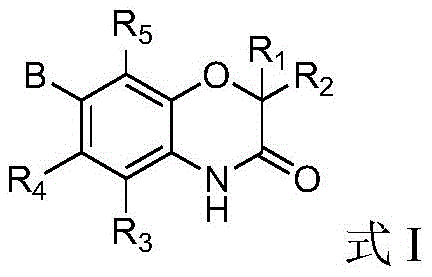
Scalable Synthesis of Novel Benzomorpholone Derivatives for Next-Generation Oncology Therapeutics
The pharmaceutical industry is constantly seeking novel scaffolds to overcome resistance in oncology treatments, particularly those targeting epigenetic regulators. Patent CN107459494B introduces a groundbreaking class of benzomorpholone derivatives that exhibit potent selective inhibition against BET (bromodomain and extra-terminal) proteins. These compounds represent a significant advancement in the development of antitumor agents, offering a unique structural framework that differs from existing clinical candidates. The patent details a robust synthetic methodology that allows for extensive structural diversification, enabling medicinal chemists to optimize pharmacokinetic properties and binding affinity efficiently. For global pharmaceutical developers, accessing these high-quality intermediates is crucial for accelerating preclinical and clinical programs aimed at treating hematological malignancies and solid tumors.
As a leading manufacturer, we recognize the strategic importance of this chemical space. The benzomorpholone core provides a rigid yet adaptable structure that facilitates specific interactions with the acetyl-lysine binding pocket of BET proteins. This specificity is vital for minimizing off-target effects, a common challenge in epigenetic therapy. By leveraging the synthetic routes disclosed in the patent, we can provide reliable partners with a steady supply of these critical building blocks, ensuring continuity in drug discovery pipelines. The versatility of the substitution patterns allowed on the aromatic rings and the side chains further enhances the potential for developing next-generation therapeutics with improved safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of heterocyclic BET inhibitors often relies on cumbersome multi-step sequences involving expensive palladium-catalyzed cross-couplings or harsh cyclization conditions that limit functional group tolerance. Many existing routes require protected intermediates and cryogenic temperatures, which drastically increase operational costs and complicate scale-up efforts in a GMP environment. Furthermore, the purification of final products from heavy metal residues poses significant regulatory hurdles and extends lead times for batch release. These inefficiencies create bottlenecks in the supply chain, making it difficult for pharmaceutical companies to secure sufficient quantities of high-purity material for toxicology studies and early-phase clinical trials without incurring prohibitive expenses.
The Novel Approach
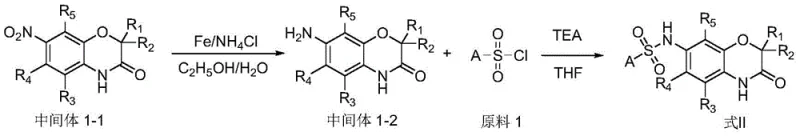
The methodology described in the patent offers a streamlined alternative by utilizing a direct cyclization strategy between substituted aminophenols and alpha-bromo esters. This approach eliminates the need for precious metal catalysts, relying instead on abundant base metals like iron for subsequent reduction steps. The reaction conditions are remarkably mild, typically proceeding at temperatures between room temperature and 90°C in common polar aprotic solvents. This simplicity not only reduces the environmental footprint but also significantly lowers the cost of goods sold (COGS). The ability to introduce diverse sulfonyl or acyl groups in the final step allows for rapid generation of analog libraries, facilitating structure-activity relationship (SAR) studies without the need for redesigning the entire synthetic pathway.
Mechanistic Insights into Base-Mediated Cyclization and Reduction
The core formation of the benzomorpholone ring system proceeds through a nucleophilic substitution mechanism where the phenolic oxygen attacks the electrophilic carbon of the alpha-bromo ester. This intramolecular cyclization is driven by the presence of a strong base, such as potassium carbonate, which deprotonates the phenol to enhance its nucleophilicity. The reaction kinetics are favorable in polar solvents like DMF, which stabilize the transition state and solubilize the inorganic salts. Following ring closure, the nitro group serves as a versatile handle for further functionalization. The reduction step utilizes iron powder in an acidic medium, a classic yet highly effective method that converts the nitro group to an amine without affecting the sensitive lactam moiety of the morpholone ring. This chemoselectivity is paramount for maintaining the integrity of the core scaffold during modification.
