Advanced Direct Tert-Butylation Technology for High-Purity p-tert-butyl o-nitrothiophenol Manufacturing
The chemical landscape for specialized sulfur-containing intermediates is undergoing a significant transformation driven by the need for more efficient and cost-effective synthetic routes. Patent CN101723864A, published in June 2010, introduces a groundbreaking methodology for the preparation of p-tert-butyl o-nitrothiophenol, a critical building block in the synthesis of advanced pharmaceutical agents and electrochromic display materials. This technology represents a paradigm shift from traditional multi-step conversions to a direct, high-yield tert-butylation strategy. By leveraging readily available starting materials such as o-nitrothiophenol or 2,2'-dinitrodiphenyl disulfide, this invention addresses long-standing challenges related to raw material scarcity and process complexity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic elegance and commercial viability of this patent is essential for securing a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of p-tert-butyl o-nitrothiophenol has been plagued by inefficient pathways that rely on the functional group interconversion of phenolic precursors. The conventional route typically begins with p-tert-butyl o-nitrophenol, necessitating a series of complex chemical transformations to convert the phenolic hydroxyl group into the desired mercapto (thiol) functionality. As illustrated in the reaction scheme below, this often involves the formation of intermediate thiocarbamates or similar derivatives, which require harsh conditions and multiple purification steps. 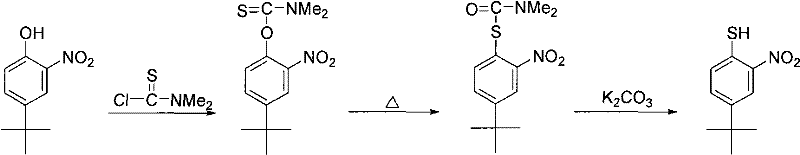 These legacy methods suffer from several critical drawbacks, including the high cost and difficult acquisition of the specific tert-butylated phenol starting material. Furthermore, the multi-step nature of the conversion leads to cumulative yield losses, making the final product economically unviable for large-scale industrial applications. The difficulty in grasping precise reaction conditions for these delicate functional group swaps further exacerbates the risk of batch-to-batch variability, posing a significant threat to supply chain stability for downstream manufacturers of sensitive electronic materials.
These legacy methods suffer from several critical drawbacks, including the high cost and difficult acquisition of the specific tert-butylated phenol starting material. Furthermore, the multi-step nature of the conversion leads to cumulative yield losses, making the final product economically unviable for large-scale industrial applications. The difficulty in grasping precise reaction conditions for these delicate functional group swaps further exacerbates the risk of batch-to-batch variability, posing a significant threat to supply chain stability for downstream manufacturers of sensitive electronic materials.
The Novel Approach
In stark contrast to the convoluted legacy pathways, the novel approach disclosed in the patent streamlines the synthesis through a direct electrophilic aromatic substitution on the thiol ring itself. By utilizing o-nitrothiophenol or its disulfide dimer as the foundational feedstock, the process introduces the tert-butyl group directly onto the aromatic ring in a single, highly efficient step. 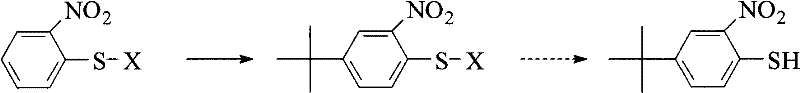 This strategic inversion of the synthetic logic eliminates the need for expensive pre-functionalized starting materials, relying instead on commodity chemicals that are abundant in the global market. The reaction proceeds smoothly under the influence of various acid catalysts, achieving high regioselectivity for the para-position relative to the thiol group. This not only drastically simplifies the operational workflow but also significantly enhances the overall atom economy of the process. For procurement managers focused on cost reduction in pharmaceutical intermediates manufacturing, this direct route offers a compelling value proposition by reducing both material costs and processing time, thereby enabling a more responsive and resilient supply network.
This strategic inversion of the synthetic logic eliminates the need for expensive pre-functionalized starting materials, relying instead on commodity chemicals that are abundant in the global market. The reaction proceeds smoothly under the influence of various acid catalysts, achieving high regioselectivity for the para-position relative to the thiol group. This not only drastically simplifies the operational workflow but also significantly enhances the overall atom economy of the process. For procurement managers focused on cost reduction in pharmaceutical intermediates manufacturing, this direct route offers a compelling value proposition by reducing both material costs and processing time, thereby enabling a more responsive and resilient supply network.
Mechanistic Insights into Acid-Catalyzed Friedel-Crafts Alkylation
The core of this innovative synthesis lies in the mechanism of the acid-catalyzed Friedel-Crafts alkylation, where the electron-rich aromatic ring of o-nitrothiophenol acts as the nucleophile. The presence of the thiol (-SH) group activates the ring towards electrophilic attack, particularly at the para-position, despite the deactivating influence of the ortho-nitro group. The catalyst, whether a Lewis acid like boron trifluoride or aluminum chloride, or a Brønsted acid like phosphoric acid, serves to generate the reactive tert-butyl carbocation from sources such as isobutylene or t-butyl chloride. This electrophile then attacks the aromatic ring, forming a sigma complex that subsequently loses a proton to restore aromaticity, yielding the desired 4-tert-butyl-2-nitrothiophenol. The versatility of the catalyst system is a key feature, allowing manufacturers to select the most appropriate reagent based on availability and waste treatment capabilities. For instance, the use of strong acidic ion exchange resins offers a heterogeneous catalysis pathway that can simplify product isolation and catalyst recycling, aligning with modern green chemistry principles.
