Scalable Synthesis of 2-(2,4,6-Trimethylphenylselenyl)-5-Methylbenzoxazole via Copper Catalysis
Introduction to Advanced Selenylation Technology
The pharmaceutical and agrochemical industries are constantly seeking efficient routes to incorporate selenium into heterocyclic frameworks, driven by the element's profound biological activity ranging from antioxidant properties to antitumor effects. Patent CN108484518B introduces a groundbreaking methodology for the synthesis of 2-(2,4,6-trimethylphenylselenyl)-5-methylbenzoxazole, a complex organoselenium compound with significant potential as a high-value intermediate. This innovation addresses long-standing challenges in organoselenium chemistry by utilizing a tandem reaction strategy that directly couples 5-methylbenzoxazole with 2,4,6-trimethyliodobenzene using elemental selenium as the selenium source. Unlike traditional methods that rely on expensive and odorous pre-functionalized selenium reagents, this protocol leverages a cooperative catalytic system involving copper acetate and ammonium bicarbonate in a polar aprotic solvent. For R&D directors and process chemists, this represents a paradigm shift towards more atom-economical and operationally simple synthetic designs that minimize waste generation while maximizing structural complexity in a single pot.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of asymmetric diaryl selenide bonds has been fraught with synthetic inefficiencies and safety concerns. Prior art, such as the work by Reddy et al. in 2009, relied heavily on the coupling of aryl halides with diaryl diselenides under strongly alkaline conditions using nano-copper oxide catalysts. While effective, this approach necessitates the prior preparation of the diaryl diselenide, adding an extra synthetic step and introducing handling issues associated with the unpleasant odor and toxicity of diselenides. Similarly, methodologies reported by Ricordi et al. utilized arylboronic acids and diaryl diselenides, which further escalates the cost of goods due to the requirement for boronic acid precursors. These conventional pathways, illustrated in earlier literature schemes, often suffer from limited substrate scope and require rigorous exclusion of moisture and oxygen, complicating the scale-up process for industrial applications.
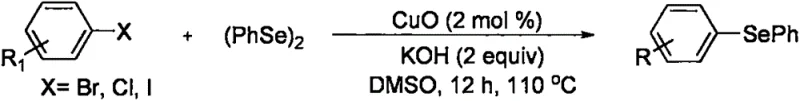
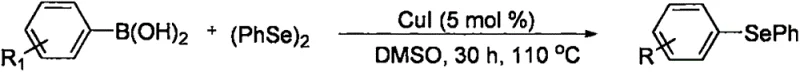
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN108484518B eliminates the need for pre-synthesized selenium reagents entirely. By employing elemental selenium powder directly alongside an aryl iodide and the benzoxazole substrate, the process achieves a direct C-H bond arylselenylation. This tandem reaction is promoted by a specific combination of copper acetate and ammonium bicarbonate, creating a highly active catalytic species in situ. The use of elemental selenium not only drastically reduces raw material costs but also simplifies the supply chain by removing the dependency on specialized selenium intermediates. Furthermore, the reaction proceeds efficiently in DMF at 140°C under a nitrogen atmosphere, demonstrating remarkable tolerance and yielding the target 2-(2,4,6-trimethylphenylselenyl)-5-methylbenzoxazole in high purity. This streamlined approach effectively bypasses the multi-step sequences typical of older protocols, offering a direct route from commodity chemicals to high-value fine chemical intermediates.
Mechanistic Insights into Copper-Catalyzed C-H Selenylation
The success of this transformation hinges on the unique interplay between the copper catalyst and the reaction components. Mechanistically, it is proposed that the copper(II) acetate species undergoes reduction in situ to generate an active copper(I) or copper(0) species capable of oxidative addition into the carbon-iodine bond of the 2,4,6-trimethyliodobenzene. Simultaneously, the elemental selenium interacts with the copper center to form a reactive copper-selenium intermediate. The presence of ammonium bicarbonate is critical, likely serving as a mild base that facilitates the deprotonation of the C2-position of the 5-methylbenzoxazole or assists in the regeneration of the active catalyst without causing decomposition of the sensitive selenium species. This cooperative catalysis allows for the cleavage of the inert C-H bond on the benzoxazole ring and the subsequent formation of the C-Se bond. The specificity of this mechanism is evidenced by the failure of other bases; strong bases like tert-butoxides or carbonates lead to no reaction, suggesting that the delicate balance of basicity provided by ammonium bicarbonate is essential for maintaining the catalytic cycle and preventing side reactions.
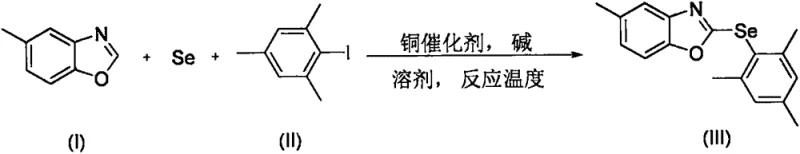
Regarding impurity control, the choice of solvent and temperature plays a pivotal role in ensuring a clean reaction profile. The use of DMF as the solvent provides the necessary polarity to solubilize the inorganic salts and selenium while stabilizing the charged intermediates involved in the catalytic cycle. Operating at 140°C ensures sufficient energy to overcome the activation barrier for C-H activation but remains below the threshold where significant thermal decomposition of the product or reagents occurs. The high selectivity observed, particularly the exclusive functionalization at the desired position without over-selenylation or degradation of the methyl groups, indicates a highly ordered transition state. This level of control is crucial for pharmaceutical manufacturing, where the removal of selenium-containing byproducts can be notoriously difficult. The protocol's ability to deliver high-purity product after simple extraction and chromatography underscores the robustness of the mechanistic design, minimizing the burden on downstream purification processes.
