Advanced Synthetic Route for Tilbroquinol: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
Advanced Synthetic Route for Tilbroquinol: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
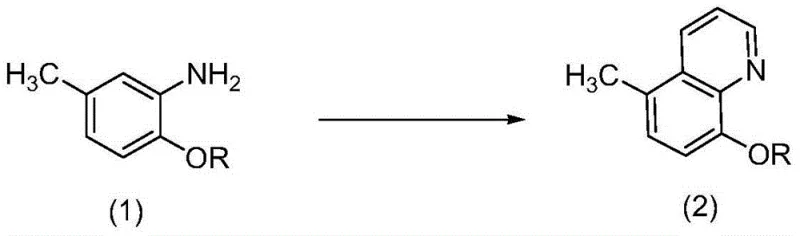
The pharmaceutical industry is constantly seeking robust manufacturing pathways that balance high purity with operational safety, a challenge vividly addressed in the recent technological disclosures surrounding the synthesis of Tilbroquinol. Patent CN103554019A introduces a transformative three-step methodology that fundamentally reimagines the production of 5-methyl-7-bromo-8-hydroxyquinoline, a critical active pharmaceutical ingredient used in treating parasitic and amoebic diseases. Unlike legacy processes that rely on hazardous alkylation agents, this innovative approach leverages a safer cyclization strategy followed by controlled hydrolysis and regioselective bromination. For R&D directors and procurement specialists evaluating reliable tilbroquinol supplier options, this patent represents a significant leap forward in process chemistry, offering a pathway that mitigates the severe safety risks associated with pyrophoric reagents while maintaining exceptional yield profiles. The strategic shift away from toxic methyl iodide not only aligns with modern green chemistry principles but also drastically simplifies the waste management infrastructure required for large-scale production facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-methyl-7-bromo-8-hydroxyquinoline has been plagued by significant safety and efficiency bottlenecks, primarily exemplified by the methods described in prior art such as patent WO02070486A1. These conventional routes typically necessitate the use of methyl iodide, a potent alkylating agent known for its high toxicity and carcinogenic potential, alongside sodium hydride, a dangerously reactive base that requires strictly anhydrous and oxygen-free conditions to prevent combustion. The reliance on n-butyllithium further exacerbates these risks, demanding specialized equipment and highly trained personnel to manage the exothermic nature of the reactions. From a supply chain perspective, these requirements translate into elevated operational costs, extended production cycles due to rigorous safety protocols, and inconsistent batch quality resulting from incomplete raw material conversion. Furthermore, the purification of the final product from these complex reaction mixtures is notoriously difficult, often leading to lower overall yields and higher impurity profiles that fail to meet the stringent standards required for clinical-grade active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology outlined in CN103554019A presents a streamlined and inherently safer alternative that bypasses the need for hazardous organometallic reagents entirely. This novel approach initiates with a robust acid-catalyzed cyclization of 2-alkoxy-5-methylaniline derivatives using readily available feedstocks like glycerol or acrolein, proceeding under mild aqueous acidic conditions that are far easier to control on an industrial scale. By substituting the dangerous alkylation step with a constructive ring-forming reaction, the process eliminates the generation of toxic halogenated waste streams and removes the threat of fire associated with sodium hydride handling. The subsequent steps involve a controlled hydrolysis or thiol-mediated demethylation followed by a precise bromination, both of which operate under moderate temperature ranges that preserve the structural integrity of the quinoline core. This strategic redesign not only enhances the safety profile for plant operators but also facilitates cost reduction in api manufacturing by utilizing commodity chemicals that are globally sourced and less subject to supply volatility than specialized organolithium reagents.
Mechanistic Insights into Acid-Catalyzed Cyclization and Bromination
The core of this synthetic innovation lies in the initial Skraup-like cyclization mechanism, where the aniline nitrogen attacks the activated carbon species generated from glycerol or acrolein under strong acidic conditions. In this step, the presence of a mass concentration greater than 30% of strong acids such as sulfuric acid or hydrochloric acid serves a dual purpose: it protonates the carbonyl or hydroxyl groups to enhance electrophilicity and simultaneously stabilizes the intermediate iminium ions formed during the ring closure. The reaction proceeds efficiently at temperatures between 80-160°C, driving the dehydration and aromatization necessary to form the stable quinoline scaffold found in compound (2). This mechanistic pathway is particularly advantageous because it tolerates a wide range of substituents and avoids the harsh basic conditions that often lead to polymerization or degradation of sensitive functional groups. The result is a high-yielding transformation that sets the stage for the subsequent functionalization steps, ensuring that the methyl group at the 5-position is correctly positioned for the final biological activity of the molecule.
Following the construction of the quinoline backbone, the process employs a highly selective bromination strategy to install the critical halogen atom at the 7-position. The mechanism relies on the activating effect of the 8-hydroxy group, which directs the electrophilic attack of the brominating agent—whether liquid bromine, N-bromosuccinimide (NBS), or dibromohydantoin—to the ortho position relative to the hydroxyl group. Conducted in organic solvents like acetonitrile or ethyl acetate at controlled temperatures ranging from -10 to 100°C, this step achieves remarkable regioselectivity, minimizing the formation of undesired poly-brominated byproducts. The ability to achieve HPLC purity levels of ≥99.8% directly from the reaction mixture underscores the precision of this mechanistic control. For quality assurance teams, this implies a significantly reduced burden on downstream purification units, as the crude product already meets rigorous specification limits, thereby accelerating the timeline from reactor to release testing and ensuring a consistent supply of high-purity pharmaceutical intermediates.
