Revolutionizing PBO Fiber Precursor Production: A Cost-Effective Selenium-Catalyzed Route for DAR
Revolutionizing PBO Fiber Precursor Production: A Cost-Effective Selenium-Catalyzed Route for DAR
The global demand for ultra-high-performance fibers, specifically poly(p-phenylene benzobisoxazole) or PBO, continues to surge across aerospace, military, and advanced industrial sectors due to its unparalleled tensile strength and thermal stability. At the heart of this material supply chain lies a critical intermediate: 4,6-diamino resorcinol dihydrochloride, commonly abbreviated as DAR. As depicted in the structural analysis below, the precise arrangement of amino and hydroxyl groups is essential for the subsequent polymerization into PBO chains. 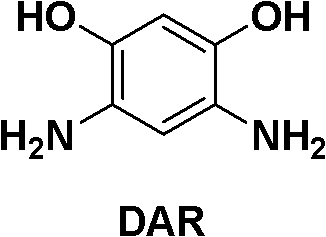 . Recent intellectual property developments, specifically patent CN102173993B, have unveiled a transformative synthetic methodology that addresses long-standing bottlenecks in DAR manufacturing. This patent outlines a robust four-step sequence—nitration, alcoholysis, reduction, and acidification—that not only simplifies the operational workflow but also dramatically enhances the economic feasibility of producing this high-value specialty chemical. For procurement leaders and R&D directors alike, understanding this shift from traditional noble metal catalysis to a selenium-based system represents a pivotal opportunity for supply chain optimization and cost restructuring in advanced material manufacturing.
. Recent intellectual property developments, specifically patent CN102173993B, have unveiled a transformative synthetic methodology that addresses long-standing bottlenecks in DAR manufacturing. This patent outlines a robust four-step sequence—nitration, alcoholysis, reduction, and acidification—that not only simplifies the operational workflow but also dramatically enhances the economic feasibility of producing this high-value specialty chemical. For procurement leaders and R&D directors alike, understanding this shift from traditional noble metal catalysis to a selenium-based system represents a pivotal opportunity for supply chain optimization and cost restructuring in advanced material manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of DAR has been plagued by reliance on expensive and logistically complex catalytic systems. The most critical bottleneck occurs during the reduction of the nitro groups to amino groups, a transformation that traditionally employs precious metal catalysts such as Palladium on Carbon (Pd/C), Platinum on Carbon (Pt/C), or Ruthenium on Carbon (Ru/C). While effective, these noble metal systems introduce severe economic and operational liabilities. Firstly, the capital expenditure required to procure and maintain inventory of palladium or platinum is substantial, directly inflating the raw material costs of the final API or intermediate. Secondly, these metal catalysts are prone to deactivation by chloride ions present in the substrate, often necessitating the addition of expensive dechlorination promoters like ammonium acetate or ammonium formiate to prevent the loss of chlorine atoms from the aromatic ring. This adds further complexity to the reaction mixture and increases the burden on downstream purification. Moreover, the recovery of these precious metals from the reaction mass is technically demanding, often requiring specialized filtration and refining processes to prevent metal contamination in the final product, which is strictly regulated for high-performance polymer applications.
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in patent CN102173993B introduces a paradigm shift by utilizing elemental selenium as the primary catalyst in conjunction with carbon monoxide (CO) and water. This selenium-catalyzed reduction system elegantly bypasses the need for costly noble metals while maintaining exceptional selectivity. As illustrated in the comprehensive reaction scheme below, the process flows seamlessly from meta-dichlorobenzene through nitration and alcoholysis before hitting the critical reduction step. 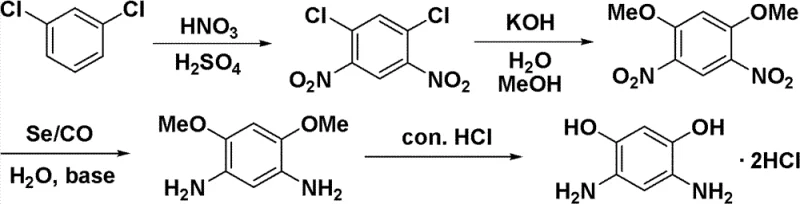 . The innovation here lies not just in the substitution of the catalyst, but in the systemic efficiency it enables. The selenium catalyst demonstrates remarkable stability and selectivity, reportedly achieving conversion rates nearing quantification with selectivity exceeding 99%. Crucially, the post-reaction processing is vastly simplified; the solid selenium catalyst can be recovered through basic filtration and recycled for subsequent batches without significant loss of activity. This eliminates the need for complex metal scavenging resins or intricate refining protocols, thereby streamlining the entire production timeline and reducing the environmental footprint associated with heavy metal waste disposal.
