Scalable Synthesis of 4-(2-Bromoethyl)indolin-2-one for High-Efficiency Ropinirole Production
Introduction to Advanced Ropinirole Intermediate Synthesis
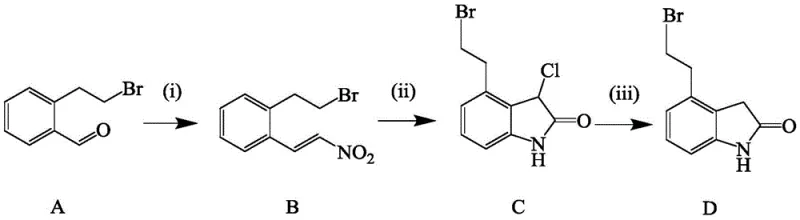
The pharmaceutical landscape for Parkinson's disease treatment relies heavily on the efficient production of dopamine agonists like Ropinirole hydrochloride. Central to this supply chain is the critical intermediate, 4-(2-bromoethyl)-1,3-dihydro-2H-indol-2-one. Recent intellectual property developments, specifically patent CN112745256B, have introduced a transformative preparation method that addresses long-standing bottlenecks in yield and purity. This technical disclosure outlines a robust synthetic pathway starting from o-bromoethyl benzaldehyde, utilizing a series of optimized reactions that bypass the limitations of legacy manufacturing protocols. For R&D directors and procurement strategists, understanding this shift is vital, as it represents a move away from expensive precious metal catalysts toward more sustainable, cost-effective chemical engineering solutions that ensure supply continuity for high-volume API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key indolinone intermediate has been plagued by inefficiencies that drive up costs and complicate regulatory compliance. In traditional routes, the nitroaldol condensation step often employs sodium formate or sodium methoxide in methanol, resulting in poor conversion rates that rarely exceed 50% even after prolonged reaction times of up to 72 hours. Furthermore, the subsequent reduction steps typically rely on Palladium on Carbon (Pd/C) or Raney Nickel. The use of Pd/C is particularly problematic; it is not only a high-cost input, accounting for a significant portion of the total route expense, but it also introduces severe risks of heavy metal contamination. Removing trace palladium to meet pharmaceutical standards requires additional purification stages, and the catalyst itself is often single-use, generating substantial solid waste. Additionally, the solvent systems in these older methods, often contaminated with water post-reaction, are difficult to recycle, leading to increased wastewater treatment burdens and environmental liabilities.
The Novel Approach
The methodology described in patent CN112745256B offers a decisive break from these inefficiencies by re-engineering the core reaction steps. As illustrated in the reaction scheme, the new process initiates with a highly efficient condensation where nitromethane serves a dual role as both reactant and solvent, catalyzed by inexpensive ammonium salts. This is followed by a cyclization step using ferric trichloride and acetyl chloride. Most critically, the final reduction replaces the problematic Pd/C hydrogenation with a chemical reduction system utilizing sodium iodide or potassium iodide alongside phosphorous acid. This substitution eliminates the need for precious metals entirely, thereby removing the risk of heavy metal residues in the final product. The streamlined nature of this approach allows for higher molar yields, reaching up to 95% in the initial step and over 86% in the final reduction, while enabling the recovery and reuse of solvents like ethanol and ethyl acetate, significantly lowering the environmental footprint.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Iodide Reduction
The success of this novel synthetic route hinges on the precise mechanistic control exerted during the cyclization and reduction phases. In the second step, ferric trichloride (FeCl3) acts as a potent Lewis acid catalyst in conjunction with acetyl chloride. This combination facilitates the intramolecular cyclization of the nitro-styrene intermediate to form the chloro-indolinone structure. The use of FeCl3 is advantageous not only for its catalytic efficiency but also for its low cost and ease of removal compared to transition metal complexes. The reaction conditions are carefully controlled, typically maintained around 5°C during addition to manage exothermicity, ensuring high selectivity for the desired ring-closed product while minimizing polymerization or side reactions that could compromise the purity profile required for downstream API synthesis.
In the final transformation, the mechanism shifts to a reductive dechlorination driven by the iodide/phosphorous acid system. Unlike catalytic hydrogenation which requires high-pressure equipment and poses safety risks with pyrophoric catalysts like Raney Nickel, this chemical reduction proceeds under atmospheric pressure in refluxing ethanol. The iodide species likely acts as a nucleophilic catalyst or a reducing mediator, facilitating the removal of the chlorine atom at the 3-position while simultaneously reducing the adjacent functionality to establish the saturated indolinone core. This mechanism is exceptionally clean, producing inorganic byproducts that are easily separated during the aqueous workup and recrystallization phases. The result is a product with a superior impurity profile, free from the vinyl byproducts often associated with over-reduction or incomplete hydrogenation in palladium-catalyzed systems.
