Scalable One-Pot Synthesis of Triazoloindole Alcohols for Advanced Drug Discovery
Introduction to Patent CN114014859A
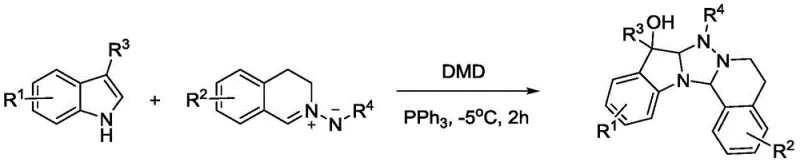
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to construct complex nitrogen-containing heterocyclic scaffolds, which serve as the backbone for numerous bioactive molecules. Patent CN114014859A introduces a groundbreaking technical solution for the synthesis of 9-substituted-4H-indolino[1,2,4]triazolo[5,1-a]isoquinolin-9-ols and their derivatives. This innovation represents a significant paradigm shift in the multifunctionalization of indole compounds, moving away from hazardous and cumbersome multi-step protocols towards a streamlined, efficient one-pot oxidative cyclization. By leveraging the unique oxidative capabilities of peroxyacetone (dimethyldioxirane) in an acetone solution, this method directly converts indole derivatives and azomethine imines into valuable triazoloindole alcohols. The technology addresses critical pain points in modern organic synthesis, including safety concerns, atom economy, and operational simplicity, making it an attractive candidate for the development of next-generation plant protection regulators and anticancer drug intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of nitrogen-rich heterocyclic systems like triazoloindoles has relied heavily on click chemistry reactions involving azides and alkynes. While effective in small-scale laboratory settings, these traditional pathways suffer from severe limitations when viewed through the lens of industrial applicability and safety. The primary concern is the inherent instability and explosive nature of organic azides, which necessitates specialized handling equipment and rigorous safety protocols, thereby inflating operational costs and limiting scalability. Furthermore, existing technologies for synthesizing these scaffolds often demand the use of expensive transition metal catalysts or harsh Lewis acids, which not only increase the raw material costs but also introduce challenging purification burdens. The requirement for high-temperature heating in many conventional routes further exacerbates energy consumption and can lead to the degradation of sensitive functional groups, resulting in lower overall yields and complex impurity profiles that are difficult to manage in a GMP environment.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN114014859A offers a remarkably elegant and efficient alternative. This novel approach utilizes a one-pot reaction system where indole compounds and azomethine imines are mixed directly with an oxidant, specifically a peroxyacetone acetone solution, under mild low-temperature conditions. The elimination of explosive azide precursors fundamentally enhances the safety profile of the synthesis, making it far more suitable for large-scale manufacturing. Moreover, the process operates without the need for additional organic solvents or metal catalysts, relying instead on the intrinsic reactivity of the peroxyacetone species to drive the cyclization and oxygenation simultaneously. This results in a reaction system that is not only scientifically rational and easy to operate but also maximizes atom economy, ensuring that a higher proportion of the starting materials are incorporated into the final product rather than being lost as waste.
Mechanistic Insights into Peroxyacetone-Mediated Oxidative Cyclization
The core of this technological breakthrough lies in the strategic use of peroxyacetone (often generated as dimethyldioxirane or DMDO in situ) as both the oxidant and the oxygen source. Mechanistically, the peroxyacetone species acts as a potent electrophilic oxidant that facilitates the activation of the indole C3 position or the azomethine imine moiety, promoting a cascade of bond-forming events that lead to the fused triazoloindole skeleton. Unlike metal-catalyzed oxidations that often proceed through radical pathways prone to side reactions, this peroxide-mediated pathway offers a controlled route to the desired hydroxylated product. The reaction proceeds efficiently at temperatures ranging from -78°C to 0°C, with optimal results observed around -10°C, indicating a low activation energy barrier that preserves the integrity of thermally labile substituents. This mildness is crucial for maintaining the structural diversity of the R-groups, allowing for the incorporation of sensitive functionalities such as halogens, methoxy groups, and various alkyl or aryl chains without decomposition.
From an impurity control perspective, the absence of transition metals eliminates the risk of metal-catalyzed side reactions, such as over-oxidation or polymerization, which are common pitfalls in traditional methods. The high selectivity of the peroxyacetone oxidation ensures that the reaction stops at the desired alcohol stage, minimizing the formation of ketone by-products or ring-opened degradation products. The one-pot nature of the synthesis further reduces the exposure of intermediates to potentially degrading conditions, as there are no isolation steps between the initial mixing and the final cyclization. This streamlined mechanistic pathway not only simplifies the reaction monitoring via TLC or HPLC but also ensures a cleaner crude product profile, significantly reducing the burden on downstream purification processes like column chromatography or recrystallization.