. The innovation here lies not just in the substitution of the catalyst, but in the systemic efficiency it enables. The selenium catalyst demonstrates remarkable stability and selectivity, reportedly achieving conversion rates nearing quantification with selectivity exceeding 99%. Crucially, the post-reaction processing is vastly simplified; the solid selenium catalyst can be recovered through basic filtration and recycled for subsequent batches without significant loss of activity. This eliminates the need for complex metal scavenging resins or intricate refining protocols, thereby streamlining the entire production timeline and reducing the environmental footprint associated with heavy metal waste disposal.
Mechanistic Insights into Selenium-Catalyzed Nitro Reduction
To fully appreciate the technical superiority of this route, one must delve into the mechanistic nuances of the selenium-carbon monoxide reduction system. Unlike heterogeneous hydrogenation which relies on surface adsorption of hydrogen gas, this homogeneous or quasi-homogeneous system utilizes CO as the reducing agent in an aqueous alkaline medium. The selenium acts as a carrier for oxygen transfer, effectively shuttling oxygen from the nitro group to the carbon monoxide to form carbon dioxide, while the nitro group is reduced to the amine. This mechanism is particularly advantageous for chlorinated substrates because it operates under milder conditions compared to high-pressure hydrogenation, thereby minimizing the risk of hydrodechlorination—a common side reaction where the valuable chlorine atoms are stripped from the ring, leading to unwanted by-products and yield loss. The presence of water and base facilitates the generation of active selenium species that are highly selective for the nitro functionality, leaving the methoxy and chloro substituents intact until the specific deprotection stages.
Furthermore, the impurity profile generated by this method is significantly cleaner than that of metal-catalyzed routes. In traditional Pd/C reductions, trace metal residues can act as pro-oxidants or degradation catalysts in the final polymer, potentially compromising the thermal stability of the resulting PBO fiber. The selenium system, being non-metallic in the traditional sense and easily removable, ensures that the final DAR product meets stringent purity specifications, often exceeding 99.9% as confirmed by HPLC analysis in the patent examples. The ability to control the reaction pressure (1-8 MPa) and temperature (60-200°C) provides a wide operating window for process engineers to optimize kinetics without sacrificing selectivity. This robustness is essential for scaling up from laboratory glassware to multi-ton industrial reactors, ensuring that the quality of the intermediate remains consistent regardless of batch size.
Visualizing the Critical Reduction Step
The efficacy of the selenium catalyst is best understood by examining the specific transformation of 1,5-dimethoxy-2,4-dinitrobenzene to the corresponding diamine. 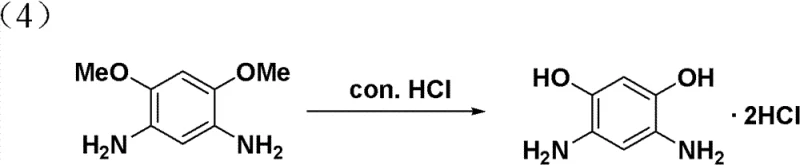 . This step represents the technological core of the patent, where the expensive and difficult nitro-reduction is achieved with near-quantitative yield. The image highlights the conversion of the nitro groups (-NO2) to amino groups (-NH2) while preserving the methoxy groups (-OMe), which are subsequently cleaved in the final acidification step. The visual clarity of this transformation underscores the chemoselectivity of the selenium system, proving its capability to handle multifunctional aromatic substrates without requiring extensive protecting group strategies.
. This step represents the technological core of the patent, where the expensive and difficult nitro-reduction is achieved with near-quantitative yield. The image highlights the conversion of the nitro groups (-NO2) to amino groups (-NH2) while preserving the methoxy groups (-OMe), which are subsequently cleaved in the final acidification step. The visual clarity of this transformation underscores the chemoselectivity of the selenium system, proving its capability to handle multifunctional aromatic substrates without requiring extensive protecting group strategies.
How to Synthesize 4,6-Diamino Resorcinol Dihydrochloride Efficiently
Implementing this synthesis requires strict adherence to the four-stage protocol outlined in the patent data, beginning with the careful nitration of meta-dichlorobenzene. The process demands precise temperature control during the exothermic nitration phase to ensure the formation of the correct 4,6-isomer, followed by a nucleophilic substitution with alkoxide to install the methoxy groups. The subsequent reduction step requires an autoclave capable of handling carbon monoxide pressure, a standard piece of equipment in modern fine chemical facilities. Finally, the demethylation and salt formation are achieved through reflux in concentrated hydrochloric acid. For a detailed, step-by-step breakdown of the exact reagent quantities, reaction times, and workup procedures necessary to replicate this high-yield process, please refer to the standardized guide below.
