Advanced Atmospheric CO2 Fixation for High-Purity Carbonyl Heterocycles Manufacturing
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, safer, and more economically viable methodologies, a transition vividly exemplified by the technological breakthroughs detailed in patent CN110878055B. This pivotal intellectual property introduces a robust and universally applicable method for preparing carbonyl heterocyclic compounds, specifically targeting the critical structural motifs of benzimidazolones, benzoxazolones, and benzothiazolones which serve as indispensable scaffolds in modern drug discovery and agrochemical development. Unlike conventional approaches that rely on hazardous reagents, this innovation leverages atmospheric carbon dioxide (CO2) as a renewable C1 building block, activated efficiently through a synergistic system of Lewis bases and hydrosilanes. For R&D directors and procurement strategists alike, this represents a monumental opportunity to decouple high-value intermediate production from volatile supply chains associated with toxic gases and precious metals. By operating under mild conditions at 100°C in diglyme solvent, the process not only ensures exceptional safety profiles but also delivers impressive isolated yields ranging from 70% to 95% across a diverse substrate scope, establishing a new benchmark for sustainable manufacturing in the pharmaceutical intermediate sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of carbonyl heterocycles has been plagued by severe safety hazards and operational complexities inherent to traditional carbonylation reagents. The most prevalent legacy methods depend heavily on the use of phosgene or carbon monoxide (CO), both of which are classified as extremely toxic and pose catastrophic risks in the event of leakage, necessitating specialized containment infrastructure and rigorous safety protocols that drive up capital expenditure. Furthermore, alternative green attempts utilizing CO2 have often been thwarted by the thermodynamic stability and kinetic inertness of the CO2 molecule, typically requiring high-pressure reactors (often exceeding several megapascals) and expensive transition metal catalysts to force the reaction forward. These high-pressure systems introduce significant engineering challenges regarding scalability and maintenance, while the reliance on noble metals creates vulnerability to price fluctuations and supply shortages. Additionally, many existing routes involve the generation and isolation of unstable isocyanate intermediates, which require complex purification steps to remove trace toxic residues, thereby increasing waste generation and prolonging production cycles significantly.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in CN110878055B offers a streamlined, metal-free pathway that operates at atmospheric pressure, fundamentally altering the risk-reward profile of carbonyl heterocycle manufacturing. By employing a dual-promoter system consisting of a Lewis base (such as CsF or KOtBu) and a hydrosilane (specifically polymethylhydrosiloxane or PMHS), the reaction effectively activates the inert CO2 molecule under mild thermal conditions without the need for high-pressure equipment. This approach not only eliminates the logistical nightmare of transporting and storing toxic phosgene but also bypasses the requirement for costly metal catalysts, resulting in a cleaner reaction profile with fewer heavy metal impurities. The versatility of this system is demonstrated by its compatibility with a wide array of ortho-substituted anilines, accommodating various functional groups including halogens, esters, and alkyl chains without compromising efficiency. As illustrated in the structural framework below, the transformation proceeds smoothly to generate the target heterocyclic core with high fidelity.