Impurity control is another critical aspect where this mechanism excels, particularly regarding the suppression of poly-alkylation and disulfide formation. The reaction conditions are carefully tuned, with temperatures ranging from -30°C to 160°C, to balance reaction rate with selectivity. By controlling the stoichiometry of the tert-butyl source and the acidity of the medium, the process minimizes the formation of di-tert-butylated byproducts or unwanted side reactions at the sulfur atom. In cases where the disulfide dimer is used as the starting material, the subsequent alkaline lysis step provides an additional purification opportunity, as the cleavage of the disulfide bond to the thiol salt allows for the removal of non-acidic organic impurities before final acidification. This robust control over the reaction profile ensures that the final product meets the stringent purity specifications required for high-value applications in electrochromism and extraction agent synthesis, providing R&D teams with a reliable source of high-purity intermediates.
How to Synthesize p-tert-butyl o-nitrothiophenol Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of charging, reacting, and isolating, designed for seamless integration into existing chemical infrastructure. The process begins with the dissolution of the thiol starting material in a common organic solvent such as toluene or methylene dichloride, followed by the controlled addition of the catalyst and tert-butylating agent. Detailed standard operating procedures regarding specific molar ratios, addition rates, and safety protocols for handling volatile alkylating agents are critical for successful execution. This structured approach ensures that operators can consistently reproduce the high yields reported in the patent examples, facilitating a smooth transition from laboratory scale to commercial production.
- Charge o-nitrothiophenol or 2,2'-dinitrodiphenyl disulfide into a reactor with a suitable solvent such as toluene or methylene dichloride.
- Add a catalytic amount of Lewis or Brønsted acid catalyst (e.g., BF3, phosphoric acid, or strong acidic ion exchange resin) and introduce the tert-butyl source (isobutylene or t-butyl chloride).
- Maintain reaction temperature between 0°C and 140°C, followed by aqueous workup, drying, and purification via distillation or crystallization to isolate the target thiol.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement executives, the adoption of this patented technology translates into tangible strategic benefits that extend far beyond simple unit price reductions. The shift from scarce, specialized phenolic precursors to abundant thiol-based feedstocks fundamentally alters the cost structure of the manufacturing process, insulating buyers from the volatility associated with niche raw material markets. By shortening the synthetic route and eliminating complex functional group interconversions, the process inherently reduces energy consumption and waste generation, contributing to a more sustainable and compliant operation. These efficiencies allow suppliers to offer more competitive pricing structures while maintaining healthy margins, creating a win-win scenario for long-term partnerships in the fine chemical sector.
- Cost Reduction in Manufacturing: The elimination of expensive pre-tert-butylated starting materials and the reduction in synthetic steps lead to a substantial decrease in overall production costs. By avoiding the low-yielding conversion of phenols to thiols, the process maximizes the utilization of raw materials, effectively lowering the cost per kilogram of the final active intermediate. Furthermore, the ability to use commodity catalysts and solvents reduces the dependency on specialized reagents, simplifying inventory management and reducing procurement overheads significantly.
- Enhanced Supply Chain Reliability: Sourcing o-nitrothiophenol and isobutylene is far more reliable than procuring specialized nitrophenol derivatives, as these are produced on a massive scale by the global petrochemical industry. This abundance ensures that production schedules are not disrupted by raw material shortages, guaranteeing consistent delivery timelines for downstream customers. The robustness of the reaction conditions also means that manufacturing can proceed with minimal sensitivity to minor fluctuations in utility supplies or environmental conditions, further stabilizing the supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with various catalyst systems including recoverable ion exchange resins. This flexibility allows for the design of continuous flow processes or large batch reactors that meet rigorous environmental standards. The simplified workup procedure, often involving simple phase separation and distillation, reduces the volume of aqueous waste and solvent emissions, facilitating easier compliance with increasingly strict environmental regulations governing chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and industrial best practices. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own value chains. These insights provide a foundational understanding of the process capabilities and limitations, aiding in informed decision-making for project planning and resource allocation.
Q: What are the primary advantages of the direct tert-butylation method over conventional phenol conversion routes?
A: The direct tert-butylation method utilizes readily available and inexpensive o-nitrothiophenol as a starting material, bypassing the complex and low-yielding functional group transformations required when starting from p-tert-butyl o-nitrophenol. This results in a significantly shorter synthetic route and improved overall process economics.
Q: Which catalysts are most effective for this specific alkylation reaction?
A: The patent discloses a wide range of effective catalysts including phosphoric acid, boron trifluoride, aluminum chloride, and zinc chloride. Notably, strong acidic ion exchange resins are also viable, offering potential advantages in catalyst recovery and waste reduction for large-scale operations.
Q: Can this process be scaled for industrial production of electronic materials?
A: Yes, the process is explicitly designed for scalability. The use of common solvents like toluene and methylene dichloride, combined with robust reaction conditions ranging from 0°C to 140°C, facilitates easy adaptation to commercial scale-up for applications in electrochromic displays and polymerization initiators.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-tert-butyl o-nitrothiophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation pharmaceuticals and electronic materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of p-tert-butyl o-nitrothiophenol meets the exacting standards required for sensitive applications like electrochromic displays and polymerization initiators. Our commitment to quality assurance ensures that your downstream processes remain uninterrupted and productive.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized manufacturing processes can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for complex fine chemical intermediates.
