Advanced Chiral Ferrocene PNO Ligands for High-Efficiency Asymmetric Catalysis and Commercial Scale-Up
Introduction to Novel Tridentate PNO Ligand Technology
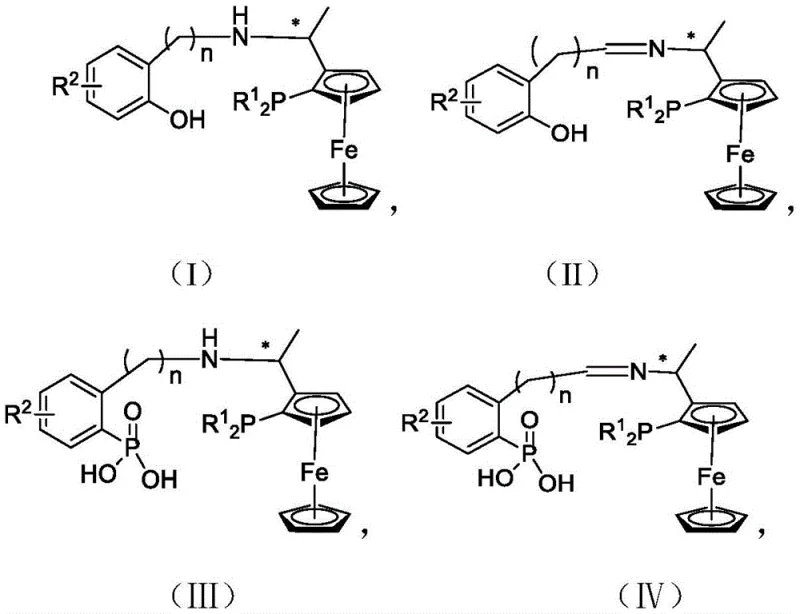
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the need for more efficient and selective chiral ligands. Patent CN112961194A introduces a groundbreaking class of tridentate PNO ligands containing planar chiral ferrocene backbones, specifically designed to overcome the limitations of traditional bidentate systems. These ligands, characterized by general formulas (I) through (VI), integrate a robust ferrocene scaffold with phenol or aryl phosphoric acid moieties, creating a unique electronic and steric environment around the catalytic center. This innovation is particularly critical for the pharmaceutical and fine chemical industries, where the demand for high-purity single-isomer intermediates continues to surge. By leveraging simple and readily available starting materials such as salicylaldehyde derivatives, this technology offers a pathway to catalysts that exhibit superior stability and enhanced bifunctional catalytic activity.
The strategic design of these ligands addresses a long-standing challenge in homogeneous catalysis: balancing high turnover numbers (TON) with exceptional enantioselectivity (ee). Unlike previous generations of ligands that often required complex, multi-step syntheses involving expensive chiral sources, this new approach simplifies the molecular architecture without compromising performance. The presence of the acidic hydroxyl group in the ligand structure plays a pivotal role, acting as a proton donor that facilitates the activation of polar double bonds in substrates like ketones and imines. For procurement managers and supply chain directors, this translates to a reliable pharmaceutical intermediate supplier capability that can support the rigorous demands of modern drug synthesis, ensuring consistent quality and reduced dependency on scarce chiral pools.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric hydrogenation has relied heavily on bidentate ligands such as BINAP or DuPhos. While effective, these systems often struggle to provide the necessary "chiral pocket" depth required for challenging substrates, leading to moderate stereoselectivity in complex molecular environments. Furthermore, many existing tridentate ligands, such as SpiroPAP, suffer from convoluted synthetic routes that involve multiple protection and deprotection steps, significantly driving up the cost of goods sold (COGS). The reliance on alkyl alcohol groups in some prior art ligands also limits the acidity and hydrogen-bonding capability of the catalyst, restricting its efficacy in reactions requiring strong substrate activation. Additionally, the stability of these conventional ligands under harsh industrial conditions can be variable, leading to catalyst decomposition and the generation of metal impurities that are difficult to remove from the final active pharmaceutical ingredient (API).