How to Synthesize Triazoloindole Alcohols Efficiently
The practical implementation of this synthesis route is designed for maximum operational efficiency and reproducibility. The protocol involves a straightforward sequence where the indole substrate and the azomethine imine precursor are combined in a reaction vessel, followed by the addition of the peroxyacetone oxidant solution. The mixture is then stirred at a controlled low temperature, typically around -10°C, for a duration of approximately 2 to 3 hours to ensure complete conversion. Following the reaction period, the workup procedure is equally simple, involving standard extraction techniques to separate the organic phase, followed by solvent removal under reduced pressure. The resulting crude solid can then be purified using standard silica gel column chromatography to afford the target triazoloindole alcohol in high purity. For detailed standardized synthesis steps and specific optimization parameters, please refer to the guide below.
- Combine indole compounds, azomethine imine compounds, and an oxidant (preferably peroxyacetone acetone solution) in a reaction vessel.
- Mix the reactants thoroughly and maintain the reaction temperature at or below 0°C, preferably around -10°C, for 2 to 3 hours.
- Upon completion, extract the crude product, remove solvents, and purify via column chromatography to isolate the target triazoloindole alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis methodology presents a compelling value proposition centered on cost efficiency and supply reliability. The elimination of expensive transition metal catalysts and the avoidance of hazardous azide reagents translate directly into substantial cost savings in raw material procurement. Furthermore, the simplified reaction workflow reduces the demand for specialized reactor equipment capable of withstanding high pressures or extreme temperatures, allowing for the utilization of standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure significantly lowers the capital expenditure required for technology transfer and scale-up.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the synthetic route. By consolidating multiple transformation steps into a single one-pot operation, the method eliminates the labor, time, and material costs associated with intermediate isolations and purifications. The absence of metal catalysts removes the need for costly and time-consuming heavy metal scavenging steps, which are often mandatory for pharmaceutical intermediates to meet strict regulatory limits. Additionally, the use of cheap and readily available starting materials, such as substituted indoles and isochroman-derived hydrazides, ensures a stable and low-cost supply chain, while the high atom economy minimizes waste disposal costs.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commodity chemicals that are widely available from multiple global suppliers, reducing the risk of bottlenecks associated with proprietary or scarce reagents. The mild reaction conditions, specifically the low-temperature requirement which is easily achievable with standard industrial chillers, ensure that the process is robust and less susceptible to thermal runaway incidents that can halt production. The high yields reported across a broad scope of substrates indicate a resilient process capable of accommodating variations in raw material quality without significant loss of productivity, thereby securing a consistent output of high-purity intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this technology aligns perfectly with green chemistry principles. The reduction in solvent usage, particularly the ability to run the reaction with minimal or no additional organic solvent beyond the oxidant solution, significantly lowers the volume of volatile organic compound (VOC) emissions. The high atom efficiency means less chemical waste is generated per kilogram of product, simplifying wastewater treatment and solid waste management. The straightforward scale-up potential, evidenced by the simple equipment requirements and mild conditions, allows for rapid expansion from pilot plant batches to multi-ton commercial production without the need for complex process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazoloindole synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: Why is this synthesis method safer than traditional click chemistry?
A: Traditional methods often rely on azide-alkyne click chemistry, where azides pose significant explosion hazards. This novel protocol utilizes stable indole and azomethine imine precursors with peroxyacetone, eliminating the safety risks associated with handling explosive azide intermediates.
Q: What are the key advantages regarding catalyst usage?
A: Unlike conventional approaches that require expensive transition metal catalysts or harsh Lewis acids, this method operates effectively without additional metal catalysts. This simplifies the downstream purification process and removes the need for costly heavy metal clearance steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly amenable to scale-up. It utilizes a one-pot strategy with mild reaction conditions (low temperature, no heating) and readily available raw materials, significantly reducing equipment complexity and energy consumption compared to multi-step high-temperature syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazoloindole Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this one-pot oxidative cyclization technology in accelerating the development of novel therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle the specific low-temperature requirements of this synthesis with precision, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cost-effective and safe synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this methodology can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for similar scaffolds and comprehensive route feasibility assessments for your custom synthesis projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
