Advanced Synthesis Of Acetyl Chroman Intermediates For Commercial Anti-Tumor Drug Development
Advanced Synthesis Of Acetyl Chroman Intermediates For Commercial Anti-Tumor Drug Development
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for complex heterocyclic intermediates that serve as the backbone for next-generation therapeutics. Patent CN114656438A introduces a significant advancement in the organic synthesis of 5,7-dihydroxy-2,2-dimethyl-6-acetyl-chroman, a critical intermediate belonging to the class of A-ring fused pyran flavonoids. This specific chemical architecture is renowned for its potent biological activities, including anti-tumor, anti-cardiovascular, and anti-viral properties, making it a highly sought-after building block for medicinal chemists. The disclosed method provides a novel pathway that not only achieves the target molecular structure with high fidelity but also yields a stable crystalline form, which is essential for downstream processing and formulation. For R&D directors and procurement specialists, understanding the nuances of this patent is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials for clinical and commercial applications.
Furthermore, the strategic importance of this synthesis lies in its potential to streamline the production of flavonoid-based drugs that are currently limited by natural extraction yields. Natural sources of A-ring fused pyran flavonoids, such as Morusin, often suffer from low abundance and complex isolation procedures, creating supply chain bottlenecks. By establishing a synthetic route that bypasses these natural limitations, manufacturers can ensure a consistent and uninterrupted supply of key intermediates. This patent details a method that is not only chemically efficient but also operationally practical, utilizing standard laboratory reagents and conditions that translate well to industrial reactors. The ability to produce this acetyl chroman derivative synthetically opens new avenues for the development of anti-tumor medicaments, addressing the growing global demand for effective cancer therapies while mitigating the risks associated with raw material scarcity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of complex flavonoid intermediates has relied heavily on extraction from medicinal plants, a process fraught with inconsistencies and inefficiencies that hinder large-scale pharmaceutical manufacturing. The natural content of A-ring fused pyran flavonoids in plant sources is typically extremely low, necessitating the processing of vast quantities of biomass to obtain minute amounts of the target compound. This approach is not only economically unsustainable due to high labor and material costs but also environmentally taxing, generating significant organic waste. Moreover, natural extracts often contain a complex matrix of impurities that are structurally similar to the target molecule, making purification to pharmaceutical-grade standards a formidable challenge that can drastically extend lead times. For a procurement manager, relying on such sources introduces unacceptable volatility into the supply chain, where crop failures or seasonal variations can abruptly halt production.
In addition to sourcing issues, existing synthetic methods for acetyl chroman derivatives have often been limited by harsh reaction conditions or the use of expensive, toxic catalysts that complicate the purification workflow. Many conventional routes require multiple protection and deprotection steps to manage the reactivity of phenolic hydroxyl groups, which reduces overall yield and increases the generation of chemical waste. The presence of residual heavy metals from catalytic processes is a particular concern for regulatory compliance, as it necessitates additional downstream processing to meet stringent purity specifications for human use. These technical barriers have historically slowed the commercialization of promising flavonoid-based therapeutics, creating a gap between laboratory discovery and market availability that this new patent aims to bridge through a more direct and efficient synthetic strategy.
The Novel Approach
The methodology outlined in patent CN114656438A represents a paradigm shift by offering a direct and efficient synthetic route that circumvents the pitfalls of both natural extraction and traditional multi-step synthesis. This novel approach utilizes a straightforward alkylation followed by an acid-catalyzed cyclization, significantly reducing the number of operational steps required to construct the chroman core. By selecting 2,4,6-trihydroxyacetophenone and 1-bromo-3-methyl-2-butene as starting materials, the process leverages readily available and cost-effective reagents that are stable and easy to handle on a large scale. The reaction conditions are optimized to promote high conversion rates without the need for exotic catalysts or extreme temperatures, thereby simplifying the engineering requirements for commercial scale-up of complex pharmaceutical intermediates. This efficiency translates directly into cost reduction in anti-tumor intermediate manufacturing, making the final drug product more accessible.
