Advanced Synthesis of 2-Trifluoromethyl Cyclopentenone Derivatives for Pharmaceutical Applications
Introduction to Next-Generation Cyclopentenone Synthesis
The field of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical development, particularly in the construction of complex heterocyclic scaffolds. Patent CN113999118A discloses a groundbreaking methodology for the preparation of 2-trifluoromethyl cyclopentenone derivatives, a structural motif prevalent in numerous bioactive natural products and drug candidates. These five-membered cyclic compounds, characterized by the presence of both carbon-carbon double bonds and carbon-oxygen double bonds, serve as pivotal intermediates in the synthesis of molecules with significant physiological activity. For instance, natural diterpenes such as pepluanol B exhibit potent cytotoxic and antitumor properties, while rupestonic acid is known for its ability to inhibit influenza viruses. 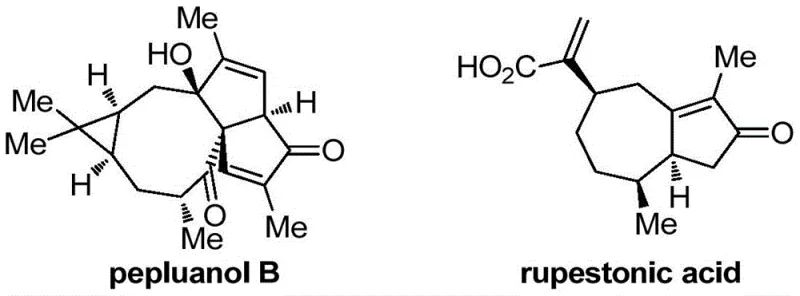 The introduction of fluorine-containing groups, specifically the trifluoromethyl group, into these scaffolds further enhances their metabolic stability and lipid solubility, making them highly desirable for drug modification strategies. The disclosed invention provides a robust and efficient route to access these valuable building blocks through a free radical serial cyclization reaction strategy. By leveraging a carbonyl-guided radical addition-migration-cyclization mechanism, this technology overcomes historical challenges associated with regioselectivity and substrate scope, offering a versatile platform for generating structural diversity in fine chemical manufacturing.
The introduction of fluorine-containing groups, specifically the trifluoromethyl group, into these scaffolds further enhances their metabolic stability and lipid solubility, making them highly desirable for drug modification strategies. The disclosed invention provides a robust and efficient route to access these valuable building blocks through a free radical serial cyclization reaction strategy. By leveraging a carbonyl-guided radical addition-migration-cyclization mechanism, this technology overcomes historical challenges associated with regioselectivity and substrate scope, offering a versatile platform for generating structural diversity in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the cyclopentenone ring skeleton has relied heavily on classical transformations such as the Pauson-Khand reaction (PKR) and Nazarov cyclization, both of which present significant operational and safety hurdles for large-scale production. The Pauson-Khand reaction, discovered in 1973, involves the reaction of alkyne cobalt hexacarbonyl complexes with alkenes to yield cyclopentenones.  However, this method suffers from severe limitations including the low reactivity of certain olefin components and unreliable regioselective control. More critically, the requirement for excess hazardous chemicals, such as pressurized carbon monoxide gas or stoichiometric amounts of toxic cobalt-carbonyl complexes, makes it environmentally unfriendly and difficult to scale safely in a commercial setting. Similarly, the Nazarov cyclization, which involves the rearrangement of divinyl ketones under strong acidic conditions, often faces issues with substrate compatibility and selectivity.
However, this method suffers from severe limitations including the low reactivity of certain olefin components and unreliable regioselective control. More critically, the requirement for excess hazardous chemicals, such as pressurized carbon monoxide gas or stoichiometric amounts of toxic cobalt-carbonyl complexes, makes it environmentally unfriendly and difficult to scale safely in a commercial setting. Similarly, the Nazarov cyclization, which involves the rearrangement of divinyl ketones under strong acidic conditions, often faces issues with substrate compatibility and selectivity. 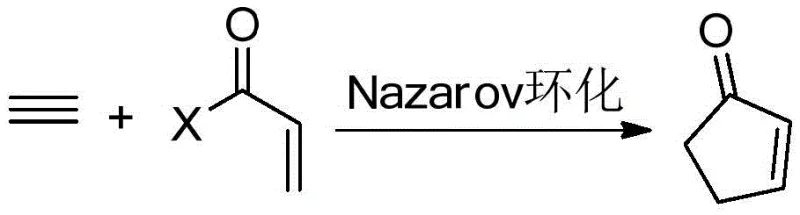 The reliance on harsh protonic acids like sulfuric acid or strong Lewis acids limits the functional group tolerance, often leading to decomposition of sensitive substrates or the formation of complex impurity profiles that are costly to remove during downstream processing.