The Novel Approach
The technology disclosed in CN112961194A represents a paradigm shift by introducing a modular design based on chiral ferrocene and salicylaldehyde derivatives. This novel approach utilizes a tridentate coordination mode (P-N-O) that locks the transition metal into a rigid, well-defined geometry, minimizing conformational flexibility that can erode enantioselectivity. The incorporation of the phenolic hydroxyl group is a masterstroke of ligand design; it not only participates in coordination but also enhances the acidity of the proton, facilitating a bifunctional mechanism where the ligand actively participates in proton transfer during the catalytic cycle. This results in catalysts that are not only more active but also more robust, capable of sustaining high turnover frequencies over extended reaction times. The simplicity of the synthesis, often achievable in just a few steps from bulk chemicals, drastically reduces the barrier to entry for commercial adoption, making cost reduction in fine chemical manufacturing a tangible reality rather than just a theoretical possibility.
Mechanistic Insights into Bifunctional Asymmetric Hydrogenation
The exceptional performance of these ferrocene-based PNO ligands stems from their unique ability to engage in bifunctional catalysis. In the catalytic cycle, the electron-rich phosphorus and nitrogen atoms donate electron density to the transition metal center (such as Iridium or Rhodium), increasing its nucleophilicity and facilitating the formation of metal-hydride species. Simultaneously, the acidic proton on the phenolic oxygen acts as a Lewis acid or hydrogen bond donor, interacting strongly with the electronegative atoms (oxygen or nitrogen) in the polar double bond of the substrate. This dual activation lowers the energy barrier for the hydride transfer step, which is typically the rate-determining step in asymmetric hydrogenation. The rigid ferrocene backbone ensures that this cooperative interaction occurs within a highly chiral environment, effectively discriminating between the pro-chiral faces of the substrate to deliver products with high enantiomeric excess.
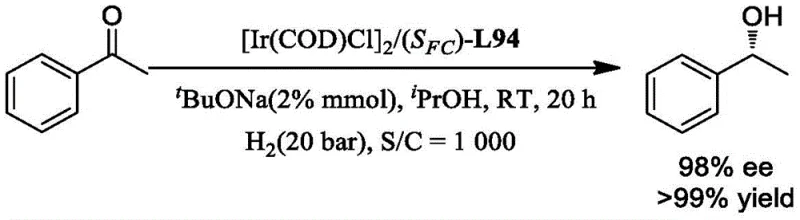
Furthermore, the modularity of the ligand allows for fine-tuning of both steric and electronic properties. By varying the substituents on the salicylaldehyde ring (e.g., introducing tert-butyl groups as seen in ligands L22 and L94), chemists can optimize the steric bulk around the metal center to accommodate different substrate sizes. This tunability is crucial for R&D directors aiming to expand the substrate scope beyond simple acetophenone to more complex pharmaceutical intermediates. The stability of the resulting metal complexes is also noteworthy; the tridentate binding mode prevents ligand dissociation, a common failure mode in bidentate systems that leads to catalyst death. This robustness ensures that the catalytic system maintains its integrity throughout the reaction, minimizing the formation of side products and simplifying downstream purification processes, which is a key consideration for maintaining stringent purity specifications in API production.
How to Synthesize Chiral Ferrocene PNO Ligands Efficiently
The synthesis of these advanced ligands is designed for operational simplicity and scalability, utilizing standard laboratory equipment and readily available reagents. The process typically begins with the condensation of a chiral ferrocene amine precursor, such as (S,R)-4, with a substituted salicylaldehyde in a solvent like ethanol. This step forms an imine intermediate which can be isolated or reduced in situ. For amine-type ligands, a subsequent reduction step using sodium borohydride (NaBH4) or lithium aluminum hydride (LiAlH4) yields the final tridentate PNO structure. The detailed standardized synthesis steps see the guide below.
- Condense chiral ferrocene amine (S,R)-4 with salicylaldehyde derivatives in ethanol under nitrogen protection to form the imine or amine linkage.
- Reduce the intermediate imine using NaBH4 or LiAlH4 to obtain the final tridentate PNO ligand with high purity.
