Scalable Metal-Free Synthesis of 3-(4-Pyridine) Indole Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and economical pathways for constructing complex heterocyclic scaffolds, particularly those exhibiting significant biological activity. Patent CN108976198B introduces a groundbreaking synthetic methodology for 3-(4-pyridine) indole compounds, a class of molecules identified as potential inosine monophosphate dehydrogenase and Rho-kinase inhibitors. This innovation addresses critical bottlenecks in the production of high-purity pharmaceutical intermediates by utilizing a transition metal-free, one-pot tandem reaction strategy. By leveraging inexpensive iodine catalysts and readily available oxime acetates, this technology offers a sustainable alternative to traditional multi-step syntheses. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering cost-effective solutions without compromising on chemical integrity or supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-(4-pyridine) indole frameworks has relied heavily on palladium-catalyzed Suzuki coupling reactions or metal-catalyzed alkyne cycloaddition processes. These conventional methodologies, while effective in academic settings, present substantial challenges for industrial application. The primary drawback lies in the reliance on precious transition metals, which not only inflate raw material costs but also introduce significant environmental and safety liabilities associated with heavy metal waste disposal. Furthermore, these multi-step sequences often require rigorous purification of intermediates at each stage, leading to cumulative yield losses and extended production timelines. The harsh reaction conditions frequently necessitated by these metal-catalyzed routes can also limit substrate compatibility, restricting the structural diversity accessible to medicinal chemists exploring structure-activity relationships.
The Novel Approach
In stark contrast, the methodology disclosed in CN108976198B utilizes a direct, one-pot tandem reaction between oxime acetate compounds and 3-formyl indoles. This approach completely bypasses the need for expensive transition metal catalysts, substituting them with economical iodine sources such as elemental iodine or N-iodosuccinimide. The reaction operates under mild thermal conditions, typically between 100-130°C, in common organic solvents like 1,4-dioxane or toluene. This simplification of the synthetic route drastically reduces the operational complexity and resource consumption associated with manufacturing. By consolidating multiple transformation steps into a single vessel, the process minimizes solvent usage and waste generation, aligning perfectly with modern green chemistry principles while enhancing the overall efficiency of producing high-purity pharmaceutical intermediates.
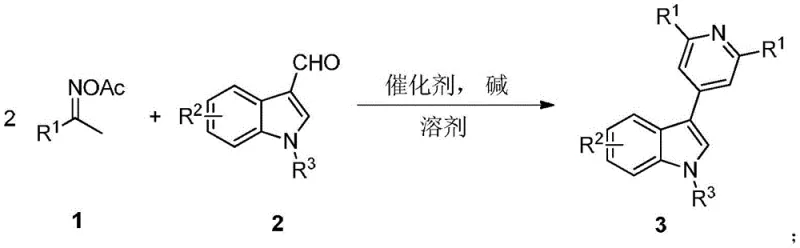
Mechanistic Insights into Iodine-Catalyzed Tandem Cyclization
The core of this technological advancement lies in the unique reactivity of the iodine catalyst in facilitating a cascade of bond-forming events. Mechanistically, the iodine species acts as a Lewis acid or electrophilic activator, promoting the initial condensation between the oxime acetate and the aldehyde functionality of the indole. This activation lowers the energy barrier for the subsequent cyclization steps that construct the pyridine ring fused to the indole core. The presence of a nitrogen-containing organic base, such as pyridine or triethylamine, plays a dual role in neutralizing acidic byproducts and potentially assisting in the deprotonation steps required for aromatization. This synergistic interaction between the iodine catalyst and the organic base ensures high regioselectivity, directing the formation of the desired 3-(4-pyridine) substitution pattern exclusively.