The reliance on harsh protonic acids like sulfuric acid or strong Lewis acids limits the functional group tolerance, often leading to decomposition of sensitive substrates or the formation of complex impurity profiles that are costly to remove during downstream processing.
The Novel Approach
In stark contrast to these traditional methods, the novel approach detailed in the patent utilizes a copper-catalyzed radical cascade that operates under remarkably mild conditions. This strategy employs a Togni reagent as a safe and efficient source of trifluoromethyl radicals, eliminating the need for hazardous gases or stoichiometric heavy metals. The reaction proceeds via a regioselective addition of the trifluoromethyl radical to an internal alkyne, guided by a proximal carbonyl group, followed by a 1,5-hydrogen atom transfer and a subsequent 5-endo cyclization. This sequence efficiently constructs the 2-trifluoromethyl cyclopentenone core with high stereoselectivity. The method is distinguished by its wide substrate applicability, accommodating diverse functional groups including esters, ethers, and complex steroid backbones without the need for extensive protecting group manipulations. Furthermore, the operational simplicity of running the reaction at moderate temperatures (25-65°C) under a nitrogen atmosphere significantly reduces energy consumption and equipment complexity compared to high-pressure or high-temperature alternatives.
Mechanistic Insights into Copper-Catalyzed Radical Cyclization
The success of this synthesis lies in the intricate mechanistic pathway that orchestrates the formation of the five-membered ring. The reaction is initiated by the copper-catalyzed generation of a trifluoromethyl radical from the Togni reagent. This electrophilic radical undergoes regioselective addition to the internal alkyne of the alkynone substrate. A critical feature of this mechanism is the role of the carbonyl group as a directing moiety; it ensures that the radical addition occurs at the correct position to facilitate the subsequent cyclization steps. Following the initial addition, the resulting alkenyl radical intermediate undergoes a 1,5-hydrogen atom transfer (HAT), a process that migrates the radical center to a position favorable for ring closure. 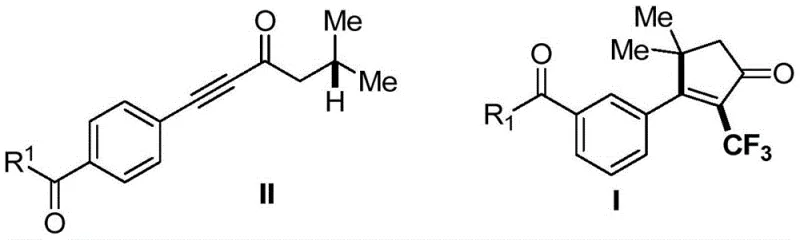 The final ring closure occurs via a 5-endo cyclization, a pathway that is traditionally disfavored due to Baldwin's rules but is rendered feasible here by the specific electronic and steric environment created by the guiding carbonyl group. The cycle concludes with an oxidative deprotonation step that restores aromaticity or conjugation and regenerates the catalyst, yielding the final 2-trifluoromethyl cyclopentenone derivative. This mechanistic elegance solves the long-standing problem of non-ideal regioselectivity associated with radical additions to internal alkynes, providing a predictable and reliable route to complex fluorinated scaffolds.
The final ring closure occurs via a 5-endo cyclization, a pathway that is traditionally disfavored due to Baldwin's rules but is rendered feasible here by the specific electronic and steric environment created by the guiding carbonyl group. The cycle concludes with an oxidative deprotonation step that restores aromaticity or conjugation and regenerates the catalyst, yielding the final 2-trifluoromethyl cyclopentenone derivative. This mechanistic elegance solves the long-standing problem of non-ideal regioselectivity associated with radical additions to internal alkynes, providing a predictable and reliable route to complex fluorinated scaffolds.
From an impurity control perspective, the mechanism offers distinct advantages. The high regioselectivity imposed by the carbonyl guide minimizes the formation of regioisomers, which are often difficult to separate. Additionally, the mild oxidative conditions prevent the over-oxidation of sensitive functional groups that might occur with stronger oxidants used in other protocols. The use of copper cyanide as a catalyst, rather than a stoichiometric reagent, ensures that residual metal levels in the final product can be kept low, simplifying the purification process. The compatibility with various R1 groups, ranging from simple alkyl chains to complex biomolecules like glucose and cholesterol derivatives, demonstrates the robustness of the catalytic cycle against steric hindrance and electronic variation, ensuring a clean reaction profile suitable for pharmaceutical grade intermediates.
