Scalable Ruthenium-Catalyzed Synthesis of Polyaryl Naphthalene Derivatives for Advanced Materials
Scalable Ruthenium-Catalyzed Synthesis of Polyaryl Naphthalene Derivatives for Advanced Materials
The landscape of fine chemical synthesis is undergoing a transformative shift towards more sustainable and economically viable catalytic processes, particularly in the realm of complex aromatic systems. Patent CN107935812B introduces a groundbreaking methodology for the preparation of polyaryl substituted naphthalene derivatives, utilizing a cost-effective ruthenium catalyst to activate aromatic ketone β-H bonds. This innovation represents a significant leap forward for manufacturers of high-performance organic fluorescent materials and semiconductor intermediates, as it eliminates the need for stoichiometric oxidants and harsh reaction conditions typically associated with traditional cyclometallation. By leveraging a simple base system and mild thermal conditions, this technology not only streamlines the synthetic pathway but also drastically reduces the environmental footprint associated with heavy metal waste disposal. For R&D directors and procurement specialists alike, this patent offers a compelling blueprint for achieving high-purity intermediates with enhanced process safety and reduced operational expenditures.
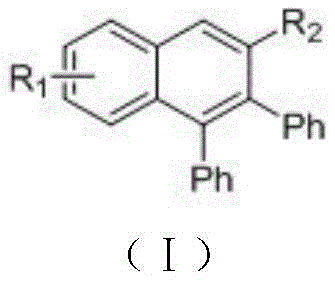
In the context of modern pharmaceutical and optoelectronic manufacturing, the ability to construct complex polycyclic aromatic hydrocarbons efficiently is paramount. The disclosed method specifically targets the synthesis of structures that serve as critical building blocks for tyrosine protein kinase inhibitors and blue light emitting materials. The versatility of this approach allows for the introduction of diverse functional groups, thereby enabling the rapid generation of compound libraries for drug discovery and material science applications. As a reliable polyaryl substituted naphthalene supplier, understanding the nuances of such catalytic cycles is essential for maintaining a competitive edge in the global supply chain. The transition from laboratory-scale curiosity to industrial reality hinges on the robustness of the catalytic system, and the ruthenium-based protocol described herein provides exactly the stability and reproducibility required for large-scale operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyaryl substituted naphthalene derivatives has been plagued by significant technical and economic hurdles that hindered their widespread adoption in commercial settings. Conventional methodologies often relied heavily on cyclometallation strategies or the use of aryl halides and aryl acids, which necessitated extremely harsh reaction conditions and generated substantial quantities of hazardous waste. A major bottleneck in these traditional pathways was the requirement for stoichiometric amounts of metal salts, such as copper or silver, to act as oxidants to complete the catalytic cycle. This reliance not only inflated the raw material costs due to the high price of these precious metals but also introduced severe challenges in downstream processing, where removing trace heavy metal contaminants to meet stringent pharmaceutical purity specifications became a costly and time-consuming endeavor. Furthermore, the environmental impact of disposing of large volumes of heavy metal-laden waste streams posed a significant regulatory burden, making these processes increasingly untenable in an era of heightened ecological scrutiny and green chemistry mandates.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel ruthenium-catalyzed approach detailed in the patent data offers a streamlined and environmentally benign alternative that redefines efficiency in aromatic coupling reactions. By employing a dimeric ruthenium complex, specifically [RuCl2(p-cymene)]2, the process achieves direct C-H activation of the aromatic ketone beta-position without the need for any external oxidants or specialized ligands. This oxidative annulation proceeds smoothly under mild thermal conditions, typically around 100°C in a non-polar solvent like toluene, utilizing simple and inexpensive bases such as potassium acetate and sodium carbonate. The elimination of stoichiometric oxidants is a game-changer for cost reduction in optoelectronic material manufacturing, as it simplifies the reaction workup and significantly lowers the E-factor of the process. This method exemplifies the principles of atom economy and waste prevention, providing a robust platform for the scalable production of high-value naphthalene derivatives that are essential for next-generation organic electronics and therapeutic agents.
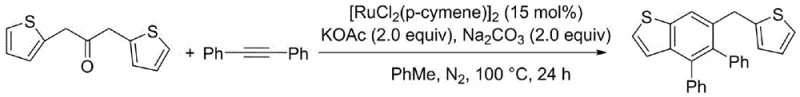
Mechanistic Insights into Ruthenium-Catalyzed C-H Activation and Cyclization
The core of this technological advancement lies in the sophisticated mechanistic pathway facilitated by the ruthenium catalyst, which enables the selective activation of inert C-H bonds under remarkably mild conditions. The catalytic cycle initiates with the coordination of the ruthenium species to the carbonyl oxygen of the alkyl aryl ketone, directing the metal center to the proximal beta-C-H bond for activation. This directed metallation forms a stable ruthenacycle intermediate, which is the key determinant of the reaction's regioselectivity and efficiency. Subsequently, the insertion of the diphenylacetylene alkyne into the ruthenium-carbon bond occurs, followed by a reductive elimination step that constructs the new carbon-carbon bonds necessary to form the six-membered naphthalene ring system. Unlike palladium-catalyzed systems that often require phosphine ligands to stabilize the active species, this ruthenium system operates effectively with the inherent stability of the p-cymene ligand, reducing the complexity and cost of the catalyst formulation. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters for specific substrate classes, ensuring maximum yield and minimal byproduct formation during the synthesis of complex polyaromatic frameworks.
