Advanced Synthesis of Doripenem Side Chain Intermediates for Commercial Scale Production
Advanced Synthesis of Doripenem Side Chain Intermediates for Commercial Scale Production
The global demand for broad-spectrum carbapenem antibiotics continues to rise, driven by the increasing prevalence of resistant bacterial infections. At the heart of manufacturing doripenem, a potent agent developed by Shionogi, lies the efficient production of its critical side chain intermediate. Recent intellectual property developments, specifically patent CN111960984B, disclose a groundbreaking preparation method that fundamentally alters the synthetic landscape for this high-value pharmaceutical building block. This technology introduces a streamlined approach that bypasses traditional bottlenecks, utilizing direct ammonolysis to achieve superior yields and purity profiles. For R&D directors and procurement strategists, understanding this shift is vital for securing a competitive edge in the antibiotic supply chain. The structural complexity of the target molecule requires precise stereochemical control, as illustrated in the core structure below.
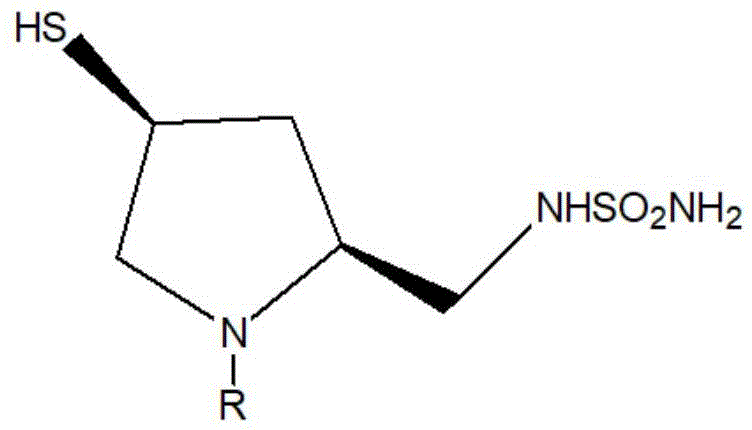
This patent represents a significant leap forward in process chemistry, moving away from atom-inefficient transformations toward greener, more direct functionalization strategies. By leveraging ammonia gas as a dual-purpose reagent, the process not only simplifies the reaction sequence but also drastically reduces the generation of hazardous solid waste associated with older methodologies. As we delve deeper into the technical specifics, it becomes clear that this innovation offers a robust pathway for reliable doripenem side chain intermediate suppliers aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the doripenem side chain has been plagued by inefficiencies inherent in classical organic transformations. Prior art routes, such as the one reported by Shionogi in Organic Process Research & Development (2003), rely heavily on stoichiometric amounts of expensive and hazardous reagents. For instance, the conversion of key precursors often necessitates the use of triphenylphosphine and azodicarboxylates in Mitsunobu-type or reduction-activation sequences. The patent data highlights a staggering material imbalance, where preparing a specific intermediate required nearly equivalent weights of triphenylphosphine relative to the substrate. This results in massive quantities of triphenylphosphine oxide waste, a solid byproduct that is notoriously difficult to remove and poses significant disposal challenges. Furthermore, alternative routes described in European patent EP0528678A1 involve excessive reaction steps, each introducing potential yield losses and impurity carryover. These conventional methods suffer from long production cycles, high energy consumption, and elevated raw material costs, creating a fragile supply chain vulnerable to reagent shortages and regulatory scrutiny regarding waste management.
The Novel Approach
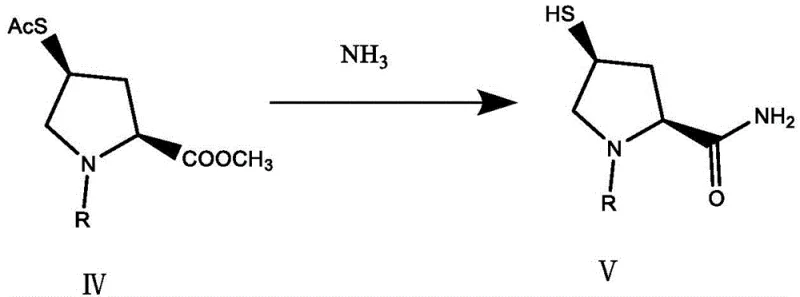
In stark contrast, the methodology outlined in CN111960984B introduces a paradigm shift by employing ammonia gas to effect a simultaneous transformation of multiple functional groups. The core innovation lies in the direct conversion of a thioacetate-ester precursor (Compound IV) into the sulfhydryl-amide intermediate (Compound V) in a single operational step. This eliminates the need for separate reduction and amination stages that characterize older routes. The process utilizes a mixed solvent system, preferably containing isopropanol and dichloromethane, which optimizes the solubility of both the organic substrate and the gaseous ammonia reagent. Crucially, the inclusion of a protective medium, such as tri-n-butylphosphine or an inert gas atmosphere, prevents the oxidation of the newly formed mercapto group into disulfides, a common side reaction that compromises yield. This novel approach not only shortens the production period by reducing the step count but also ensures a cleaner reaction profile with fewer byproducts, directly addressing the cost and quality pain points of traditional manufacturing.
