Advanced Radical Cyclization Strategy for Scalable 1,2-Dihydronaphthalene Derivative Manufacturing
Introduction to Patent CN108129287B Technology
The chemical industry is constantly seeking more efficient and sustainable pathways to construct complex cyclic scaffolds essential for modern drug discovery. Patent CN108129287B introduces a groundbreaking methodology for the synthesis of 1,2-dihydronaphthalene derivatives through the direct reaction of methylenecyclopropane compounds (MCPs) with aldehyde compounds. This innovation represents a significant leap forward in C-C bond activation and functionalization, specifically targeting the challenging aldehyde C(sp2)-H bond. Unlike traditional methods that often rely on expensive and toxic transition metals, this protocol utilizes a combination of a Lewis acid catalyst and a radical initiator to drive the cyclization. The ability to seamlessly integrate highly strained three-membered rings with readily available aldehydes opens up vast possibilities for generating diverse molecular libraries. For R&D teams focused on building complex natural product analogs or bioactive skeletons, this technology offers a robust platform for rapid structural diversification without the need for pre-functionalized starting materials.
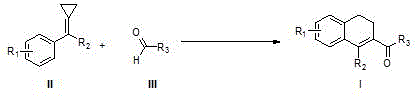
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of methylenecyclopropanes has been heavily dependent on noble or specific transition metal catalysts such as Copper(I) or Manganese(III) acetate. These conventional approaches often suffer from significant drawbacks including high catalyst costs, sensitivity to air and moisture, and limited substrate tolerance. For instance, previous literature describes Cu(I)-catalyzed intramolecular trifluoromethylation or Mn(OAc)3 promoted reactions which require strict anhydrous conditions and often produce stoichiometric amounts of metal waste. Furthermore, many existing protocols are restricted to reactions with heteroatoms or specific activated methylene compounds like malonates, failing to address the direct utilization of simple aldehyde C-H bonds. This limitation restricts the chemical space accessible to medicinal chemists and imposes a heavy burden on downstream purification processes due to metal contamination. The reliance on specialized reagents also complicates supply chain logistics, making large-scale production economically unviable for many fine chemical applications.
The Novel Approach
The methodology disclosed in CN108129287B overcomes these barriers by employing an earth-abundant Iron(II) catalyst coupled with a peroxide-based radical initiator. This system enables the direct radical cyclization between MCPs and the formyl C-H bond of aldehydes, a transformation that was previously unreported or inefficient. By shifting from precious metal catalysis to a radical manifold mediated by FeCl2, the process drastically reduces raw material costs and simplifies the reaction setup. The use of di-tert-butyl peroxide (DTBP) as the initiator ensures a steady generation of radicals at moderate temperatures, facilitating the homolytic cleavage required for C-H activation. This approach not only expands the scope of compatible substrates to include a wide variety of substituted benzaldehydes but also improves the overall atom economy of the synthesis. The result is a streamlined process that delivers high-purity 1,2-dihydronaphthalene cores with minimal environmental footprint, aligning perfectly with the goals of green chemistry and sustainable manufacturing.
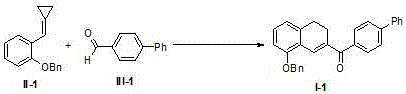
Mechanistic Insights into FeCl2-Catalyzed Radical Cyclization
The success of this transformation lies in the intricate interplay between the Lewis acid catalyst and the radical species generated in situ. The mechanism initiates with the thermal decomposition of the radical initiator, DTBP, which cleaves to form tert-butoxy radicals and methyl radicals. These highly reactive oxygen-centered radicals then abstract a hydrogen atom from the aldehyde substrate, generating a nucleophilic acyl radical intermediate. This acyl radical subsequently adds across the electron-rich carbon-carbon double bond of the methylenecyclopropane moiety. The addition triggers the relief of ring strain inherent in the three-membered cyclopropane ring, leading to a ring-opening event that generates a stabilized benzylic radical species. This intermediate then undergoes an intramolecular cyclization onto the adjacent aromatic ring, forming the new six-membered ring characteristic of the dihydronaphthalene skeleton. Finally, hydrogen abstraction from the radical intermediate by another tert-butoxy radical terminates the cycle and yields the final product. Throughout this cascade, the FeCl2 catalyst plays a crucial role in stabilizing the radical intermediates and potentially coordinating with the carbonyl oxygen to enhance electrophilicity, thereby lowering the activation energy for the key bond-forming steps.
Impurity control in this radical process is managed through the careful selection of reaction parameters and the inherent selectivity of the radical cascade. The use of an inert argon atmosphere prevents the quenching of radical intermediates by atmospheric oxygen, which could otherwise lead to peroxidation side products or polymerization. Additionally, the specific choice of n-Butyl acetate as the solvent provides a stable medium that does not participate in unwanted hydrogen abstraction reactions, unlike ethers or alcohols which might compete as radical traps. The Lewis acid nature of FeCl2 also helps in directing the regioselectivity of the radical addition, ensuring that the ring-opening occurs at the correct bond to yield the desired 1,2-dihydronaphthalene isomer rather than alternative rearrangement products. By optimizing the molar ratios of the initiator and catalyst, the concentration of free radicals is kept at an ideal level to promote the desired cyclization pathway while minimizing bimolecular radical termination events that lead to dimerization impurities.
