Optimizing Glupa-C Production: A Safe, Scalable Route for Diagnostic Reagents
The chemical synthesis landscape for diagnostic reagent substrates is undergoing a significant transformation driven by the urgent need for safer, more scalable, and higher-purity intermediates. Patent CN113045446A introduces a groundbreaking preparation method for Glupa-C (Gamma-glutamyl-3-carboxyl-4-nitroaniline monoammonium salt), a critical substrate used in the clinical determination of transpeptidase activity. This innovation addresses long-standing safety hazards and yield limitations inherent in previous synthetic routes by shifting the reaction environment from toxic alkaline conditions to a controlled acidic system. By replacing hazardous reagents like tri-n-butylamine with benign acidic solvents such as acetic acid, the patent outlines a pathway that not only mitigates explosion risks associated with high-temperature processing but also streamlines the purification process through advanced ion exchange techniques. For global supply chain leaders and R&D directors, this represents a pivotal shift towards sustainable and reliable pharmaceutical intermediate manufacturing, ensuring consistent quality for vital biochemical test kits.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
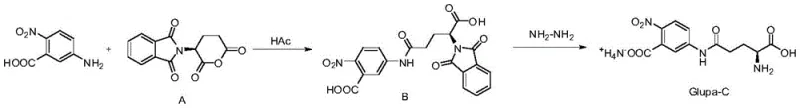
Historically, the synthesis of Glupa-C has been plagued by severe safety and efficiency bottlenecks that hindered its widespread industrial adoption. Early methodologies, such as those disclosed in U.S. Pat. No. 5,397,9447A, relied heavily on the use of tri-n-butylamine as a condensing agent. While chemically effective, tri-n-butylamine is a highly toxic substance that poses significant occupational health risks and complicates waste disposal protocols, making it increasingly undesirable for modern green chemistry initiatives. Furthermore, alternative routes described in Chinese patent CN1076688A attempted to drive the condensation reaction at excessively high temperatures ranging from 140°C to 142°C. This thermal approach introduced a critical safety vulnerability, as nitro-compounds are inherently unstable under such extreme thermal stress, creating a tangible risk of explosion during scale-up. Additionally, these legacy processes suffered from difficult downstream processing, where separating the desired Glupa-C from by-products like phthalhydrazide was notoriously inefficient, leading to inconsistent purity profiles and low overall yields that failed to meet the stringent demands of clinical diagnostics.
The Novel Approach
In stark contrast to these hazardous precedents, the novel approach detailed in CN113045446A leverages a mild acidic environment to drive the synthesis with exceptional control and safety. The core innovation lies in the use of acetic acid or formic acid as the reaction medium for the initial condensation step, which proceeds efficiently at a moderate temperature of 100°C. This eliminates the thermal instability issues of the nitro-group while avoiding the toxicity of tertiary amines. The subsequent hydrazinolysis step is conducted at ambient temperatures (0-50°C) in solvents like acetonitrile or methanol, further reducing energy consumption and operational risk. Crucially, the integration of ion exchange resin technology for the final ammonium salt formation allows for a highly selective purification mechanism. This ensures that the final product is isolated as a high-purity monoammonium salt, effectively resolving the separation difficulties that plagued earlier iterations. This robust methodology establishes a new standard for the commercial scale-up of complex pharmaceutical intermediates, offering a reproducible route that aligns perfectly with modern regulatory and safety expectations.
Mechanistic Insights into Acidic Condensation and Hydrazinolysis
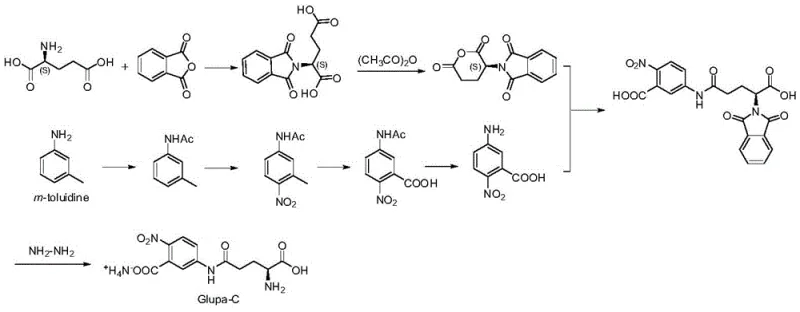
The mechanistic elegance of this synthesis lies in the precise manipulation of nucleophilic attacks under acidic catalysis. In the first step, the amino group of 5-amino-2-nitrobenzoic acid acts as a nucleophile, attacking the electrophilic carbonyl carbon of the phthaloyl-L-glutamic anhydride (Compound A). The presence of the acidic solvent, specifically acetic acid, plays a dual role: it solubilizes the reactants effectively and protonates the intermediate species to prevent unwanted side reactions or polymerization that might occur in basic media. The reaction is monitored via TLC or HPLC until the complete disappearance of the anhydride starting material, typically achieved within 2 hours at 100°C. This results in the formation of Intermediate B, a stable phthaloyl-protected amide, which serves as the crucial precursor for the final deprotection step. The careful control of the molar ratio, preferably around 0.9:1 of amine to anhydride, ensures minimal waste of the valuable chiral glutamic derivative while driving the equilibrium towards the desired product.