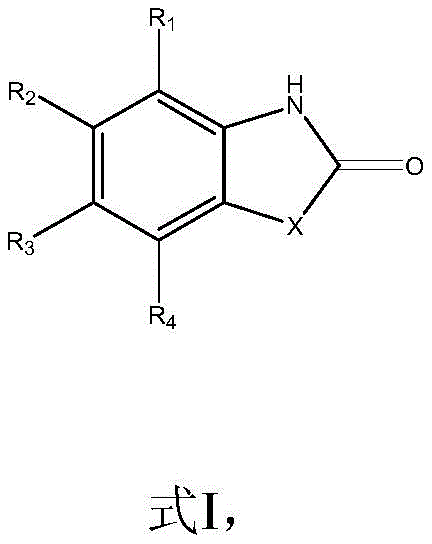
Mechanistic Insights into Lewis Base and Hydrosilane Promoted Cyclization
The mechanistic elegance of this synthesis lies in the cooperative activation of CO2 by the Lewis base and the subsequent reduction by the hydrosilane, which together overcome the high energy barrier of CO2 insertion. The Lewis base acts as a nucleophilic activator or a coordination site modifier, facilitating the attack of the amine nitrogen on the electrophilic carbon of the CO2 molecule to form a carbamate-like intermediate in situ. Crucially, the hydrosilane serves as a potent oxygen scavenger and reducing agent, driving the equilibrium forward by removing oxygen atoms from the intermediate species, effectively dehydrating the system to form the stable carbonyl bond within the heterocyclic ring. This mechanism avoids the formation of free isocyanate species that typically require isolation in other pathways, thereby minimizing side reactions such as polymerization or hydrolysis that often plague batch processes. The choice of PMHS as the hydrosilane source is particularly strategic for industrial applications, as it is an inexpensive byproduct of the silicone industry, offering a cost-effective alternative to discrete silanes like Et3SiH while maintaining high reactivity.
From an impurity control perspective, this mechanism offers distinct advantages for pharmaceutical grade manufacturing by inherently limiting the generation of difficult-to-remove byproducts. Since the reaction does not utilize transition metals, the final product is free from residual palladium, nickel, or copper contaminants that often require extensive scavenging treatments to meet strict ICH Q3D guidelines. The reaction conditions are sufficiently mild to prevent the decomposition of sensitive functional groups on the aromatic ring, ensuring that substituents such as esters, nitriles, and halogens remain intact throughout the cyclization process. The substrate scope is remarkably broad, accepting ortho-substituted anilines where the substituent X can be NH, S, or O, allowing for the seamless production of benzimidazolones, benzothiazolones, and benzoxazolones respectively. The structural diversity achievable with this method is highlighted by the generic precursor structure shown below, which accommodates various R groups to tailor the electronic and steric properties of the final API intermediate.
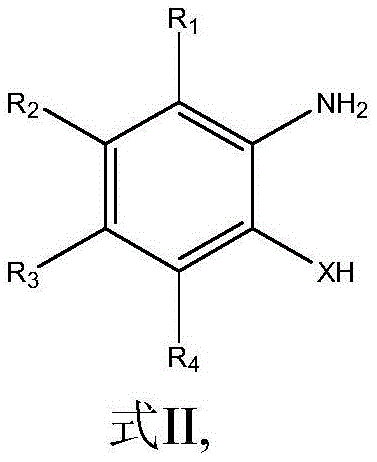
How to Synthesize Carbonyl Heterocycles Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, enabling chemists to achieve high conversions with minimal procedural overhead. The process begins by charging a reaction vessel with the ortho-substituted aniline substrate, a catalytic amount of Lewis base, and a polar aprotic solvent such as diglyme, followed by the introduction of CO2 gas at ambient pressure. Upon the addition of the hydrosilane promoter and heating to 100°C, the cyclization proceeds over a period of approximately 20 hours to reach completion. The detailed standardized synthetic steps, including precise molar ratios and workup parameters, are outlined in the technical guide below.
- Prepare the reaction system by mixing ortho-substituted aniline compounds with a Lewis base promoter such as CsF in a suitable organic solvent like diglyme.
- Introduce atmospheric pressure CO2 gas into the reaction vessel and add polymethylhydrosiloxane (PMHS) as the hydrosilane reducing agent.
