Advanced Catalytic Systems for Commercial Scale-Up of Complex Electronic Chemical Intermediates
Advanced Catalytic Systems for Commercial Scale-Up of Complex Electronic Chemical Intermediates
The rapid evolution of the organic electronics sector demands increasingly sophisticated synthetic methodologies capable of delivering high-purity aromatic compounds with precise structural control. Patent CN110003271B introduces a groundbreaking advancement in the field of transition metal-catalyzed cross-coupling, specifically targeting the synthesis of pi-conjugated aromatic systems essential for next-generation display and optoelectronic materials. This technology leverages a novel class of sterically demanding phosphine ligands, represented by general formulas (C) through (F), to drive Suzuki-Miyaura coupling reactions with unprecedented efficiency. By optimizing the electronic and steric environment around the palladium center, this process overcomes traditional limitations associated with catalyst deactivation and low molecular weight propagation. For R&D directors seeking reliable electronic chemical supplier partnerships, understanding the mechanistic nuances of this patent is critical for developing robust manufacturing routes for OLED host materials and conductive polymers.
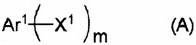
The core innovation lies in the strategic design of the ligand system, which facilitates the oxidative addition and reductive elimination steps crucial for polymer growth. Unlike conventional triphenylphosphine systems that often suffer from instability under rigorous reaction conditions, the disclosed di-tert-butyl(aryl)phosphines and dicyclohexyl(aryl)phosphines exhibit superior thermal stability and resistance to oxidation. This stability translates directly into process reliability, allowing for extended reaction times necessary to achieve high molecular weights without significant loss of catalytic activity. The versatility of the method is further evidenced by its compatibility with a wide range of substrates, including various boronic acid esters and aryl halides, enabling the construction of complex copolymer architectures required for tuning bandgaps in organic semiconductors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high molecular weight conjugated polymers has been plagued by challenges related to catalyst longevity and side reaction management. Traditional Suzuki coupling protocols often rely on simple phosphine ligands that dissociate easily from the metal center, leading to the formation of palladium black and premature termination of the polymerization chain. This phenomenon results in broad molecular weight distributions and lower than desired number-average molecular weights, which negatively impact the charge mobility and film-forming properties of the final electronic material. Furthermore, conventional methods frequently require excessive catalyst loading to compensate for deactivation, thereby increasing the residual metal content in the product. For procurement managers focused on cost reduction in electronic chemical manufacturing, the need for extensive purification steps to remove trace palladium represents a significant hidden cost and supply chain bottleneck that erodes profit margins.
The Novel Approach
The methodology outlined in CN110003271B addresses these deficiencies through the implementation of bulky, electron-rich phosphine ligands that form highly stable yet reactive palladium complexes. As illustrated in the structural definitions within the patent, ligands such as di-tert-butyl(3,5-di-tert-butylphenyl)phosphine create a protective pocket around the catalytic site. This steric shielding prevents the aggregation of palladium atoms while simultaneously promoting the dissociation of the leaving group during the oxidative addition phase. The result is a catalytic cycle that maintains high turnover frequencies over prolonged periods, enabling the synthesis of polymers with weight-average molecular weights (Mw) reaching up to 5.0 x 10^5. This dramatic improvement in performance metrics allows manufacturers to reduce lead time for high-purity electronic chemical intermediates by minimizing batch failures and ensuring consistent product quality across large-scale production runs.
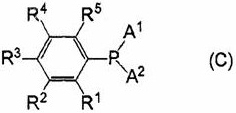
Mechanistic Insights into Bulky Phosphine-Palladium Catalysis
The efficacy of this production process is rooted in the delicate balance of steric and electronic properties imparted by the specific phosphine ligands employed. In the catalytic cycle, the bulky tert-butyl or cyclohexyl groups on the phosphorus atom exert a strong cone angle effect, which forces the coordination geometry of the palladium center to favor the monoligated species active for oxidative addition. This is particularly advantageous when reacting sterically hindered substrates, such as ortho-substituted aryl halides, which are common building blocks in advanced OLED materials. The electron-donating nature of the alkyl groups increases the electron density on the palladium, facilitating the insertion into the carbon-halogen bond. Subsequently, the same steric bulk accelerates the reductive elimination step, which is often the rate-determining step in polymerization, by destabilizing the crowded intermediate state. This dual acceleration mechanism ensures that the propagation of the polymer chain outpaces termination events, leading to the observed high molecular weights.
From an impurity control perspective, the robust nature of these ligand-metal complexes significantly suppresses common side reactions such as homocoupling of the boronic acid or deboronation. In conventional systems, free boronic acids can undergo oxidative homocoupling in the presence of oxygen or trace metal impurities, leading to structural defects in the polymer backbone that act as charge traps. The rapid transmetallation and reductive elimination promoted by the novel ligands minimize the lifetime of reactive intermediates susceptible to these pathways. Additionally, the use of specific bases and phase transfer catalysts, such as tetraethylammonium hydroxide or Aliquat 336, creates a biphasic environment that enhances the solubility of inorganic byproducts while keeping the growing polymer chain in the organic phase. This phase separation not only drives the equilibrium towards product formation but also simplifies the workup procedure, contributing to a cleaner final product profile that meets the stringent purity specifications required for display applications.