How to Synthesize 4-(2-Bromoethyl)indolin-2-one Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to maximize yield and purity. The process is designed for scalability, moving seamlessly from laboratory validation to commercial production. The initial condensation sets the tone for the entire sequence, requiring precise temperature control between 90°C and 100°C to drive the equilibrium forward while allowing for the distillation and recovery of excess nitromethane.
- Condense o-bromoethyl benzaldehyde with nitromethane using an ammonium salt catalyst at 90-100°C to form the nitro-styrene intermediate.
- Perform cyclization using ferric trichloride and acetyl chloride in dichloromethane to generate the chloro-indolinone precursor.
- Execute the final dechlorination and reduction using sodium iodide or potassium iodide with phosphorous acid in ethanol to yield the target indolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages beyond simple chemical yield. The most immediate impact is seen in the raw material cost structure. By eliminating the dependency on Palladium on Carbon, which historically represents a volatile and expensive cost center, manufacturers can stabilize their bill of materials. The replacement reagents—sodium iodide, potassium iodide, and phosphorous acid—are commodity chemicals with stable pricing and abundant global availability. This shift mitigates the supply risk associated with precious metals, which are subject to geopolitical fluctuations and mining constraints. Furthermore, the ability to recycle solvents such as nitromethane and ethanol reduces the ongoing operational expenditure related to solvent purchase and disposal, contributing to a leaner manufacturing cost base.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling when analyzing the total cost of ownership. Traditional routes incur hidden costs related to the disposal of spent palladium catalysts and the extensive processing required to scavenge heavy metals from the final API. By switching to a non-metallic reduction system, these downstream purification costs are effectively eliminated. Additionally, the high conversion rates in the initial condensation step mean less raw material is wasted, and the ability to distill and reuse nitromethane creates a closed-loop system that minimizes waste treatment fees. This holistic approach to cost reduction ensures that the manufacturing of this high-purity pharmaceutical intermediate remains economically viable even under tight margin pressures.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for API intermediates, and this new route strengthens resilience by diversifying the chemical inputs. The reliance on bulk industrial chemicals like ferric chloride and ammonium salts reduces the risk of bottlenecks that can occur with specialized catalysts. Moreover, the simplified workup procedures, which avoid complex filtration of fine catalyst powders that can clog industrial filters, lead to faster batch cycle times. This operational efficiency allows for more flexible production scheduling and quicker response to market demand surges for Ropinirole, ensuring that downstream drug manufacturers receive their materials on time without interruption.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently safer and more scalable. The avoidance of high-pressure hydrogenation equipment reduces the facility's safety risk profile. The reduction in wastewater generation, achieved through solvent recycling and the absence of aqueous quenching steps that contaminate organic solvents, aligns with increasingly stringent global environmental regulations. This compliance advantage prevents potential production shutdowns due to environmental violations and positions the manufacturer as a sustainable partner for multinational pharmaceutical companies who prioritize green chemistry in their supplier audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy systems in terms of purity, yield, and operational safety.
Q: How does this new method eliminate heavy metal contamination risks?
A: Unlike conventional routes relying on Palladium on Carbon (Pd/C) which pose significant heavy metal residue risks requiring complex purification, this patented method utilizes sodium iodide or potassium iodide combined with phosphorous acid. This transition metal-free reduction system ensures the final API intermediate meets stringent heavy metal specifications without expensive scavenging steps.
Q: What are the yield improvements compared to traditional nitroaldol condensation?
A: Traditional methods using sodium formate in methanol often struggle with conversion rates below 50% even after 72 hours. The disclosed protocol employs nitromethane as both solvent and reagent with ammonium salt catalysis, achieving molar yields of 90% to 95% within just 6 to 8 hours, drastically improving throughput.
Q: Is the solvent system in this process environmentally sustainable?
A: Yes, the process is designed for green chemistry compliance. Excess nitromethane in step one is distilled and recycled. In the final reduction step, solvents like ethanol and ethyl acetate are used, which are easily recoverable, unlike the methanol/water mixtures in older routes that create difficult-to-treat wastewater streams.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(2-Bromoethyl)indolin-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN112745256B are fully realized in practice. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, particularly for critical parameters like heavy metal content and residual solvents, guaranteeing that every batch of 4-(2-bromoethyl)-1,3-dihydro-2H-indol-2-one meets the exacting standards required for Parkinson's disease medication.
We invite pharmaceutical partners to leverage our technical proficiency to optimize their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to validate the superior quality and economic advantages of our next-generation manufacturing process for this vital pharmaceutical intermediate.