From an impurity control perspective, this metal-free mechanism offers distinct advantages over transition metal catalysis. The absence of palladium or other heavy metals eliminates the risk of metal leaching into the final product, a critical quality attribute for API intermediates. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups often present on the indole or phenyl rings, such as esters, halogens, or nitro groups. The patent data demonstrates that substrates bearing electron-withdrawing or electron-donating groups are well-tolerated, yielding clean reaction profiles with minimal side products. This inherent selectivity simplifies downstream processing, reducing the burden on purification teams and ensuring consistent batch-to-batch quality essential for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize 3-(4-Pyridine) Indole Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The general procedure involves dissolving the specific oxime acetate derivative and the 3-formyl indole substrate in a dry aprotic solvent. Following the addition of the iodine catalyst and the organic base, the mixture is heated in a sealed vessel to maintain pressure and prevent solvent loss. The reaction progress is typically monitored until completion, after which standard aqueous workup procedures involving extraction with ethyl acetate and washing with sodium thiosulfate solution effectively remove residual iodine. The crude product is then purified via silica gel chromatography to afford the target compound. For detailed operational specifics regarding stoichiometry and temperature optimization, please refer to the standardized guide below.
- Dissolve the oxime acetate compound and 3-formyl indole derivative in a suitable solvent such as 1,4-dioxane or toluene.
- Add an iodine-based catalyst (e.g., elemental iodine) and a nitrogen-containing organic base like pyridine or triethylamine to the reaction mixture.
- Heat the sealed reaction vessel to a temperature between 100-130°C for approximately 12 hours, then quench with water and extract with ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-catalyzed protocol translates into tangible strategic benefits beyond mere chemical curiosity. The elimination of precious metal catalysts represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. By replacing expensive palladium reagents with commodity-grade iodine, the direct material costs are substantially lowered, improving the overall margin profile of the final active ingredient. Additionally, the simplified one-pot nature of the reaction reduces the number of unit operations required, thereby decreasing labor costs and facility occupancy time. This efficiency gain allows for faster throughput and more responsive supply chains capable of meeting fluctuating market demands without the need for extensive capital investment in new equipment.
- Cost Reduction in Manufacturing: The shift away from transition metal catalysis removes the financial burden associated with purchasing and recovering expensive metals like palladium. Since iodine is abundant and inexpensive, the raw material expenditure is drastically minimized. Furthermore, the avoidance of multi-step purification processes for intermediates reduces solvent consumption and waste disposal fees, contributing to a leaner and more economically viable production model that enhances competitiveness in the global marketplace.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically oxime acetates and 3-formyl indoles, are commercially available and easy to prepare from bulk chemicals. This accessibility mitigates the risk of supply disruptions often caused by the scarcity of specialized organometallic reagents. The robustness of the reaction conditions also means that production is less susceptible to variations in raw material quality, ensuring a steady and predictable flow of intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simplicity and the use of standard organic solvents. The absence of heavy metal waste simplifies environmental compliance and wastewater treatment protocols, reducing the regulatory burden on manufacturing sites. This green chemistry profile not only aligns with corporate sustainability goals but also facilitates smoother regulatory approvals for new drug applications by minimizing the toxicological risks associated with metal residues in the final drug substance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential partners and licensees.
Q: What are the advantages of this iodine-catalyzed method over traditional palladium-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts like palladium, significantly reducing raw material costs and simplifying the purification process by avoiding heavy metal residue removal.
Q: What is the substrate scope for this synthesis regarding substituents on the indole ring?
A: The protocol demonstrates high tolerance for various functional groups including methyl, methoxy, fluoro, chloro, nitro, and ester groups on the indole ring, allowing for diverse derivative synthesis.
Q: Does this process require harsh reaction conditions or specialized equipment?
A: No, the reaction proceeds under relatively mild conditions (100-130°C) in common organic solvents using standard sealed tube apparatus, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(4-Pyridine) Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methods described in CN108976198B for the production of advanced pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-(4-pyridine) indole derivatives meets the highest international standards for safety and efficacy required by top-tier pharmaceutical companies.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