From an impurity control perspective, the absence of external oxidants plays a pivotal role in maintaining the integrity of the final product profile. In traditional oxidative couplings, side reactions involving the oxidant can lead to over-oxidation of sensitive functional groups or the formation of homocoupling byproducts, which complicate purification and reduce overall yield. The reductive nature of this specific ruthenium cycle, driven solely by the internal hydrogen transfer and base mediation, minimizes these parasitic pathways, resulting in a cleaner crude reaction mixture. This inherent selectivity translates directly to simplified downstream processing, as the burden on chromatographic separation is significantly reduced. For quality assurance teams, this means a more consistent impurity profile across different batches, which is critical when supplying intermediates for regulated industries like pharmaceuticals. The robustness of the catalytic system against various functional groups, including halogens and trifluoromethyl groups, further underscores its utility in synthesizing diverse analogues without compromising the purity standards required for high-performance applications.
How to Synthesize Polyaryl Substituted Naphthalene Efficiently
The practical implementation of this synthesis route involves a straightforward procedure that balances reaction efficiency with operational simplicity, making it highly attractive for process chemists looking to translate laboratory findings to pilot plant scales. The standard protocol dictates the combination of diphenylacetylene and the chosen aromatic ketone substrate in a sealed reaction vessel, ensuring an inert atmosphere to prevent catalyst deactivation. The addition of the ruthenium catalyst at a loading of approximately 15 mol% alongside a dual-base system creates the optimal environment for the cyclization to proceed. Heating the mixture to 100°C for a duration of 24 hours allows for complete conversion, after which standard aqueous workup and column chromatography yield the target polyaryl naphthalene derivative. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during execution.
- Combine diphenylacetylene and aromatic ketone substrates in a sealed tube with a non-polar solvent like toluene.
- Add the ruthenium catalyst [RuCl2(p-cymene)]2 along with a mixed base system of potassium acetate and sodium carbonate.
- Heat the reaction mixture to 100°C under a nitrogen atmosphere for 24 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed technology presents a multitude of strategic benefits that extend far beyond mere chemical novelty. The primary value proposition lies in the substantial cost savings achieved through the elimination of expensive stoichiometric oxidants and the use of a relatively cheap ruthenium precursor compared to other noble metals. By removing the need for copper or silver salts, the raw material bill of materials is significantly optimized, and the costs associated with hazardous waste treatment are drastically curtailed. Furthermore, the use of common solvents like toluene and simple inorganic bases ensures that the supply chain remains resilient against fluctuations in the availability of exotic reagents. This robustness enhances supply chain reliability, as the risk of production delays due to reagent shortages is minimized, ensuring a steady flow of critical intermediates to downstream manufacturing units. The simplified purification process also contributes to shorter cycle times, allowing for faster turnover and improved responsiveness to market demands.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the removal of stoichiometric oxidants which traditionally account for a significant portion of reagent costs in oxidative coupling reactions. By relying on a catalytic amount of ruthenium and inexpensive bases, the direct material costs are lowered, while the indirect costs related to waste disposal and environmental compliance are also significantly reduced. The simplified workup procedure means less solvent consumption and reduced energy usage for purification, contributing to a leaner and more cost-effective manufacturing operation. Additionally, the high atom economy of the reaction ensures that a greater proportion of the starting materials end up in the final product, maximizing resource utilization and minimizing waste generation throughout the production lifecycle.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as toluene, sodium carbonate, and potassium acetate ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized ligands or sensitive organometallic reagents. This accessibility guarantees a consistent supply of raw materials, which is critical for maintaining continuous production schedules and meeting tight delivery deadlines for global clients. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, further stabilizing the supply chain. For supply chain planners, this translates to reduced safety stock requirements and lower inventory carrying costs, as the risk of batch failure due to reagent instability is negligible.
- Scalability and Environmental Compliance: From a scalability perspective, the mild reaction conditions and the absence of hazardous oxidants make this process inherently safer and easier to scale from kilogram to tonne levels. The reduced generation of heavy metal waste aligns perfectly with increasingly stringent environmental regulations, facilitating smoother permitting processes and reducing the risk of regulatory shutdowns. The use of a closed system under nitrogen atmosphere further enhances safety by minimizing exposure to potential allergens or irritants. This environmental compatibility not only future-proofs the manufacturing process against tightening green chemistry standards but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B negotiations with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium-catalyzed synthesis method, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for assessing the feasibility of integrating this technology into existing production workflows and for optimizing the supply of high-purity intermediates.
Q: What are the primary advantages of this ruthenium-catalyzed method over traditional cyclometallation?
A: Unlike traditional methods that require stoichiometric amounts of expensive and toxic metal oxidants like copper or silver salts, this novel approach utilizes a cheap ruthenium catalyst and simple bases without any external oxidants, significantly reducing waste and production costs.
Q: Can this synthesis method be scaled for industrial production of optoelectronic materials?
A: Yes, the process operates under mild conditions (100°C) in common solvents like toluene and avoids hazardous additives, making it highly suitable for commercial scale-up and compliant with strict environmental regulations for electronic chemical manufacturing.
Q: What types of functional groups are tolerated in the aromatic ketone substrates?
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituents including alkyl groups like methyl and ethyl, as well as electron-withdrawing groups such as fluorine and trifluoromethyl, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyaryl Substituted Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the ruthenium-mediated C-H activation described in CN107935812B for the production of high-value fine chemicals. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Whether you require custom synthesis of complex polyaryl naphthalene derivatives for OLED applications or pharmaceutical intermediates, our infrastructure is designed to meet the most demanding quality and volume requirements of the global market.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green chemistry approach can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to drive innovation and efficiency in your supply chain. Together, we can achieve superior results in the synthesis of next-generation functional materials.
