. Unlike processes that consume chiral auxiliaries stoichiometrically, this catalytic-like recovery model ensures that the cost per kilogram of the final ligand remains competitive even at smaller scales. Furthermore, the use of stable [2.2]paracyclophane scaffolds reduces the risk of product degradation during storage and transport, minimizing losses due to spoilage and ensuring consistent quality upon delivery to the end user.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive chiral starting materials by relying on a resolution strategy that utilizes a recoverable auxiliary. The high recovery rate of the palladium complex means that the effective cost of the chiral inducer is amortized over multiple batches, drastically lowering the variable cost of production. Additionally, the simplified purification steps, primarily relying on silica gel chromatography with standard eluents like petroleum ether and ethyl acetate, reduce the need for specialized preparative HPLC equipment, further driving down capital and operational expenditures.
- Enhanced Supply Chain Reliability: The starting material, 4-bromo[2.2]paracyclophane, is a well-established intermediate with a stable supply chain, reducing the risk of bottlenecks associated with exotic reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and standard organic solvents, ensures that production can be maintained consistently without sensitive control requirements. This reliability is critical for maintaining continuous manufacturing schedules and meeting the strict delivery timelines demanded by downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram production without fundamental changes in chemistry. The ability to recycle the palladium complex aligns with green chemistry principles by minimizing heavy metal discharge, simplifying waste treatment protocols, and ensuring compliance with increasingly stringent environmental regulations. This sustainability profile enhances the marketability of the final product to eco-conscious global partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within CN101003549B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating these ligands into their existing catalytic processes.
Q: What is the primary advantage of this cyclopalladium resolution method?
A: The method allows for the efficient separation of enantiomers through diastereomer formation, and crucially, the chiral cyclopalladium auxiliary can be recovered and recycled with high efficiency using hydrochloric acid treatment.
Q: Can these ligands be used for asymmetric hydrogenation?
A: Yes, the patent explicitly demonstrates their utility as ligands in asymmetric catalytic hydrogenation reactions, achieving high conversion rates and enantioselectivity with substrates like dehydroamino acid esters.
Q: How stable are these planar chiral phosphine compounds?
A: Unlike many traditional chiral monophosphines that suffer from oxidation issues, these [2.2]paracyclophane-based compounds exhibit high stability, making them suitable for long-term storage and robust industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Planar Chiral Phosphine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. With our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we are uniquely positioned to manufacture these complex planar chiral phosphine compounds to your exact specifications. Our facility is equipped with stringent purity specifications and rigorous QC labs to ensure that every batch meets the highest standards of optical purity and chemical integrity required for sensitive asymmetric catalysis applications. We understand the critical nature of chiral ligands in drug synthesis and are committed to delivering products that drive your R&D success.
We invite you to collaborate with us to explore the full potential of this technology for your specific application needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our supply of high-purity planar chiral cyclophane phosphines can optimize your catalytic processes and reduce your overall manufacturing costs.
