Advanced Oxidation Control for High-Purity Rosuvastatin Calcium Intermediate Manufacturing
Advanced Oxidation Control for High-Purity Rosuvastatin Calcium Intermediate Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes that guarantee exceptional purity while maintaining economic viability, particularly for high-volume cardiovascular medications like Rosuvastatin calcium. Patent CN109456300B introduces a groundbreaking preparation method for a critical Rosuvastatin calcium intermediate, specifically addressing the persistent issue of oxidation by-products that have plagued previous manufacturing attempts. This technical disclosure outlines a sophisticated TEMPO-catalyzed oxidation process that successfully suppresses the formation of the structurally similar carboxylic acid impurity, designated as Compound I', to levels below 0.05%. By achieving such stringent impurity control, this innovation not only enhances the quality of the intermediate industrialized production finished product but also provides substantial support for the regulatory registration and quality improvement of the final Rosuvastatin calcium medicament. For global supply chain leaders, this represents a pivotal shift towards more reliable and consistent sourcing of high-value pharmaceutical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tert-butyl 2-((4R,6S)-6-formyl-2,2-dimethyl-1,3-dioxane-4-yl)acetate, known as Compound I, has been fraught with significant technical hurdles regarding yield and purity profiles. Prior art, including patents such as CN1035022234A and CN102186869A, typically relied on oxidation strategies that lacked precise selectivity control, leading to the unavoidable generation of oxidation impurities. These impurities, specifically the over-oxidized carboxylic acid derivative Compound I', possess a chemical structure remarkably similar to the target aldehyde, making their separation via standard purification techniques extremely difficult and costly. Experimental data from these conventional routes indicated that the content of such impurities could reach alarming levels of 10% to 25%, severely compromising the quality of the pure product and necessitating complex, yield-depleting purification steps. This high impurity load not only increased the cost of goods sold but also introduced significant variability into the supply chain, posing risks for downstream API manufacturers who require consistent raw material specifications.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a highly controlled reaction environment to dictate the selectivity of the oxidation transformation. The core of this innovation lies in the meticulous adjustment of the reaction pH to a weakly alkaline state, specifically within the range of 7 to 9, during the oxidation of the precursor alcohol, Compound II. By employing a catalytic system comprising tetramethylpiperidine oxynitride (TEMPO) and a carefully prepared sodium hypochlorite solution, the process effectively halts the oxidation at the aldehyde stage, preventing further conversion to the carboxylic acid. 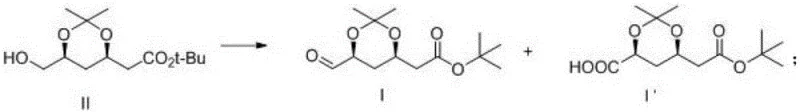
This strategic modulation of reaction conditions ensures that the content of the troublesome impurity Compound I' is rigorously controlled to be less than or equal to 0.05%, a dramatic improvement over the double-digit percentages seen in prior art. Furthermore, the process demonstrates remarkable flexibility regarding reaction temperature, operating efficiently between -25°C and 25°C, and accommodates various organic solvents such as dichloromethane, chloroform, ethyl acetate, and toluene. This versatility allows manufacturers to optimize the process for their specific equipment and safety protocols without sacrificing the critical purity metrics required for pharmaceutical grade intermediates.
Mechanistic Insights into TEMPO-Catalyzed Selective Oxidation
The success of this synthetic route is fundamentally rooted in the mechanistic behavior of the TEMPO-mediated oxidation cycle under buffered alkaline conditions. In this catalytic system, TEMPO acts as a stable nitroxyl radical that facilitates the transfer of oxygen to the primary alcohol substrate, Compound II, converting it into the desired aldehyde, Compound I. The critical factor preventing over-oxidation is the precise pH control; in strongly alkaline environments, aldehydes are prone to hydrate formation and subsequent oxidation to carboxylic acids, but by maintaining the pH between 7 and 9, the concentration of the reactive aldehyde hydrate species is minimized. Additionally, the use of a sodium hypochlorite solution that has been pre-adjusted to a pH of 8.5-8.7 at low temperatures ensures that the oxidant is introduced in a controlled manner, avoiding local hotspots of high oxidative potential that could drive the reaction toward the thermodynamic carboxylic acid product. This kinetic control is essential for stopping the reaction at the intermediate aldehyde stage, thereby preserving the structural integrity of the sensitive dioxane ring system present in the molecule.
From an impurity control perspective, the mechanism also accounts for the fate of any Compound I' that is inadvertently formed. The patent elucidates that while the primary goal is prevention, the process design acknowledges the inevitability of trace by-product formation and incorporates a robust workup procedure to manage it. During the aqueous workup, the carboxylic acid impurity I' exhibits different solubility characteristics compared to the neutral aldehyde Compound I, preferentially partitioning into the aqueous phase when the system is treated appropriately. This physicochemical difference allows for a highly effective separation where the organic phase retains the high-purity target compound, while the aqueous phase concentrates the impurity. This mechanistic understanding of phase distribution is crucial for R&D directors aiming to replicate the process, as it highlights that purity is achieved not just through reaction selectivity but also through intelligent downstream processing that leverages the acid-base properties of the impurity.