The second phase involves the strategic removal of the phthaloyl protecting group through hydrazinolysis. Hydrazine hydrate acts as a potent nucleophile, attacking the imide carbonyls of the phthaloyl ring in Intermediate B. This reaction cleaves the cyclic imide structure, releasing phthalhydrazide as a by-product and freeing the primary amine on the glutamic acid side chain. The reaction is conducted at room temperature in polar aprotic or protic solvents like acetonitrile, which facilitates the dissolution of the intermediate while maintaining the stability of the nitro-aniline moiety. Following the cleavage, the crude mixture undergoes a sophisticated purification sequence involving pH adjustment and extraction. The final conversion to the monoammonium salt is achieved using an ion exchange resin column. This step is mechanistically significant as it selectively binds the carboxylic acid groups and exchanges protons for ammonium ions, ensuring the final product exists in the specific salt form required for biochemical stability and solubility in diagnostic assays, thereby guaranteeing a purity profile exceeding 99%.
How to Synthesize Glupa-C Efficiently
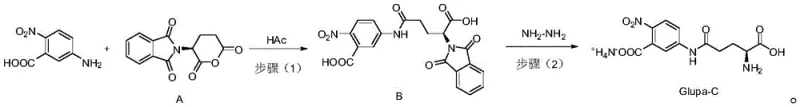
The synthesis of Glupa-C via this patented route offers a streamlined protocol that balances reaction kinetics with operational simplicity, making it ideal for both laboratory optimization and plant-scale production. The process begins with the precise weighing of 5-amino-2-nitrobenzoic acid and phthaloyl-L-glutamic anhydride, which are suspended in glacial acetic acid. The mixture is heated to reflux conditions (approximately 100°C) under vigorous stirring to ensure homogeneous reaction progress. Upon completion, confirmed by the absence of starting anhydride, the reaction mass is cooled to induce crystallization of Intermediate B, which is then filtered and washed. The second stage involves dissolving this intermediate in acetonitrile and treating it with a controlled addition of hydrazine hydrate solution. The reaction is allowed to proceed overnight at room temperature to ensure complete deprotection. The detailed standardized operating procedures, including specific workup parameters and crystallization conditions necessary to achieve GMP-grade quality, are outlined in the comprehensive guide below.
- Condense 5-amino-2-nitrobenzoic acid and phthaloyl-L-glutamic anhydride in acetic acid at 100°C to form Intermediate B.
- React Intermediate B with hydrazine hydrate in acetonitrile at room temperature to remove the phthaloyl protecting group.
- Purify the crude product using ion exchange resin to obtain the final monoammonium salt Glupa-C with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates directly into enhanced operational resilience and significant cost optimization opportunities. By eliminating the need for highly toxic reagents like tri-n-butylamine, manufacturers can drastically reduce the costs associated with hazardous waste disposal and specialized containment infrastructure. The shift to milder reaction conditions also lowers energy consumption, as the process no longer requires the extreme heating or cryogenic cooling often demanded by less efficient routes. Furthermore, the improved yield and simplified purification workflow mean that less raw material is required to produce the same amount of finished goods, effectively lowering the cost of goods sold (COGS). This efficiency gain is critical for maintaining competitive pricing in the volatile market of diagnostic reagents, allowing companies to secure better margins while passing savings on to healthcare providers.
- Cost Reduction in Manufacturing: The transition to an acidic solvent system eliminates the expensive and complex downstream processing required to remove toxic amines, leading to substantial cost savings in utility and waste management. The high selectivity of the ion exchange purification step reduces the loss of product during refining, maximizing the output from every batch of raw materials. Additionally, the use of commodity chemicals like acetic acid and hydrazine hydrate ensures that input costs remain stable and predictable, shielding the supply chain from the price volatility often seen with specialty catalysts.
- Enhanced Supply Chain Reliability: By removing the explosion risks associated with high-temperature nitro-compound processing, this method significantly improves plant safety and reduces the likelihood of unplanned shutdowns due to safety incidents. The robustness of the reaction conditions allows for consistent batch-to-batch reproducibility, ensuring that delivery schedules are met without delay. Sourcing of raw materials is also simplified, as the process relies on widely available bulk chemicals rather than scarce or regulated toxic substances, thereby strengthening the overall continuity of the supply chain for this critical pharmaceutical intermediate.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations and avoiding exotic equipment requirements. The reduced environmental footprint, characterized by lower toxicity and safer waste streams, ensures compliance with increasingly stringent global environmental regulations. This forward-looking approach future-proofs the manufacturing asset against regulatory tightening, allowing for seamless expansion of production capacity to meet growing global demand for transpeptidase test kits without the need for major capital reinvestment in safety mitigation systems.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial aspects of the Glupa-C synthesis process, derived directly from the innovative strategies presented in the patent literature. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this technology for integration into their production portfolios. Understanding these nuances is essential for making informed decisions regarding process validation and supplier qualification.
Q: Why is the acidic solvent method superior to traditional alkaline methods for Glupa-C?
A: The acidic method avoids the use of highly toxic tri-n-butylamine and eliminates the explosion risks associated with high-temperature condensation (140-142°C) found in prior art, ensuring safer industrial operations.
Q: How does the new process improve product purity?
A: By utilizing ion exchange resin chromatography during the final ammonium treatment step, the process effectively separates the product from by-products like phthalhydrazide, achieving purity levels up to 99%.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the process operates at moderate temperatures (100°C for condensation, room temperature for hydrazinolysis) and uses common solvents like acetic acid and acetonitrile, making it highly scalable and compliant with industrial safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glupa-C Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality substrates like Glupa-C play in the accuracy and reliability of clinical diagnostics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of materials that meet the most rigorous industry standards. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying every batch against the highest benchmarks. We are committed to delivering not just a chemical product, but a guaranteed solution that supports the integrity of your diagnostic assays and the health of your end-users.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable, high-quality supply of Glupa-C for your global operations.
