Advanced Visible Light Promoted Synthesis of 3-Methyl-3-Difluoroethyl-2-Oxindole Intermediates
The pharmaceutical and fine chemical industries are constantly seeking efficient, sustainable pathways to access complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. A significant breakthrough in this domain is documented in patent CN112194608A, which discloses a novel synthesis method for 3-methyl-3-difluoroethyl-2-oxindole compounds. This technology leverages visible light promotion to drive a multi-component one-pot reaction, effectively merging N-aryl acrylamide, iodobenzene diacetic acid, and difluoroacetic acid into a unified synthetic sequence. The strategic incorporation of the difluoroethyl moiety is of paramount importance, as fluorine atoms are known to drastically improve the metabolic stability and bioavailability of drug candidates. By utilizing a self-photocatalytic mechanism without the need for expensive external photocatalysts or transition metals, this invention represents a paradigm shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of 2-oxindole frameworks bearing fluorinated side chains has relied on harsh reaction conditions that pose significant challenges for industrial scale-up and environmental compliance. Conventional routes often necessitate the use of stoichiometric amounts of toxic transition metal catalysts, strong oxidants, or extreme temperatures that can degrade sensitive functional groups. Furthermore, many existing protocols require multi-step sequences with intermediate isolation, leading to accumulated waste and reduced overall atom economy. The reliance on precious metal catalysts not only inflates the raw material costs but also introduces stringent purification requirements to meet residual metal limits mandated by regulatory bodies for active pharmaceutical ingredients. These factors collectively create bottlenecks in the supply chain, extending lead times and complicating the procurement of high-purity intermediates necessary for clinical development.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN112194608A introduces a streamlined, visible-light promoted protocol that operates under remarkably mild conditions. The core innovation lies in the use of blue light irradiation, specifically within the 430-445 nm range, to activate the reaction system in situ. This approach eliminates the need for external photocatalysts, as the reaction intermediate itself exhibits sufficient photosensitivity to drive the transformation. The process utilizes readily available and inexpensive reagents such as iodobenzene diacetic acid and difluoroacetic acid, which react to form a reactive hypervalent iodine species capable of generating difluoromethyl radicals. This one-pot strategy significantly simplifies the operational workflow, reducing solvent consumption and processing time while delivering excellent yields exceeding 90% under optimized conditions.
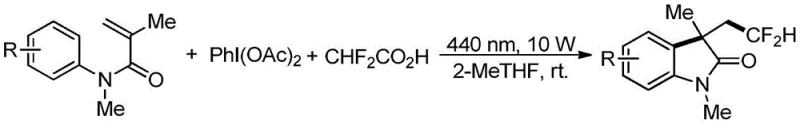
Mechanistic Insights into Visible Light Promoted Radical Cyclization
To fully appreciate the robustness of this synthetic route, it is essential to understand the underlying mechanistic pathway that governs the formation of the 3-methyl-3-difluoroethyl-2-oxindole core. The reaction initiates with the interaction between iodobenzene diacetic acid and difluoroacetic acid, which generates a bis(difluoroacetoxy)iodobenzene intermediate in situ. Upon irradiation with 440 nm blue light, this hypervalent iodine species undergoes homolytic cleavage to produce an iodine-centered radical and a difluoroacetoxy radical. The latter rapidly decarboxylates to release carbon dioxide and generate the critical difluoromethyl radical species. This highly reactive radical then adds across the electron-deficient double bond of the N-aryl acrylamide substrate, forming a new carbon-carbon bond and a transient carbon-centered radical intermediate adjacent to the aromatic ring.
Following the initial radical addition, the cyclization event is triggered through an intramolecular attack on the aromatic ring, facilitated by single electron transfer processes involving the iodine species. The resulting cationic intermediate undergoes deprotonation, driven by the difluoroacetate anions present in the reaction medium, to restore aromaticity and finalize the formation of the oxindole ring system. This elegant cascade ensures high regioselectivity and minimizes the formation of byproducts, as the radical intermediates are short-lived and efficiently consumed in the productive cycle. The absence of transition metals further simplifies the impurity profile, making downstream purification significantly more straightforward compared to metal-catalyzed alternatives.
How to Synthesize 3-Methyl-3-Difluoroethyl-2-Oxindole Efficiently
Implementing this visible-light promoted synthesis in a laboratory or pilot plant setting requires careful attention to light source specifications and reagent ratios to maximize efficiency. The standard protocol involves dissolving the N-aryl acrylamide substrate along with iodobenzene diacetic acid and difluoroacetic acid in a benign solvent such as 2-methyltetrahydrofuran. The mixture is then subjected to irradiation from a 10W LED blue light source at room temperature for a duration of approximately 3 to 5 hours. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and optimal yield.
