Revolutionizing Alpha-Alkyl Ketone Production: A Green Iridium-Catalyzed Approach for Industrial Scale
Revolutionizing Alpha-Alkyl Ketone Production: A Green Iridium-Catalyzed Approach for Industrial Scale
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the urgent need for sustainable and economically viable manufacturing processes. A pivotal development in this arena is documented in Chinese Patent CN112047797A, which discloses a highly efficient method for preparing alpha-alkyl substituted ketone compounds. This technology leverages a sophisticated cyclic iridium complex catalyst to facilitate the coupling of primary and secondary alcohols directly in an aqueous medium. For R&D directors and procurement strategists in the pharmaceutical and agrochemical sectors, this patent represents more than just a new reaction; it offers a tangible pathway to reduce reliance on hazardous alkylating agents and volatile organic solvents. The ability to construct carbon-carbon bonds using simple alcohols as feedstocks aligns perfectly with the principles of green chemistry, addressing both regulatory pressures and cost containment goals simultaneously.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of alpha-alkyl substituted ketones has relied heavily on the alkylation of ketone enolates using alkyl halides. While chemically effective on a small scale, this classical approach presents severe drawbacks when evaluated through the lens of modern industrial manufacturing. The process typically necessitates the use of strong bases to generate reactive enolate intermediates, which often leads to competing side reactions such as self-condensation or over-alkylation, thereby compromising selectivity and yield. Furthermore, the stoichiometric generation of inorganic salt waste creates a substantial burden on downstream waste treatment facilities, driving up operational costs and environmental liabilities. The use of toxic alkyl halides also introduces significant safety hazards regarding worker exposure and requires rigorous containment protocols, making the process less attractive for large-scale production of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes a "borrowing hydrogen" strategy that fundamentally alters the reaction economy. By employing primary alcohols and secondary alcohols as the sole reactants, the process avoids the use of pre-functionalized halides entirely. The reaction proceeds through a catalytic cycle where the alcohol is transiently oxidized to an aldehyde or ketone, undergoes condensation, and is subsequently reduced back to the saturated ketone product. 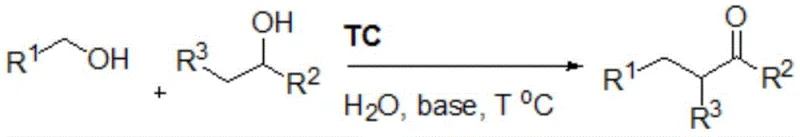 This transformation is mediated by a specialized cyclic iridium complex (denoted as TC) in the presence of a mild base. The most striking feature of this novel approach is the use of water as the reaction medium. This not only eliminates the cost and safety risks associated with organic solvents but also simplifies the workup procedure, as the product can often be extracted directly from the aqueous phase, streamlining the isolation process for supply chain teams.
This transformation is mediated by a specialized cyclic iridium complex (denoted as TC) in the presence of a mild base. The most striking feature of this novel approach is the use of water as the reaction medium. This not only eliminates the cost and safety risks associated with organic solvents but also simplifies the workup procedure, as the product can often be extracted directly from the aqueous phase, streamlining the isolation process for supply chain teams.
Mechanistic Insights into Iridium-Catalyzed Borrowing Hydrogen
To fully appreciate the technical robustness of this method, one must understand the underlying catalytic mechanism. The cyclic iridium complex acts as a multifunctional catalyst capable of reversible dehydrogenation and hydrogenation. Initially, the catalyst activates the secondary alcohol substrate, abstracting hydrogen to form a metal-hydride species and releasing the corresponding ketone intermediate. Simultaneously, the primary alcohol is oxidized to an aldehyde. These two carbonyl species then undergo an aldol-type condensation to form an alpha,beta-unsaturated ketone intermediate. Crucially, the metal-hydride species generated in the first step is not wasted; it is utilized to reduce the carbon-carbon double bond of the unsaturated intermediate, delivering the final saturated alpha-alkyl ketone product and regenerating the active catalyst. This closed-loop hydrogen transfer ensures high atom economy, as the only byproducts are water and trace amounts of hydrogen gas, avoiding the accumulation of stoichiometric waste.