How to Synthesize 2-(2,4,6-Trimethylphenylselenyl)-5-Methylbenzoxazole Efficiently
To implement this synthesis effectively, precise adherence to the molar ratios and reaction conditions specified in the patent is essential. The process begins with the careful weighing of 5-methylbenzoxazole, 2,4,6-trimethyliodobenzene, and elemental selenium in a molar ratio of approximately 1:3:3, ensuring an excess of the selenium source and aryl iodide to drive the equilibrium forward. The addition of copper acetate (10 mol%) and ammonium bicarbonate (3 equivalents) into the reaction vessel containing DMF solvent must be performed under an inert nitrogen atmosphere to prevent oxidation of the selenium species. Detailed standard operating procedures for the heating, stirring, and workup phases are critical for reproducibility.
- Combine 5-methylbenzoxazole, 2,4,6-trimethyliodobenzene, elemental selenium, copper acetate catalyst, and ammonium bicarbonate in DMF solvent under nitrogen.
- Heat the reaction mixture to 140°C and stir for 24 hours to facilitate the C-H bond arylselenylation tandem reaction.
- Cool the mixture, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical advantages over traditional selenylation methods. The primary driver for cost reduction is the substitution of expensive, pre-functionalized diaryl diselenides or arylboronic acids with commodity-grade elemental selenium and aryl iodides. Elemental selenium is widely available as a bulk chemical, significantly lowering the raw material entry cost per kilogram of the final product. Furthermore, the elimination of the preliminary step to synthesize the selenium reagent reduces labor costs, reactor occupancy time, and solvent consumption, leading to a substantially simplified manufacturing workflow. The high yield reported in the scale-up example (90%) further enhances the overall process economics by maximizing the throughput of the existing infrastructure and minimizing waste disposal costs associated with low-yielding reactions.
- Cost Reduction in Manufacturing: The strategic use of elemental selenium as a direct reagent removes the markup associated with specialized organoselenium intermediates. By consolidating the synthesis into a single tandem reaction, the process eliminates the need for separate reactor campaigns dedicated to reagent preparation. This consolidation translates to reduced utility consumption and lower capital expenditure requirements for equipment, as fewer unit operations are needed to achieve the final API intermediate. Additionally, the use of inexpensive copper acetate and ammonium bicarbonate as promoters ensures that the catalyst and additive costs remain negligible relative to the value of the output.
- Enhanced Supply Chain Reliability: Relying on broadly available starting materials like elemental selenium and substituted iodobenzenes mitigates the risk of supply chain disruptions often associated with niche fine chemicals. These commodities are produced by multiple global suppliers, ensuring competitive pricing and consistent availability. The robustness of the reaction conditions, specifically the tolerance for standard organic solvents like DMF, means that the process can be executed in most standard multipurpose chemical plants without requiring specialized corrosion-resistant equipment often needed for harsh acidic or basic selenylation protocols. This flexibility allows for easier technology transfer and multi-site manufacturing capabilities.
- Scalability and Environmental Compliance: The patent data explicitly demonstrates successful scale-up from milligram to gram scales with maintained efficiency, indicating a clear path to kilogram and ton-scale production. The simplified workup procedure, involving standard extraction and chromatography, avoids the generation of complex aqueous waste streams laden with heavy metals or toxic selenium byproducts that are difficult to treat. By improving atom economy and reducing the number of synthetic steps, the overall E-factor (environmental factor) of the process is significantly improved, aligning with modern green chemistry principles and facilitating easier regulatory approval for environmental discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this copper-catalyzed selenylation technology. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing clarity on critical process parameters. Understanding these nuances is vital for process engineers aiming to adapt this methodology for specific production environments or to troubleshoot potential variations in raw material quality.
Q: Why is elemental selenium preferred over diaryl diselenides in this synthesis?
A: Elemental selenium is significantly more cost-effective and atom-economical compared to pre-synthesized diaryl diselenides, which require additional preparation steps and generate more waste.
Q: What is the critical role of ammonium bicarbonate in this reaction system?
A: Screening data indicates that ammonium bicarbonate is uniquely effective as a base promoter; other common bases like carbonates or phosphates resulted in no reaction, suggesting a specific mechanistic requirement for this mild base.
Q: Can this process be scaled for industrial production?
A: Yes, the patent demonstrates successful scale-up in Example 2, achieving a 90% isolated yield on a multi-gram scale, indicating robustness suitable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2,4,6-Trimethylphenylselenyl)-5-Methylbenzoxazole Supplier
As the demand for selenium-containing heterocycles grows in the pharmaceutical and agrochemical sectors, having a partner with deep technical expertise in C-H functionalization is invaluable. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 2-(2,4,6-trimethylphenylselenyl)-5-methylbenzoxazole meets the highest standards required for drug substance manufacturing. We understand the critical nature of intermediate quality on the final API performance and are committed to delivering products that facilitate smooth downstream processing.
We invite you to engage with our technical team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this elemental selenium-based protocol for your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project timelines, helping you secure a reliable and cost-effective source for this high-value intermediate.