How to Synthesize 1,2-Dihydronaphthalene Derivatives Efficiently
Implementing this synthesis route requires precise control over reaction conditions to maximize yield and purity. The protocol involves charging a reactor with the methylenecyclopropane derivative, the aldehyde coupling partner, and the iron catalyst, followed by rigorous degassing to establish an inert environment. Once the system is purged, the radical initiator and solvent are introduced, and the mixture is heated to facilitate the radical cascade. The simplicity of the workup procedure, involving solvent evaporation and standard chromatographic purification, makes this method highly attractive for both laboratory scale optimization and pilot plant operations. Detailed standardized synthesis steps are provided below to ensure reproducibility and safety during implementation.
- Charge a reactor with methylenecyclopropane derivative, aldehyde compound, and FeCl2 catalyst under inert atmosphere.
- Add radical initiator DTBP and organic solvent n-BuOAc after purging the system with argon gas multiple times.
- Heat the reaction mixture to 90°C for 24 hours, then evaporate solvent and purify the residue via chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial advantages by eliminating the dependency on scarce and volatile transition metal markets. The substitution of expensive copper or palladium catalysts with ferrous chloride results in a drastic reduction in raw material costs, directly impacting the bottom line for large-scale manufacturing campaigns. Furthermore, the reagents involved, such as DTBP and n-Butyl acetate, are commodity chemicals with stable global supply chains, ensuring consistent availability and reducing the risk of production delays due to material shortages. The robustness of the reaction conditions also means that specialized equipment for handling air-sensitive materials is less critical, allowing for production in standard stainless steel reactors commonly found in multi-purpose chemical facilities. This flexibility enhances supply chain resilience and allows for faster turnaround times from order to delivery.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the need for costly metal scavenging steps and complex waste treatment protocols associated with heavy metal disposal. This simplification of the downstream processing significantly lowers the operational expenditure per kilogram of product. Additionally, the high atom economy of the direct C-H functionalization reduces the amount of waste generated, further contributing to cost savings in waste management and environmental compliance. The use of inexpensive iron salts instead of proprietary ligand-metal complexes ensures that the cost of goods sold remains competitive even at commercial volumes.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit from the use of widely available starting materials that are produced by multiple suppliers globally. This diversification of the supply base mitigates the risk of single-source bottlenecks that often plague specialty chemical manufacturing. The stability of the reagents allows for longer shelf-life and easier storage requirements, reducing inventory carrying costs and minimizing the risk of material degradation before use. Consequently, manufacturers can maintain leaner inventory levels while still guaranteeing continuous production schedules to meet customer demand.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and pressures, making it inherently safer and easier to scale from gram to ton quantities without significant engineering hurdles. The solvent system is relatively benign compared to chlorinated solvents often used in traditional organic synthesis, facilitating easier solvent recovery and recycling. This alignment with green chemistry principles not only reduces the environmental footprint but also simplifies regulatory approvals and permits for new manufacturing lines. The process generates minimal hazardous waste, streamlining the path to commercial validation and market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. Understanding these details is crucial for evaluating the feasibility of this route for specific project requirements. The answers are derived directly from the experimental data and technical disclosures within the patent documentation to ensure accuracy and reliability for decision-making purposes.
Q: What is the primary catalyst used in this novel synthesis method?
A: The process utilizes Ferrous Chloride (FeCl2) as a Lewis acid catalyst, which is significantly more cost-effective and earth-abundant compared to traditional transition metal catalysts like Copper or Manganese.
Q: How does this method activate the C-H bond of the aldehyde?
A: The reaction employs Di-tert-butyl peroxide (DTBP) as a radical initiator. Upon heating, DTBP decomposes to generate tert-butoxy radicals which abstract a hydrogen atom from the aldehyde C(sp2)-H bond, forming a reactive acyl radical intermediate.
Q: What are the typical reaction conditions for optimal yield?
A: Optimal conditions involve using n-Butyl acetate (n-BuOAc) as the solvent, maintaining a temperature of 90°C, and reacting for approximately 24 hours under an argon atmosphere to prevent oxidative side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Dihydronaphthalene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this radical cyclization technology for the development of next-generation pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your innovative molecules can reach the market efficiently. Our state-of-the-art facilities are equipped to handle radical reactions safely, and our rigorous QC labs enforce stringent purity specifications to meet the highest industry standards. We are committed to translating complex academic methodologies into robust, GMP-compliant manufacturing processes that deliver consistent quality.
We invite you to collaborate with our technical team to explore how this cost-effective synthesis route can be integrated into your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and commercialization goals, ensuring a seamless partnership from process development to full-scale supply.