Moreover, this new method specifically addresses the issue of product isolation by facilitating the formation of a well-defined crystalline structure. The ability to crystallize the 5,7-dihydroxy-2,2-dimethyl-6-acetyl-chroman directly from the reaction mixture or through a simple solvent exchange process is a major advantage for quality control. Crystalline materials generally exhibit superior stability and handling characteristics compared to amorphous oils or gums, which simplifies storage and transportation logistics for the supply chain head. The process also incorporates a purification step using silica gel column chromatography with a specific eluent system, ensuring that the final product meets the rigorous purity standards required for pharmaceutical applications. This combination of synthetic efficiency and robust isolation techniques positions this patent as a cornerstone for developing a reliable supply chain for high-value flavonoid intermediates.
Mechanistic Insights into K2CO3-Mediated Alkylation and Cyclization
The core of this synthetic innovation lies in the precise control of reaction kinetics during the alkylation and subsequent cyclization phases, which dictates the purity and yield of the final acetyl chroman. The process begins with the deprotonation of the phenolic hydroxyl groups on 2,4,6-trihydroxyacetophenone using anhydrous potassium carbonate in a tetrahydrofuran solvent system. This base-mediated activation is critical for generating the nucleophilic phenoxide species required to attack the electrophilic carbon of the 1-bromo-3-methyl-2-butene. The stoichiometry is carefully balanced at a molar ratio of 1:2:1 for the acetophenone, base, and bromide, respectively, to ensure complete conversion while minimizing side reactions such as over-alkylation or polymerization of the alkene. The use of dry THF is essential to prevent hydrolysis of the alkylating agent and to maintain the reactivity of the base throughout the extended reflux period of 6.0 to 6.5 hours.
Following the initial alkylation, the intermediate undergoes an acid-catalyzed intramolecular cyclization to close the pyran ring, forming the characteristic chroman structure. The addition of hydrochloric acid promotes the protonation of the alkene moiety, facilitating a nucleophilic attack by the adjacent phenolic oxygen to complete the heterocyclic ring system. This step is performed under reflux for 1 to 1.5 hours, a duration that is optimized to drive the equilibrium towards the cyclic product without degrading the sensitive acetyl group. The mechanistic pathway avoids the formation of difficult-to-remove regioisomers, which is a common issue in flavonoid synthesis. By controlling the acidity and temperature, the process ensures that the acetyl group remains intact at the 6-position, preserving the biological activity profile of the molecule. 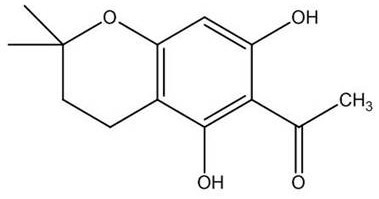
How to Synthesize 5,7-Dihydroxy-2,2-Dimethyl-6-Acetyl-Chroman Efficiently
Implementing this synthesis in a production environment requires strict adherence to the procedural parameters defined in the patent to ensure batch-to-batch consistency and regulatory compliance. The process is designed to be robust, utilizing standard chemical engineering unit operations such as reflux, distillation, and filtration, which are familiar to most contract development and manufacturing organizations. The initial alkylation step demands careful monitoring of temperature and addition rates to manage the exotherm and ensure safety, while the subsequent workup involves standard liquid-liquid extraction techniques to remove inorganic salts and byproducts. The final purification via silica gel chromatography, while laboratory-scale in the patent examples, can be adapted to preparative HPLC or recrystallization strategies for larger volumes depending on the required purity profile. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-value pathway.
- Dissolve 2,4,6-trihydroxyacetophenone in dry THF with anhydrous K2CO3 and reflux before adding 1-bromo-3-methyl-2-butene.
- Maintain reflux for 6.0 to 6.5 hours to ensure complete alkylation, then evaporate solvent under reduced pressure.
