Advanced Synthesis of Allene Thiocyanide Derivatives for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex sulfur-containing scaffolds, particularly allene thiocyanide derivatives, which serve as pivotal building blocks for bioactive molecules. A significant breakthrough in this domain is documented in patent CN108424343B, which discloses a novel, efficient, and inherently safer synthetic route for these valuable compounds. Unlike traditional approaches that rely on hazardous gaseous reagents, this innovation utilizes silver trifluoromethyl sulfide (AgSCF3) in conjunction with halogen salts to drive a two-step tandem reaction at ambient temperature. This technological advancement addresses critical pain points regarding operator safety, environmental compliance, and process efficiency, positioning it as a superior choice for the manufacturing of high-purity pharmaceutical intermediates. The method demonstrates exceptional chemical selectivity and broad substrate tolerance, accommodating various substituted phenyl and alkyl groups, thereby offering a versatile platform for medicinal chemistry applications.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allene thiocyanides and related sulfur-containing heterocycles has been plagued by significant safety and operational challenges. Conventional protocols frequently depend on thiophosgene or similar highly toxic, volatile sulfur sources that pose severe health risks and require rigorous engineering controls to prevent leakage and exposure. These traditional methods often necessitate multi-step sequences where unstable intermediates must be isolated and purified under stringent conditions, leading to increased processing time and material loss. Furthermore, the use of cryogenic temperatures or strong bases in older methodologies limits the functional group compatibility, often resulting in side reactions or decomposition of sensitive substrates. The cumulative effect of these factors is a manufacturing process that is not only expensive due to safety infrastructure requirements but also suffers from inconsistent yields and poor scalability, making it ill-suited for the demanding supply chains of modern pharmaceutical production.
The Novel Approach
The methodology outlined in patent CN108424343B represents a paradigm shift by replacing hazardous gaseous reagents with stable, easy-to-handle solid precursors. By employing silver trifluoromethyl sulfide and a simple halogen salt additive, the reaction generates the necessary reactive species in situ, effectively bypassing the need to store or transport dangerous chemicals. This tandem reaction proceeds smoothly at room temperature, eliminating the energy costs associated with heating or cooling and significantly simplifying the reactor setup. The process exhibits high atom economy and step efficiency, as the transformation from propargylamine to the final allene thiocyanide occurs in a single pot without the isolation of intermediates. This streamlined approach not only enhances the overall yield but also drastically reduces the generation of chemical waste, aligning perfectly with green chemistry principles and the sustainability goals of forward-thinking chemical enterprises.
Mechanistic Insights into AgSCF3-Mediated Tandem Reaction
The elegance of this synthesis lies in its intricate yet efficient mechanistic pathway, which leverages the unique reactivity of silver trifluoromethyl sulfide in the presence of halide ions. The reaction initiates with a metathesis between AgSCF3 and the halogen salt, such as potassium bromide, resulting in the precipitation of silver bromide and the formation of soluble potassium trifluoromethyl sulfide. This soluble species subsequently undergoes decomposition to generate thiofluorophosgene, a potent electrophile, directly within the reaction medium. The immediate capture of this transient species by the propargylamine substrate prevents its accumulation and potential side reactions, ensuring high chemoselectivity. Following the initial nucleophilic attack, the resulting propargyl isothiocyanate intermediate undergoes a spontaneous [3,3]-sigmatropic rearrangement. This pericyclic process drives the structural reorganization required to form the characteristic allene moiety,最终 yielding the stable allene thiocyanide derivative with high fidelity.
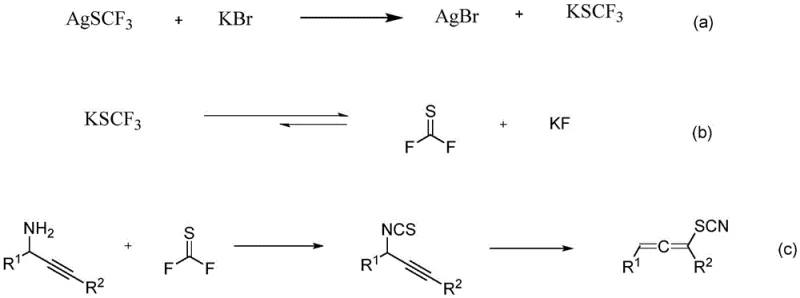
Understanding the impurity profile is crucial for R&D directors aiming for regulatory compliance, and this mechanism offers inherent advantages in that regard. Because the reactive thiofluorophosgene is generated in situ and consumed immediately, the concentration of free toxic species remains negligible, minimizing the formation of oligomeric byproducts often seen in batch additions of thiophosgene. Furthermore, the mild reaction conditions preserve sensitive functional groups on the aromatic rings or alkyl chains, preventing degradation or unwanted substitution that could complicate downstream purification. The precipitation of silver bromide acts as a driving force for the initial metathesis step and can be easily removed during workup, contributing to a cleaner crude product profile. This level of control over the reaction trajectory ensures that the final API intermediate meets stringent purity specifications with minimal effort, reducing the burden on analytical quality control teams.