How to Synthesize 2-Trifluoromethyl Cyclopentenone Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of execution in a standard laboratory or pilot plant setting. The protocol involves a straightforward one-pot procedure where the catalyst, radical source, and base are combined prior to the addition of the substrate. This pre-mixing ensures the active catalytic species are generated in situ before the reaction commences. The use of common solvents like ethyl acetate and acetonitrile further enhances the industrial viability of the process, avoiding the need for exotic or highly toxic solvents. The reaction times are moderate, typically completing within 8 to 15 hours, allowing for convenient overnight processing. Workup procedures are equally simple, involving standard aqueous quenching and extraction techniques followed by silica gel chromatography, which is a familiar unit operation for most chemical manufacturers. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction system by mixing copper cyanide, Togni reagent, and potassium carbonate in a reaction vessel under nitrogen atmosphere.
- Add the alkynone compound (Formula II) dissolved in ethyl acetate/acetonitrile to the mixture and heat to 50°C for 8-15 hours.
- Quench the reaction with water, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits regarding cost efficiency and supply reliability. The shift away from hazardous reagents and extreme reaction conditions translates directly into lower operational expenditures and reduced regulatory burden. By eliminating the need for pressurized carbon monoxide and stoichiometric cobalt complexes, the process removes significant safety risks and the associated costs of specialized containment equipment and hazardous waste disposal. This simplification of the safety profile allows for manufacturing in a broader range of facilities, thereby increasing supply chain flexibility and resilience. Furthermore, the high functional group tolerance means that fewer synthetic steps are required to protect and deprotect sensitive moieties, leading to a shorter overall synthesis timeline and higher overall throughput.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven primarily by the replacement of expensive and toxic stoichiometric reagents with a catalytic system. Traditional methods often require large excesses of cobalt complexes which are not only costly to purchase but also expensive to dispose of in compliance with environmental regulations. In contrast, the copper-catalyzed system uses minimal amounts of catalyst and generates less heavy metal waste, significantly lowering the cost of goods sold (COGS). Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to further operational savings. The high yields and selectivity observed across diverse substrates minimize material loss, ensuring that raw materials are converted efficiently into the desired high-value intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commercially available and stable reagents. Togni reagents and copper salts are widely sourced commodities, reducing the risk of supply bottlenecks associated with specialized organometallic complexes. The robustness of the reaction against moisture and air (when conducted under nitrogen) simplifies logistics and storage requirements for intermediates. Moreover, the ability to synthesize a wide variety of derivatives from a common alkynone precursor allows for a modular manufacturing approach. This flexibility enables rapid response to changing market demands for different API intermediates without the need for extensive retooling or process re-validation, ensuring a steady flow of materials to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and ton scales is facilitated by the absence of exothermic hazards and high-pressure requirements. The reaction can be safely performed in standard glass-lined or stainless steel reactors, making it compatible with existing multipurpose manufacturing assets. From an environmental standpoint, the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The elimination of stoichiometric heavy metals and hazardous gases simplifies wastewater treatment and废气 (exhaust gas) management. This improved environmental profile not only reduces compliance costs but also enhances the sustainability credentials of the final pharmaceutical products, a factor of increasing importance to global regulatory bodies and end consumers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 2-trifluoromethyl cyclopentenone derivatives. The answers are derived directly from the experimental data and mechanistic understanding provided in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this technology into existing production pipelines or new drug discovery programs. We encourage potential partners to review these details to fully appreciate the versatility and robustness of the described methodology.
Q: What are the key advantages of this radical cyclization method over traditional Pauson-Khand reactions?
A: Unlike the Pauson-Khand reaction which requires hazardous pressurized CO gas and stoichiometric cobalt complexes, this novel method utilizes mild copper catalysis and Togni reagents under atmospheric pressure, significantly improving operational safety and reducing heavy metal waste.
Q: Can this synthesis method accommodate complex bioactive substrates?
A: Yes, the method demonstrates excellent functional group tolerance, successfully synthesizing derivatives linked to fenofibrate, amino acids, glucose, and cholesterol scaffolds without compromising the integrity of sensitive moieties.
Q: How does the carbonyl guiding group influence the reaction selectivity?
A: The carbonyl group acts as a critical directing moiety that facilitates the regioselective addition of the trifluoromethyl radical to the internal alkyne, enabling the challenging 5-endo cyclization pathway that is typically difficult to achieve with terminal alkynes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Cyclopentenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced radical cyclization technology in accelerating the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry and radical reactions, maintaining stringent purity specifications through our rigorous QC labs. We are committed to delivering high-purity cyclopentenone derivatives that meet the exacting standards of the global pharmaceutical industry, supporting your goals in bringing life-saving medicines to market faster.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this method can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary intermediates. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and supply chain management.