- Complex the ligand with transition metal precursors like [Ir(COD)Cl]2 or [Rh(COD)Cl]2 in situ using a base such as tBuONa for asymmetric hydrogenation reactions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ligand technology offers compelling strategic advantages that extend beyond mere technical performance. The primary driver for cost optimization lies in the raw material sourcing; salicylaldehyde and its derivatives are commodity chemicals produced on a massive global scale, ensuring a stable and low-cost supply chain that is immune to the volatility often associated with exotic chiral building blocks. This accessibility directly translates to significant cost savings in the overall manufacturing process, as the ligand itself becomes a negligible fraction of the total production cost. Moreover, the simplified synthetic route eliminates the need for cryogenic conditions or hazardous reagents often required in traditional ligand synthesis, thereby reducing operational expenditures related to safety compliance and energy consumption.
- Cost Reduction in Manufacturing: The streamlined synthesis of these ligands eliminates multiple synthetic steps and expensive chiral auxiliaries found in older technologies. By utilizing a convergent synthesis strategy where the chiral ferrocene unit is coupled with a simple aldehyde, the overall process mass intensity (PMI) is drastically reduced. This efficiency means less solvent waste, lower energy usage for heating and cooling, and reduced labor time per batch. Consequently, the cost per kilogram of the final catalyst is substantially lower, allowing pharmaceutical manufacturers to achieve better margins on their high-value chiral intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for complex chiral ligands poses a significant risk to production continuity. The modular nature of this technology allows for the synthesis of a wide library of ligand variants (L1-L144) using a common set of precursors. This flexibility ensures that if one specific derivative faces supply constraints, alternative ligands with similar steric and electronic profiles can be rapidly deployed. Furthermore, the robustness of the ligands allows for longer shelf-life and easier storage conditions, reducing the risk of degradation during transit and warehousing, which is critical for maintaining a resilient global supply chain for critical drug substances.
- Scalability and Environmental Compliance: The catalytic systems derived from these ligands operate efficiently at relatively low catalyst loadings, with turnover numbers (TON) reaching up to 100,000 in optimized conditions as demonstrated in the patent examples. High TON means less metal waste and lower residual metal levels in the final product, simplifying the purification workflow and reducing the environmental footprint of the manufacturing process. The ability to run reactions at ambient temperatures and moderate hydrogen pressures (e.g., 20 bar) also enhances process safety and reduces the capital expenditure required for high-pressure reactor infrastructure, facilitating easier scale-up from pilot plant to commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ferrocene PNO ligand technology in industrial settings. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering a clear picture of the technology's capabilities and limitations.
Q: What are the key advantages of these tridentate PNO ligands over traditional bidentate ligands?
A: These tridentate ligands provide a deeper and more stable chiral pocket around the transition metal center. The inclusion of an acidic phenolic hydroxyl group enables a bifunctional catalytic mechanism, significantly enhancing interaction with polar double bonds and improving both activity and stereoselectivity compared to standard bidentate systems.
Q: Can these ligands be used with metals other than Iridium?
A: Yes, the patent demonstrates versatility across multiple transition metals. While Iridium and Rhodium complexes show excellent results in asymmetric hydrogenation, the ligands also successfully form stable complexes with Ruthenium, Manganese, Palladium, and Nickel, expanding their utility to various reaction types including cross-coupling and transfer hydrogenation.
Q: Is the synthesis of these ligands scalable for industrial production?
A: Absolutely. The synthesis utilizes commercially available bulk raw materials like salicylaldehyde and simple chiral ferrocene amines. The reaction conditions are mild (often room temperature or reflux in common solvents like ethanol or toluene), and the purification involves standard column chromatography or recrystallization, making the process highly amenable to large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ferrocene PNO Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial success requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to multi-ton manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral ligand or catalyst delivered meets the highest industry standards for enantiomeric excess and chemical purity.
We invite you to collaborate with our technical team to explore how this novel ligand technology can optimize your specific synthetic routes. Whether you require a Customized Cost-Saving Analysis for an existing process or need to develop a new asymmetric hydrogenation protocol from scratch, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and comprehensive quotations tailored to your volume requirements, ensuring a secure and efficient supply chain for your critical chiral intermediates.