How to Synthesize Rosuvastatin Intermediate Efficiently
The practical implementation of this high-purity synthesis requires strict adherence to the optimized parameters regarding reagent preparation and addition rates to ensure reproducibility on a commercial scale. The process begins with the careful preparation of the oxidant, where sodium hypochlorite is mixed with water and cooled to 0-5°C before a two-step pH adjustment involving hydrochloric acid and solid sodium bicarbonate to reach the target window of 8.5-8.7. This pre-conditioning of the oxidant is vital to prevent shock to the reaction system upon addition. Subsequently, the substrate Compound II is dissolved in a suitable solvent like dichloromethane along with catalytic amounts of TEMPO and potassium bromide, and the mixture is cooled to maintain thermal control during the exothermic oxidation.
- Prepare a sodium hypochlorite solution by mixing with water, cooling to 0-5°C, and adjusting pH to 8.5-8.7 using hydrochloric acid and sodium bicarbonate.
- Dissolve Compound II in an organic solvent like dichloromethane, add TEMPO and potassium bromide, and cool the mixture to 0°C under nitrogen protection.
- Slowly add the prepared sodium hypochlorite solution while maintaining pH between 7-9, stir for 1 hour, quench with sodium thiosulfate, and separate phases to isolate Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound advantages that extend far beyond simple chemical yield improvements, directly impacting the bottom line through waste reduction and material efficiency. The most significant commercial benefit arises from the drastic reduction in impurity levels, which eliminates the need for extensive and costly chromatographic purification steps that are often required to meet pharmaceutical specifications when using older methods. By achieving a purity of over 99.7% directly from recrystallization, the process significantly reduces solvent consumption, labor hours, and processing time, leading to a streamlined manufacturing workflow that enhances overall throughput. Furthermore, the ability to consistently produce material with impurity levels below 0.05% mitigates the risk of batch rejection, ensuring a more predictable and reliable supply of critical intermediates for downstream API production schedules.
A key driver for cost reduction in this manufacturing process is the innovative strategy for handling the oxidation by-product, Compound I', which transforms a waste stream into a valuable resource. Instead of discarding the aqueous phase containing the carboxylic acid impurity, the patent discloses a recycling loop where Compound I' is separated and chemically reduced back to the starting material, Compound II, using reducing agents such as borane complexes. 
This closed-loop approach means that raw materials are utilized with maximum efficiency, as the carbon skeleton of the over-oxidized by-product is recovered and fed back into the production cycle. Qualitatively, this recycling capability represents a substantial decrease in the effective cost of raw materials per kilogram of final product, as the loss of expensive chiral precursors is minimized. Additionally, the robustness of the reaction conditions, which tolerate a wide temperature range from -25°C to 25°C and various common solvents, simplifies the engineering requirements for scale-up. This flexibility allows manufacturers to utilize existing infrastructure without needing specialized cryogenic equipment, thereby lowering capital expenditure barriers and facilitating a faster transition from pilot scale to commercial production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced oxidation technology for Rosuvastatin intermediate production. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential partners.
Q: What is the primary impurity challenge in synthesizing Rosuvastatin intermediates?
A: The primary challenge is the over-oxidation of the aldehyde group to a carboxylic acid, forming impurity I' (tert-butyl 2-((4R,6S)-6-carboxy-2,2-dimethyl-1,3-dioxan-4-yl)acetate), which historically reached levels of 10-25% and was difficult to remove.
Q: How does the new method control impurity levels?
A: The method strictly controls the reaction pH within a weakly alkaline range of 7 to 9 and utilizes a specific TEMPO/sodium hypochlorite system, reducing the content of impurity I' to below 0.05%.
Q: Can the oxidation by-product be recycled?
A: Yes, the separated impurity I' can be effectively reduced back to the starting material Compound II using reducing agents like borane/tetrahydrofuran, allowing for material recovery and improved overall process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and a commitment to quality excellence. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated pH control and recycling mechanisms described in this patent are executed with precision at every batch size. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every shipment of Rosuvastatin intermediate meets the exacting standards required for global pharmaceutical registration. We understand that consistency is the cornerstone of a secure supply chain, and our process validation protocols are designed to deliver the high-purity, low-impurity profiles necessary for your downstream synthesis.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that details how implementing this recycling-enabled oxidation route can optimize your specific budget and timeline. We encourage you to contact us today to request specific COA data from our recent batches and to discuss route feasibility assessments tailored to your volume requirements. By partnering with NINGBO INNO PHARMCHEM, you secure not just a chemical supplier, but a strategic ally dedicated to enhancing the efficiency and reliability of your cardiovascular drug manufacturing supply chain.