From an impurity control perspective, this mechanism offers distinct advantages over traditional base-mediated alkylations. Because the reaction does not rely on highly reactive carbanions generated by strong bases, the risk of uncontrolled polymerization or degradation of sensitive functional groups is significantly minimized. The patent data indicates excellent tolerance for various substituents, including halogens, methoxy groups, and heterocycles like thiophene. This chemoselectivity is vital for R&D teams synthesizing complex drug candidates where protecting group strategies can add multiple steps and cost to the overall route. The high selectivity reported (up to 99:1) suggests that the catalyst effectively discriminates between the desired cross-coupling pathway and potential homocoupling side reactions, ensuring a cleaner crude profile that facilitates easier purification.
How to Synthesize Alpha-Alkyl Substituted Ketones Efficiently
Implementing this synthesis route requires careful attention to catalyst loading and reaction parameters to maximize the turnover number. The patent specifies a molar ratio of primary to secondary alcohol of approximately 1:1 to 1.1:1, ensuring complete conversion of the limiting reagent while minimizing excess raw material costs. The reaction is typically conducted at temperatures between 60°C and 100°C under an inert atmosphere to prevent catalyst oxidation.
- Combine primary alcohol and secondary alcohol substrates with a base (e.g., KOH) and the cyclic iridium catalyst (TC) in water.
- Heat the reaction mixture to 60-100°C under inert gas protection (Nitrogen/Argon) and stir for 10-24 hours.
- Cool to room temperature, extract with ethyl acetate, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iridium-catalyzed aqueous protocol offers compelling economic and logistical benefits. The shift away from alkyl halides and organic solvents directly impacts the cost of goods sold (COGS) by reducing raw material expenses and waste disposal fees. The simplicity of the workup procedure, involving basic extraction and chromatography, reduces the processing time and equipment occupancy, thereby enhancing overall plant throughput. Furthermore, the use of water as a solvent mitigates the regulatory complexities associated with VOC emissions, ensuring smoother compliance with increasingly stringent environmental regulations in major manufacturing hubs.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic alkylating agents, combined with the use of water as a free and safe solvent, drastically lowers the input costs for raw materials. Additionally, the high catalytic efficiency (S/C ratios exceeding 10,000) means that the precious metal catalyst loading can be kept extremely low, minimizing the impact of iridium prices on the final budget. The reduction in salt waste generation further decreases the operational expenditure related to effluent treatment and hazardous waste disposal, contributing to substantial long-term savings.
- Enhanced Supply Chain Reliability: The starting materials for this process—primary and secondary alcohols—are commodity chemicals that are widely available from multiple global suppliers, reducing the risk of supply chain bottlenecks. Unlike specialized alkyl halides which may have limited vendors or long lead times, alcohols offer a stable and secure sourcing strategy. The robustness of the reaction conditions also implies a lower risk of batch failure due to sensitivity to moisture or oxygen, ensuring consistent delivery schedules and reliable inventory management for downstream customers.
- Scalability and Environmental Compliance: The protocol has been demonstrated to work effectively on the gram scale with high yields, indicating a clear path toward kilogram and ton-scale production. The absence of volatile organic solvents simplifies the engineering controls required for scale-up, such as explosion-proof reactors and solvent recovery systems. This inherently safer design accelerates the technology transfer from the laboratory to the pilot plant and eventually to commercial manufacturing, while simultaneously meeting the sustainability goals of modern chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alpha-alkyl ketone synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation.
Q: What are the primary advantages of using water as a solvent in this ketone synthesis?
A: Using water eliminates the need for expensive and hazardous organic solvents, significantly reducing VOC emissions and fire risks while simplifying waste treatment protocols.
Q: How does the iridium catalyst improve reaction efficiency compared to traditional methods?
A: The cyclic iridium complex enables a 'borrowing hydrogen' mechanism with high turnover numbers (S/C > 10,000), allowing for milder conditions and higher selectivity without toxic alkylating agents.
Q: Is this method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the patent demonstrates gram-level synthesis with high yields (up to 95%) and broad substrate tolerance, indicating strong potential for scale-up to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Alkyl Substituted Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iridium-catalyzed borrowing hydrogen methodology for producing high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our facility is equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of alpha-alkyl ketones meets the exacting standards required for drug substance manufacturing. We are committed to leveraging this green chemistry innovation to deliver superior quality products while maintaining competitive pricing structures.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your target molecule. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your supply chain with reliable, high-purity alpha-alkyl substituted ketones produced via this state-of-the-art catalytic process.