Mechanistic Insights into Ammonia-Mediated Dual Functionalization
The mechanistic elegance of this synthesis lies in the chemoselective reactivity of ammonia gas under controlled conditions. In the transformation of Compound IV to Compound V, ammonia acts as a nucleophile attacking two distinct electrophilic centers: the carbonyl carbon of the ester moiety and the sulfur-bound carbonyl of the thioacetate group. The reaction proceeds through a tetrahedral intermediate at the ester site, leading to the formation of the primary amide, while simultaneously cleaving the thioester bond to liberate the free sulfhydryl group. The presence of the protecting group R on the pyrrolidine nitrogen (such as p-nitrobenzoxycarbonyl or t-butoxycarbonyl) is critical, as it stabilizes the ring system and prevents unwanted N-alkylation or ring-opening side reactions during the ammonolysis. The use of tri-n-butylphosphine serves a dual role: it acts as a scavenger for any trace oxidants that might convert the sensitive thiol to a disulfide, and it may also facilitate the solubilization of ammonia in the organic phase. This dual functionalization strategy is highly atom-economical, as the only byproducts are volatile or easily removable species like methanol and thioacetamide derivatives, avoiding the heavy metal or phosphine oxide contamination seen in other routes.
Furthermore, the subsequent steps involving the reduction of the amide to an amine (Compound V to VI) and the final sulfamoylation demonstrate high fidelity in stereochemical retention. The reduction utilizes borane complexes, which are known for their ability to reduce amides to amines without affecting the carbamate protecting groups or the sulfhydryl functionality when properly managed. The final coupling with sulfamoyl chloride, activated by trimethylchlorosilane, ensures the efficient installation of the sulfonamide tail, which is essential for the biological activity of the final doripenem API. The entire sequence, from the initial hydroxymethyl esterification to the final product, maintains rigorous control over impurity profiles, ensuring that the final intermediate meets the stringent specifications required for GMP manufacturing of beta-lactam antibiotics.
How to Synthesize Doripenem Side Chain Intermediate Efficiently
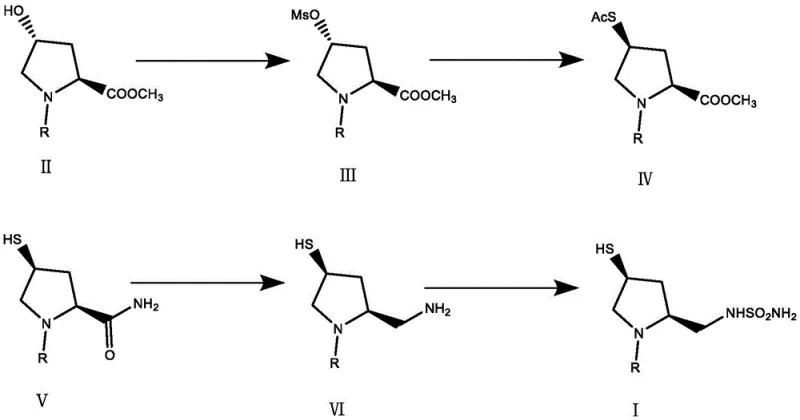
The practical implementation of this synthesis route requires careful attention to reaction conditions, particularly temperature control and reagent addition rates, to maximize yield and safety. The process begins with the activation of the hydroxyl group in Compound II, followed by thioacetylation to install the sulfur handle. The pivotal step involves the introduction of ammonia gas into the reactor containing Compound IV, which must be conducted at low temperatures (preferably around -5°C to 0°C) to minimize side reactions while maintaining sufficient reaction kinetics. The use of a mixed solvent system is essential to ensure homogeneity throughout the gas-liquid reaction. Following the ammonolysis, the workup involves pH adjustment and crystallization, which are designed to be operationally simple and scalable. The subsequent reduction and sulfamoylation steps follow standard protocols but benefit from the high purity of the incoming intermediate. For a detailed breakdown of the specific operational parameters, reagent ratios, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Convert the hydroxyl precursor (Compound II) to a mesylate (Compound III) using methylsulfonyl chloride, followed by nucleophilic substitution with potassium thioacetate (KSAc) to form the thioacetate intermediate (Compound IV).