- Prepare the reaction mixture by combining N-aryl acrylamide, iodobenzene diacetic acid, and difluoroacetic acid in a suitable solvent such as 2-methyltetrahydrofuran.
- Irradiate the reaction mixture with a blue LED light source, specifically optimized at a wavelength of 440 nm and a power of 10W, while stirring at room temperature.
- Monitor the reaction progress via TLC, and upon completion, purify the crude product using column chromatography to isolate the target 3-methyl-3-difluoroethyl-2-oxindole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this visible-light promoted methodology offers substantial strategic advantages over traditional manufacturing routes. The primary benefit stems from the drastic simplification of the raw material portfolio; the process relies on commodity chemicals like difluoroacetic acid and hypervalent iodine reagents rather than scarce, price-volatile transition metals. This shift significantly mitigates supply risk and stabilizes input costs, ensuring a more predictable budget for long-term production campaigns. Additionally, the elimination of heavy metal catalysts removes the need for expensive scavenging resins and complex analytical testing for metal residues, thereby reducing the overall cost of goods sold and accelerating the release of batches for downstream processing.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the use of low-energy LED light sources which are inexpensive to operate and maintain compared to high-pressure mercury lamps or thermal heating systems. By conducting the reaction at room temperature, the facility saves significantly on energy consumption associated with heating and cooling cycles. Furthermore, the high selectivity of the reaction minimizes the generation of difficult-to-separate impurities, leading to higher recovery rates of the final product and reduced waste disposal costs. The avoidance of noble metals also means that the capital expenditure for specialized corrosion-resistant equipment is lowered, as the reaction medium is less aggressive than many traditional acidic or basic conditions.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain resilience. Since the reagents are stable and commercially available in bulk quantities, manufacturers are less susceptible to disruptions caused by the shortage of specialized catalysts. The tolerance of the reaction to various functional groups, as demonstrated by the successful synthesis of derivatives with methyl and bromo substituents, allows for a flexible production schedule where multiple analogues can be produced on the same line with minimal changeover time. This flexibility is crucial for CDMOs managing diverse portfolios of clinical candidates, enabling rapid response to changing customer demands without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: From an environmental perspective, this technology aligns perfectly with green chemistry principles by utilizing visible light as a traceless reagent and generating minimal hazardous waste. The use of 2-methyltetrahydrofuran, a bio-based solvent, further enhances the sustainability profile of the process, appealing to pharmaceutical companies with strict environmental, social, and governance (ESG) mandates. Scaling this reaction is feasible because the light penetration issues common in photochemistry can be managed using flow chemistry reactors or optimized vessel geometries, ensuring consistent irradiation even at larger volumes. This scalability ensures that the transition from gram-scale discovery to kilogram-scale commercial production is seamless and reliable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light promoted synthesis technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the critical reaction conditions for this visible light promoted synthesis?
A: The reaction requires irradiation with blue visible light, optimally at a wavelength of 440 nm using a 10W LED source. The process operates efficiently at room temperature in solvents like 2-methyltetrahydrofuran or acetonitrile, avoiding the need for external photocatalysts or transition metals.
Q: Does this method support diverse functional groups on the substrate?
A: Yes, the methodology exhibits excellent substrate compatibility. It tolerates various substituents on the N-aryl ring, including alkyl groups like methyl, as well as halogens such as bromine, maintaining high yields across different electronic environments.
Q: Why is the difluoroethyl group significant in pharmaceutical intermediates?
A: The introduction of a 1,1-difluoroethyl group significantly enhances the metabolic stability and bioavailability of bioactive molecules. This modification is crucial for developing drug candidates with improved pharmacokinetic profiles, making this synthesis route highly valuable for medicinal chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methyl-3-Difluoroethyl-2-Oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light promoted chemistry in the production of advanced pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic discoveries are successfully translated into robust industrial processes. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We are committed to delivering high-quality 3-methyl-3-difluoroethyl-2-oxindole derivatives that empower your drug discovery programs with reliable supply and superior consistency.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can optimize your specific project requirements. By engaging with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume needs, helping you identify opportunities for margin improvement without sacrificing quality. Please contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary targets, and let us help you accelerate your path to market.