- Perform acid-catalyzed cyclization using hydrochloric acid under reflux for 1 to 1.5 hours, followed by silica gel column purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers substantial strategic benefits that extend beyond simple chemical transformation. The primary advantage is the significant simplification of the raw material portfolio, as the key starting materials are commodity chemicals with established global supply chains, reducing the risk of sourcing bottlenecks. Unlike processes that rely on specialized chiral catalysts or rare earth metals, this method utilizes inexpensive inorganic bases and common organic solvents, which drastically lowers the direct material costs and simplifies waste management protocols. This economic efficiency allows for more competitive pricing models in the final drug substance, providing a clear advantage in cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and complex protection group strategies results in a leaner manufacturing process with lower operational expenditures. By avoiding the need for specialized metal scavenging steps to meet residual metal limits, the downstream processing costs are significantly reduced, contributing to substantial cost savings. The high atom economy of the alkylation-cyclization sequence ensures that a greater proportion of the raw materials are converted into the desired product, minimizing waste disposal fees and maximizing yield efficiency. These factors combine to create a highly cost-effective production model that is sustainable for long-term commercial manufacturing.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 2,4,6-trihydroxyacetophenone and 1-bromo-3-methyl-2-butene ensures that production schedules are not held hostage by the availability of niche reagents. These chemicals are produced by multiple suppliers globally, providing redundancy and flexibility in sourcing strategies that protect against market volatility. The synthetic nature of the process also decouples production from agricultural cycles and environmental factors that affect plant-based extraction, guaranteeing a consistent and predictable supply of the intermediate. This stability is crucial for reducing lead time for high-purity pharmaceutical intermediates and ensuring uninterrupted drug development timelines.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, utilizing standard reflux and atmospheric pressure operations that can be easily transferred from laboratory glassware to industrial stainless steel reactors. The solvent system, primarily based on THF and ethyl acetate, is well-understood in terms of recovery and recycling, allowing for the implementation of closed-loop solvent management systems that minimize environmental impact. The absence of heavy metals and the use of benign inorganic salts simplify the treatment of process effluents, ensuring compliance with increasingly stringent environmental regulations. This ease of scale-up facilitates the commercial scale-up of complex flavonoid intermediates from kilogram to multi-ton quantities without requiring significant capital investment in specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific acetyl chroman intermediate. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your existing drug development pipeline. The information covers aspects of scalability, purity, and biological application to provide a comprehensive overview of the technology's value proposition.
Q: What are the primary advantages of this synthesis route for commercial production?
A: The method utilizes readily available starting materials like 2,4,6-trihydroxyacetophenone and avoids expensive transition metal catalysts, significantly simplifying the purification process and reducing overall manufacturing costs while ensuring high purity suitable for pharmaceutical applications.
Q: How does this intermediate contribute to anti-tumor drug development?
A: This acetyl chroman derivative serves as a critical A-ring fused pyran flavonoid intermediate, which has been verified to exhibit inhibitory effects on various human cancer cell lines including colon, lung, breast, and liver cancer cells, making it a valuable precursor for novel anti-tumor medicaments.
Q: Is the process scalable for industrial manufacturing?
A: Yes, the reaction conditions involving standard reflux in THF and common acid catalysis are highly amenable to scale-up, allowing for consistent production from kilogram to multi-ton scales without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,7-Dihydroxy-2,2-Dimethyl-6-Acetyl-Chroman Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving anti-tumor medications. Our team of expert chemists has thoroughly analyzed the synthetic pathway described in patent CN114656438A and is fully prepared to execute this route with the highest standards of quality and efficiency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met whether you are in the pre-clinical phase or full-scale commercialization. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5,7-dihydroxy-2,2-dimethyl-6-acetyl-chroman meets the exacting requirements of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. By leveraging our technical expertise and manufacturing capabilities, you can secure a stable source of this vital intermediate while achieving significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our partnership can drive value and innovation in your anti-tumor drug programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