Subsequent functionalization at the amine position involves nucleophilic attack on sulfonyl chlorides or activated carboxylic acids. In the case of sulfonamide formation, triethylamine acts as a proton scavenger, driving the equilibrium towards product formation. For amide coupling, the use of coupling reagents like HATU ensures high efficiency even with sterically hindered substrates. This mechanistic robustness ensures consistent quality across different batches, a critical factor for regulatory compliance. The detailed understanding of these reaction pathways allows for precise control over impurity profiles, ensuring that the final intermediates meet the stringent purity specifications required for pharmaceutical applications. By optimizing stoichiometry and addition rates, side reactions such as over-alkylation or hydrolysis are effectively minimized.
How to Synthesize Benzomorpholone Derivatives Efficiently
The synthesis of these valuable intermediates follows a logical three-stage progression that balances yield with operational simplicity. Initially, the construction of the heterocyclic core is achieved through thermal cyclization, followed by a heterogeneous reduction to unveil the reactive amine. Finally, the installation of the pharmacophore is accomplished via acylation or sulfonylation. This modular approach allows manufacturers to stockpile the common amino-intermediate and rapidly diverge into multiple final compounds based on demand. Detailed standardized operating procedures for each transformation have been established to ensure reproducibility and safety during scale-up operations.
- Cyclization of substituted aminophenols with alpha-bromo esters using potassium carbonate in DMF at 90°C to form the benzomorpholone core.
- Reduction of the nitro-intermediate using iron powder and ammonium chloride in an ethanol-water mixture under reflux conditions.
- Final coupling of the amino-intermediate with sulfonyl chlorides or carboxylic acids using triethylamine or HATU/DIPEA to yield the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic benefits regarding cost stability and sourcing reliability. The reliance on commodity chemicals such as iron powder, potassium carbonate, and common organic solvents insulates the production process from the volatility associated with specialized reagents. This fundamental shift in raw material sourcing mitigates the risk of supply disruptions that often plague complex pharmaceutical syntheses dependent on single-source catalysts. Furthermore, the elimination of heavy metal catalysts simplifies the downstream processing, reducing the need for expensive scavenging resins and extensive analytical testing for residual metals.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing expensive transition metal catalysts with economical iron powder for the reduction step. This substitution removes the necessity for costly metal removal protocols and reduces the overall reagent expenditure per kilogram of product. Additionally, the high atom economy of the cyclization step minimizes waste generation, leading to lower disposal costs and improved process efficiency. The use of standard laboratory equipment without the need for specialized high-pressure or cryogenic reactors further decreases capital expenditure requirements for manufacturing facilities.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials like substituted nitrophenols and alpha-bromo esters, the supply chain becomes more resilient to market fluctuations. These precursors are produced by multiple global suppliers, preventing bottlenecks caused by vendor consolidation. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without extensive re-validation, ensuring business continuity. This flexibility allows for agile response to sudden increases in demand from clinical partners, securing the timeline for drug development programs.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been demonstrated to perform consistently from gram to kilogram scales without loss of yield or purity. The avoidance of hazardous reagents and the use of recyclable solvents align with green chemistry principles, facilitating easier regulatory approval and reducing the environmental impact of manufacturing operations. Waste streams are less toxic and easier to treat, lowering the compliance burden on environmental health and safety teams. This sustainable approach not only meets current regulatory standards but also future-proofs the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzomorpholone derivatives. Understanding these aspects helps stakeholders make informed decisions about integrating these intermediates into their development pipelines. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation.
Q: What is the primary therapeutic target of these benzomorpholone derivatives?
A: These compounds are designed as small molecule inhibitors targeting the Bromodomain and Extra-Terminal (BET) family of proteins, specifically showing activity against BRD2, BRD3, BRD4, and BRDT, which are critical in epigenetic regulation and tumor proliferation.
Q: How does this synthesis method improve upon conventional routes?
A: The patented method utilizes readily available starting materials like substituted aminophenols and alpha-bromo esters, avoiding complex multi-step sequences. It employs mild reaction conditions and standard reagents like iron powder for reduction, significantly simplifying purification and enhancing overall yield.
Q: Are these intermediates suitable for large-scale commercial production?
A: Yes, the process avoids expensive transition metal catalysts and uses robust solvents like DMF, ethanol, and THF. The operational simplicity, including straightforward work-up procedures like filtration and crystallization, makes it highly amenable to kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzomorpholone Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications for complex heterocyclic intermediates. We understand the critical nature of oncology drug development and are committed to providing a seamless supply of high-quality benzomorpholone derivatives that adhere to global regulatory standards. Our technical team works closely with clients to optimize processes for maximum efficiency and cost-effectiveness.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to specific COA data and comprehensive route feasibility assessments that validate the viability of your supply chain. Let us support your innovation with reliable manufacturing capabilities and deep chemical expertise, ensuring your BET inhibitor programs advance without interruption. Reach out today to discuss how we can accelerate your path to clinical success.