- Perform nitration of meta-dichlorobenzene using a mixed acid system of sulfuric and nitric acid to obtain 4,6-dinitro-meta-dichlorobenzene.
- Conduct alcoholysis of the dinitro compound in the presence of potassium hydroxide and an alcoholic solvent to yield 1,5-dimethoxy-2,4-dinitrobenzene.
- Execute the critical reduction step using selenium as a catalyst with carbon monoxide and water to convert nitro groups to amino groups.
- Finalize the synthesis by reacting the dimethoxy diamine with concentrated hydrochloric acid to form the target DAR dihydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this selenium-catalyzed route offers compelling strategic advantages that extend far beyond simple chemistry. The primary value driver is the drastic reduction in raw material costs associated with eliminating precious metals. By replacing palladium or platinum with selenium, a commodity chemical with a fraction of the cost, manufacturers can significantly lower the Cost of Goods Sold (COGS) for DAR. This cost efficiency is compounded by the operational savings gained from the simplified catalyst recovery process; the ability to filter and reuse the catalyst multiple times reduces both material consumption and waste disposal fees. Furthermore, the high selectivity of the reaction minimizes the formation of isomers and by-products, which translates to higher overall yields and less material lost to purification streams, effectively increasing the throughput of existing production assets without the need for capital expansion.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts removes a major variable cost component from the production budget. Unlike palladium, which fluctuates wildly in price and requires secure storage and insurance, selenium is stable and inexpensive. Additionally, the removal of dechlorination promoters further strips away unnecessary reagent costs. The high yield of the reduction step, reported as nearly quantitative, ensures that expensive intermediates are not wasted, maximizing the return on investment for every kilogram of starting material processed. This lean manufacturing approach allows for competitive pricing in the global market for PBO precursors.
- Enhanced Supply Chain Reliability: Relying on precious metal catalysts often exposes supply chains to geopolitical risks and mining bottlenecks. By shifting to a selenium-based system, manufacturers diversify their supply risk, sourcing from a more abundant and stable pool of raw materials. The robustness of the reaction conditions also means that production is less susceptible to minor variations in feedstock quality, ensuring consistent output even when supply chains are stressed. The simplified purification process reduces the dependency on specialized chromatography resins or complex extraction solvents, streamlining the logistics of raw material procurement and inventory management.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior. The reduction of heavy metal waste aligns with increasingly stringent global environmental regulations, reducing the liability and cost associated with hazardous waste treatment. The process generates fewer by-products, leading to a cleaner effluent stream that is easier and cheaper to treat. The scalability is proven by the use of standard unit operations like nitration reactors and autoclaves, making the transition from pilot scale to commercial production seamless. This ensures that supply can be ramped up quickly to meet surging demand for high-performance fibers without encountering technical barriers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of DAR using this novel methodology. These insights are derived directly from the experimental data and claims presented in patent CN102173993B, providing a factual basis for evaluating the technology's viability. Understanding these details is crucial for technical teams assessing the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is selenium preferred over palladium catalysts for DAR synthesis?
A: Selenium is significantly cheaper than precious metals like palladium or platinum. Furthermore, unlike noble metals which often require complex recovery processes or dechlorination promoters, selenium can be easily recovered via simple filtration and reused multiple times without losing catalytic efficiency, drastically lowering operational costs.
Q: What purity levels can be achieved with this synthetic route?
A: The patented method achieves exceptional purity, with the final 4,6-diamino resorcinol product reaching up to 99.9% purity. The selectivity of the key reduction step is reported to be as high as 99%, minimizing the formation of isomers and by-products that typically complicate downstream polymerization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes standard reactor equipment (autoclaves for the reduction step) and avoids the environmental burdens associated with heavy metal waste. The simplicity of the catalyst recovery and the high yield of each step make it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-Diamino Resorcinol Dihydrochloride Supplier
The technological advancements described in patent CN102173993B highlight the immense potential for optimizing the production of critical polymer intermediates like DAR. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, commercial-scale realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the selenium-catalyzed route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of DAR meets the exacting standards required for PBO fiber synthesis.
We invite forward-thinking partners to collaborate with us to leverage this cost-effective synthesis route for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss specific COA data, route feasibility assessments, and how we can support your long-term goals in the advanced materials sector with reliable, high-quality chemical solutions.