- Heat the mixture to 100°C for approximately 20 hours, then isolate the pure carbonyl heterocyclic product via simple suction filtration and washing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this CO2 fixation technology translates directly into tangible economic benefits and enhanced operational resilience. The elimination of phosgene and high-pressure CO2 infrastructure drastically reduces the capital investment required for plant setup and lowers the ongoing costs associated with safety compliance and hazardous material handling. By utilizing atmospheric CO2, a readily available and virtually limitless feedstock, manufacturers can insulate themselves from the price volatility associated with specialized carbonylation reagents, ensuring a stable and predictable cost base for long-term production contracts. Furthermore, the use of PMHS, an industrial waste product, as a key reagent transforms a disposal liability into a value-added input, contributing to a circular economy model that aligns with modern corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the complete avoidance of precious metal catalysts which represent a significant portion of raw material costs in traditional cross-coupling or carbonylation reactions. Without the need for expensive ligands or metal salts, the bill of materials is significantly streamlined, and the downstream processing costs are reduced since there is no requirement for specialized metal scavenging resins or complex purification columns. The simple workup procedure, which often involves merely cooling the reaction mixture and filtering the precipitated product, minimizes solvent consumption and energy usage compared to multi-step extraction and chromatography protocols. This efficiency gain allows for a substantial reduction in the overall cost of goods sold (COGS), making the final intermediates more competitive in the global market.
- Enhanced Supply Chain Reliability: Relying on atmospheric CO2 and commodity chemicals like Lewis bases and siloxanes creates a robust supply chain that is less susceptible to geopolitical disruptions or single-source bottlenecks. Unlike specialized reagents that may have limited suppliers and long lead times, the inputs for this reaction are widely available from multiple chemical distributors globally, ensuring continuity of supply even during market fluctuations. The mild reaction conditions also reduce the wear and tear on reactor vessels and ancillary equipment, leading to lower maintenance downtime and higher asset utilization rates. This reliability is crucial for meeting tight delivery schedules for downstream API manufacturers who depend on consistent intermediate availability to maintain their own production timelines.
- Scalability and Environmental Compliance: The transition from bench scale to commercial production is facilitated by the absence of high-pressure requirements, allowing the reaction to be run in standard glass-lined or stainless steel reactors without the need for autoclaves rated for extreme pressures. This simplifies the scale-up process and reduces the regulatory burden associated with operating high-pressure systems, accelerating the time to market for new drug candidates. Environmentally, the process generates minimal waste, as the byproducts are primarily siloxanes which are relatively benign, and the use of CO2 as a feedstock contributes to carbon utilization efforts. The high atom economy and reduced solvent load align perfectly with green chemistry principles, helping companies meet increasingly stringent environmental regulations and sustainability targets without compromising on yield or quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this atmospheric CO2 fixation technology in industrial settings. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the primary safety advantages of this CO2 fixation method over traditional phosgene routes?
A: This method eliminates the need for highly toxic phosgene and carbon monoxide, replacing them with non-toxic atmospheric CO2. Additionally, it avoids the isolation of hazardous isocyanate intermediates, significantly reducing operational risks and regulatory compliance burdens.
Q: Can this process be scaled for industrial production without expensive metal catalysts?
A: Yes, the process utilizes inexpensive Lewis bases and industrial silicon waste (PMHS) as promoters, completely avoiding precious metal catalysts. This simplifies the supply chain and removes the need for costly metal removal steps, making it highly suitable for large-scale commercial manufacturing.
Q: What is the typical purity profile and workup procedure for these heterocycles?
A: The reaction yields high-purity products often exceeding 90% isolated yield. The workup is exceptionally simple, requiring only cooling, water quenching, and suction filtration to separate the solid product, thereby minimizing solvent usage and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl Heterocycle Supplier
As the demand for sustainable and cost-effective pharmaceutical intermediates continues to rise, NINGBO INNO PHARMCHEM stands ready to leverage advanced synthetic methodologies like the one described in CN110878055B to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of benzimidazolone or benzoxazolone meets the exacting standards required for GMP manufacturing. Our commitment to quality is matched only by our dedication to safety and environmental stewardship, making us the ideal partner for companies seeking to optimize their supply chains.
We invite you to engage with our technical procurement team to discuss how this innovative CO2 fixation route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits this technology offers for your specific portfolio. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your manufacturing operations. Let us collaborate to build a more sustainable and resilient future for the fine chemical industry.