How to Synthesize High-Purity Aromatic Compounds Efficiently
The practical implementation of this technology involves a straightforward yet precisely controlled sequence of mixing and heating steps designed to maximize catalyst activation and monomer conversion. The process typically begins with the charging of the boronic acid derivative and the aryl halide monomer into a reaction vessel under an inert nitrogen atmosphere to prevent catalyst oxidation. A suitable aprotic organic solvent, such as toluene or tetrahydrofuran, is selected based on the solubility of the growing polymer chain to ensure homogeneous reaction conditions throughout the process. The detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction vessel with boronic acid derivatives and aryl halides under inert atmosphere.
- Add the specific bulky phosphine ligand and palladium source to generate the active catalytic species in situ.
- Heat the mixture in an aprotic solvent system with base to facilitate polymerization and achieve high molecular weight.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders evaluating new technologies, the adoption of this phosphine-mediated coupling process offers tangible strategic benefits beyond mere technical performance. The ability to consistently produce high molecular weight materials with narrow polydispersity reduces the variability in downstream device fabrication, thereby enhancing overall yield in the manufacturing of OLED panels or organic photovoltaics. This consistency is a key factor in establishing long-term supply agreements with major electronics manufacturers who prioritize reliability over spot pricing. Furthermore, the robustness of the catalyst system allows for flexibility in raw material sourcing, as the process tolerates a wider range of substrate purities without compromising the final product quality, providing a buffer against supply chain disruptions for specialized monomers.
- Cost Reduction in Manufacturing: The high catalytic efficiency of the bulky phosphine ligands means that significantly lower loadings of expensive palladium precursors are required to achieve complete conversion. This reduction in precious metal usage directly lowers the bill of materials cost per kilogram of product. Moreover, the suppression of side reactions minimizes the formation of difficult-to-remove impurities, which simplifies the purification workflow. Eliminating complex chromatography steps or multiple recrystallizations in favor of simpler precipitation and filtration methods drastically reduces solvent consumption and waste disposal costs, leading to substantial cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: The ligands and catalysts described are synthesized from readily available starting materials such as tert-butyl chloride and aryl bromides, ensuring a stable supply of the catalytic system itself. The process conditions, utilizing common solvents like toluene and standard inorganic bases, do not require exotic or hazardous reagents that might be subject to strict regulatory controls or shipping restrictions. This chemical simplicity enhances the resilience of the supply chain, allowing for multi-sourcing of raw materials and reducing the risk of production stoppages due to logistical bottlenecks or regulatory compliance issues associated with specialized reagents.
- Scalability and Environmental Compliance: The reaction protocol is inherently scalable, having been demonstrated effectively in batch processes that can be translated to large-scale reactors without significant re-optimization. The use of biphasic systems with aqueous bases facilitates the separation of inorganic salts, reducing the load on wastewater treatment facilities. Additionally, the high atom economy of the coupling reaction, combined with the ability to recover and recycle the organic solvent, aligns with modern green chemistry principles. This environmental compatibility simplifies the permitting process for new manufacturing lines and supports corporate sustainability goals, making the technology attractive for companies aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the operational parameters and expected outcomes for potential licensees or manufacturing partners.
Q: What are the key advantages of the phosphine ligands described in CN110003271B?
A: The ligands, particularly di-tert-butyl(aryl)phosphines, provide exceptional steric bulk and electron donation, which significantly accelerates the reductive elimination step in the catalytic cycle, leading to higher molecular weight polymers.
Q: Can this process be scaled for industrial production of OLED materials?
A: Yes, the process utilizes common solvents like toluene and robust bases, and the catalysts demonstrate high turnover numbers, making the commercial scale-up of complex electronic chemical intermediates feasible and cost-effective.
Q: How does this method improve impurity profiles compared to conventional Suzuki coupling?
A: The specialized ligand architecture suppresses homocoupling side reactions and beta-hydride elimination, resulting in a cleaner impurity spectrum and reducing the burden on downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Conjugated Polymers Supplier
The technological breakthroughs detailed in CN110003271B represent a significant leap forward in the synthesis of functional aromatic materials, yet translating laboratory success into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this transformation, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive handling of phosphine ligands and palladium catalysts under strictly controlled inert atmospheres, ensuring that the stringent purity specifications demanded by the electronics industry are met consistently. With our rigorous QC labs and advanced analytical capabilities, we guarantee that every batch of conjugated polymer delivered meets the exacting standards required for high-performance optoelectronic applications.
We invite global innovators in the display and energy sectors to collaborate with us to unlock the full potential of this proprietary technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to reach out today to request specific COA data and route feasibility assessments for your next generation of organic electronic materials, ensuring a secure and efficient supply chain for your critical components.