How to Synthesize Allene Thiocyanide Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize yield and reproducibility. The protocol involves dissolving the halogen salt and silver trifluoromethyl sulfide in a polar aprotic solvent such as acetonitrile, followed by the controlled addition of the propargylamine substrate. The mixture is then stirred at room temperature for a defined period to allow the tandem sequence to reach completion. Detailed standardized operating procedures, including precise molar ratios and workup techniques, are essential for consistent results. For a comprehensive guide on the exact experimental steps and purification methods, please refer to the structured synthesis guide below.
- Mix silver trifluoromethyl sulfide (AgSCF3) and a halogen salt additive (such as KBr) in an organic solvent like acetonitrile.
- Add the propargylamine substrate to the reaction mixture and stir at room temperature to initiate the tandem reaction sequence.
- Upon completion, remove the solvent under reduced pressure and purify the crude allene thiocyanide derivative via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthesis route offers transformative benefits that extend beyond mere technical feasibility. The shift away from hazardous gaseous reagents to stable solids fundamentally alters the risk profile of the manufacturing process, leading to substantial reductions in insurance premiums and safety compliance costs. The ability to operate at room temperature significantly lowers energy consumption, contributing to a smaller carbon footprint and reduced utility expenses. Moreover, the use of commercially available and shelf-stable reagents ensures a reliable supply chain, mitigating the risks associated with the sourcing of specialized or regulated chemicals. This robustness translates into shorter lead times and greater flexibility in production scheduling, allowing manufacturers to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous thiophosgene removes the need for specialized gas handling infrastructure and scrubbing systems, resulting in significant capital expenditure savings. Additionally, the one-pot tandem nature of the reaction reduces labor costs and solvent usage by removing intermediate isolation steps. The high yields reported across a broad range of substrates mean less raw material is wasted, further driving down the cost of goods sold. These cumulative efficiencies make the production of allene thiocyanide derivatives economically viable even at smaller scales, opening up new opportunities for niche pharmaceutical projects.
- Enhanced Supply Chain Reliability: Sourcing stable solid reagents like AgSCF3 and common halide salts is far more predictable than managing the logistics of toxic gases, which often face strict transportation regulations and supply bottlenecks. This stability ensures continuous production capability without the fear of sudden raw material shortages. The simplified process flow also reduces the number of potential failure points in the manufacturing line, enhancing overall equipment effectiveness. Consequently, supply chain managers can forecast delivery timelines with greater accuracy, strengthening relationships with downstream clients who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The mild conditions and absence of volatile toxic emissions make this process inherently scalable from gram to ton quantities without requiring major process re-engineering. The reduced generation of hazardous waste simplifies disposal protocols and lowers environmental compliance costs. As regulatory pressures on chemical manufacturing intensify globally, adopting such green and safe methodologies future-proofs the production facility against stricter environmental laws. This proactive approach to sustainability not only safeguards the license to operate but also enhances the corporate brand reputation among environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of allene thiocyanide derivatives, based on the detailed disclosures within the patent literature. These insights are designed to clarify the operational benefits and mechanistic underpinnings of this advanced chemical transformation. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is this new synthesis method safer than conventional thiophosgene routes?
A: Conventional methods often require thiophosgene, a highly toxic and volatile gas that demands specialized containment equipment. This patented method generates the reactive species in situ from stable solid reagents (AgSCF3 and halide salts) at room temperature, drastically reducing operational hazards.
Q: What represents the key mechanistic advantage of using AgSCF3 in this reaction?
A: The use of AgSCF3 combined with a halide salt allows for the controlled, in situ generation of thiofluorophosgene. This reactive intermediate immediately engages with the propargylamine substrate, facilitating a seamless [3,3]-sigmatropic rearrangement to form the allene thiocyanide structure without isolating unstable intermediates.
Q: Is this synthesis protocol scalable for industrial production?
A: Yes, the protocol operates under mild conditions (room temperature) and utilizes commercially available, stable reagents. The elimination of cryogenic conditions and toxic gas handling makes it highly suitable for commercial scale-up in standard pharmaceutical manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allene Thiocyanide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates through safe and efficient synthetic routes. Our team of expert chemists has thoroughly analyzed the technology described in patent CN108424343B and is fully equipped to leverage this methodology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain for allene thiocyanide derivatives and other fine chemical intermediates. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate how our capabilities can accelerate your drug development timeline while reducing overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