How to Synthesize 5-methyl-7-bromo-8-hydroxyquinoline Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and safety, beginning with the preparation of the quinoline core through acid-catalyzed condensation. Operators must maintain strict control over the acid concentration and temperature profile during the initial cyclization to ensure complete conversion of the aniline starting material while preventing thermal degradation of the product. Following isolation, the demethylation step can be tuned via either acidic hydrolysis or thiol treatment depending on the specific substrate sensitivity, offering flexibility in process design. The final bromination is conducted under mild conditions to preserve the integrity of the heterocyclic ring, with workup procedures designed to recover solvents and minimize waste. The detailed standardized synthesis steps see the guide below for specific molar ratios and processing times.
- React 2-alkoxy-5-methylaniline with glycerol or acrolein in strong acidic aqueous solution at 80-160°C to form the quinoline core.
- Perform acidic hydrolysis or thiol-mediated reaction on the methoxy-quinoline intermediate at 30-150°C to generate the hydroxy-quinoline precursor.
- Conduct regioselective bromination of the hydroxy-quinoline using liquid bromine or NBS in organic solvent at -10 to 100°C to yield Tilbroquinol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers profound strategic benefits that extend far beyond simple chemical transformation. By eliminating the dependency on methyl iodide and sodium hydride, manufacturers can drastically reduce the costs associated with hazardous material storage, specialized containment infrastructure, and expensive waste disposal services. The shift towards using commodity reagents like glycerol and mineral acids creates a more resilient supply chain, as these materials are produced in massive volumes globally and are less susceptible to the geopolitical or logistical disruptions that often plague niche fine chemical suppliers. This stability translates directly into enhanced supply chain reliability, ensuring that production schedules remain uninterrupted even during periods of market volatility. Furthermore, the simplified operational workflow reduces the need for highly specialized labor, allowing facilities to optimize their workforce allocation and focus on scaling output rather than managing complex safety protocols.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous reagents such as methyl iodide and n-butyllithium leads to substantial cost savings in raw material procurement and handling. Without the need for strict anhydrous conditions or inert atmosphere gloveboxes, the capital expenditure for reactor setup is significantly lowered, and energy consumption is reduced due to milder reaction temperatures. The high conversion rates and minimal byproduct formation mean that less material is lost to waste streams, effectively increasing the overall mass balance efficiency of the plant. Additionally, the avoidance of toxic reagents simplifies regulatory compliance and reduces the financial liability associated with environmental permits and safety audits, creating a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials like 2-methoxy-5-methylaniline and glycerol ensures that production is not bottlenecked by the scarcity of specialized organometallic reagents. This accessibility allows for larger batch sizes and more frequent production runs, effectively reducing lead time for high-purity pharmaceutical intermediates and enabling faster response to market demand fluctuations. The robustness of the aqueous acid steps also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply output. Consequently, partners can rely on a consistent flow of material without the risk of sudden stoppages caused by the unavailability of critical, hard-to-source catalysts or bases.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, as the exothermic profiles are manageable and the reaction media are compatible with standard stainless steel equipment. The reduction in toxic waste generation aligns with increasingly strict global environmental regulations, minimizing the ecological footprint of the manufacturing site. Waste streams are easier to treat due to the absence of heavy metals and persistent organic pollutants, facilitating smoother interactions with local regulatory bodies. This environmental stewardship not only protects the corporate reputation but also future-proofs the manufacturing asset against tightening emission standards, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and comparative analysis with prior art. These insights are intended to clarify the operational benefits and quality assurances provided by this novel route for stakeholders evaluating potential partnerships. Understanding these nuances is crucial for making informed decisions about process adoption and supply agreements.
Q: How does this new synthesis method improve safety compared to traditional routes?
A: The novel process eliminates the use of highly toxic methyl iodide and pyrophoric sodium hydride required in prior art (WO02070486A1), significantly reducing operational hazards and environmental pollution risks.
Q: What represents the key advantage for supply chain stability in this method?
A: By utilizing common industrial reagents like glycerol, acrolein, and mineral acids instead of specialized organolithium reagents, the method ensures consistent raw material availability and reduces lead time for high-purity pharmaceutical intermediates.
Q: Can this process achieve the purity required for clinical applications?
A: Yes, the optimized bromination step yields products with HPLC purity ≥99.8%, meeting stringent quality specifications without requiring complex purification workflows that often delay commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tilbroquinol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthesis routes is critical for the long-term success of pharmaceutical supply chains. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN103554019A are fully realized in practical, large-volume manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Tilbroquinol meets the exacting standards required for global regulatory submission. We combine deep technical expertise with a commitment to operational excellence, providing a secure foundation for your drug development and commercialization efforts.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can specifically benefit your project timelines and budgetary goals. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume requirements. We encourage potential partners to contact us immediately to obtain specific COA data and route feasibility assessments, allowing you to validate the superior quality and reliability of our manufacturing capabilities firsthand.