- Perform a direct one-pot ammonolysis on Compound IV using ammonia gas in the presence of a protective medium like tri-n-butylphosphine to simultaneously generate the sulfhydryl and amide groups, yielding Compound V.
- Reduce the amide group of Compound V to an amine (Compound VI) using a carbonyl reducing agent such as borane, followed by reaction with sulfamoyl chloride to finalize the doripenem side chain intermediate (Compound I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages that go beyond mere technical novelty. The primary value driver is the drastic simplification of the supply chain for raw materials. By eliminating the need for triphenylphosphine, azodicarboxylates, and other specialized coupling agents, manufacturers can source cheaper, commodity-grade chemicals like ammonia and potassium thioacetate. This shift significantly reduces the exposure to price volatility associated with fine chemical reagents and mitigates the risk of supply disruptions. Moreover, the reduction in reaction steps directly correlates to a shorter manufacturing cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations. The simplified purification processes, characterized by straightforward crystallizations rather than complex chromatographic separations, further enhance throughput and reduce facility occupancy time, effectively lowering the cost of goods sold (COGS) without compromising quality.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reagents like triphenylphosphine and the reduction in total processing steps lead to substantial cost savings. The process avoids the generation of hazardous solid waste, which significantly lowers waste disposal costs and environmental compliance fees. Additionally, the use of ammonia gas, a low-cost industrial commodity, replaces pricier amination reagents, driving down the overall raw material expenditure for carbapenem antibiotic manufacturing.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals rather than specialized fine chemicals, the supply chain becomes more resilient. The shortened synthesis timeline reduces the lead time for high-purity doripenem intermediates, ensuring a steady flow of materials for downstream API production. The robustness of the crystallization-based purification steps ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by out-of-specification materials.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with commercial production in mind. The avoidance of hazardous solid byproducts aligns with increasingly strict environmental regulations, facilitating easier permitting and operation in regulated jurisdictions. The mild reaction conditions and use of common solvents simplify the engineering requirements for commercial scale-up of complex pharmaceutical intermediates, allowing for seamless technology transfer from pilot plant to multi-ton production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology resolves historical manufacturing challenges. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this new process for their own production lines or for procurement teams negotiating supply agreements based on these improved specifications.
Q: How does the new ammonia gas method improve upon traditional doripenem side chain synthesis?
A: The novel method described in patent CN111960984B replaces hazardous and waste-generating reagents like triphenylphosphine and azodiisopropyl ester with ammonia gas. This eliminates difficult-to-remove byproducts and dangerous solid waste, significantly simplifying purification and reducing environmental impact.
Q: What are the critical quality control parameters for the intermediate Compound V?
A: Critical parameters include maintaining a reaction temperature between -10°C and 40°C during ammonolysis to prevent side reactions, and ensuring the use of a protective medium like tri-n-butylphosphine to prevent oxidative coupling of the generated mercapto group into disulfides, thereby ensuring high purity (>98%).
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the route is highly scalable. It reduces the total number of reaction steps compared to prior art (such as the Shionogi route), utilizes common industrial solvents like isopropanol and dichloromethane, and avoids expensive transition metal catalysts, making it ideal for cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doripenem Side Chain Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your antibiotic production line depends on the quality and reliability of your starting materials. As a leading CDMO partner, we have closely analyzed the technological advancements presented in patent CN111960984B and possess the technical capability to implement this optimized route immediately. Our facilities are equipped to handle the specific gas-liquid reaction requirements and the stringent temperature controls necessary for this synthesis. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards, guaranteeing that our doripenem side chain intermediates are ready for immediate use in your API synthesis.
We invite you to collaborate with us to leverage these process improvements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this new route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of critical carbapenem intermediates for the global market.
