Advanced Copper-Catalyzed Synthesis of Benzophosphaindole Derivatives for High-Performance Optoelectronic Materials
Advanced Copper-Catalyzed Synthesis of Benzophosphaindole Derivatives for High-Performance Optoelectronic Materials
The rapid evolution of the organic electronics sector demands increasingly sophisticated heterocyclic building blocks that offer superior charge transport properties and thermal stability. Patent CN110577556B introduces a groundbreaking methodology for the preparation of benzophosphaindole derivatives, a class of compounds critical for next-generation optoelectronic applications and homogeneous catalysis. This intellectual property details a robust synthetic pathway that leverages a copper-catalyzed radical cyclization strategy, effectively transforming simple alkynes and phosphorus reagents into complex phosphorus-containing heterocycles under remarkably mild conditions. By operating within a temperature window of 50-100°C and utilizing commercially accessible oxidants, this technology addresses the longstanding challenges of harsh reaction environments and low atom economy associated with traditional phosphole synthesis. For R&D leaders seeking to integrate high-purity phosphorus heterocycles into their material pipelines, this patent represents a significant leap forward in process efficiency and structural versatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of phosphorus-containing heterocyclic frameworks, such as phospholanes and benzo[b]phospholes, has been plagued by severe synthetic bottlenecks that hinder large-scale adoption. Traditional routes often rely on the utilization of difficult-to-obtain precursors that require multi-step preparation, thereby inflating the overall cost of goods and extending lead times for material development. Furthermore, classical methodologies frequently necessitate the use of strong bases or harsh dehydrating agents to drive cyclization, which can lead to poor functional group tolerance and the generation of complex impurity profiles that are difficult to separate. In the context of synthesizing specific catalysts like 5-phenylbenzo[b]phosphaindole-5-oxide, prior art describes processes where raw materials are scarce and reaction conditions are excessively rigorous, resulting in suboptimal yields that are economically unviable for commercial manufacturing. These legacy methods also struggle with the synthesis of conjugated systems required for photoelectric materials, often failing to provide the necessary structural precision without compromising throughput.
The Novel Approach
In stark contrast to these archaic techniques, the novel approach disclosed in the patent utilizes a direct oxidative cyclization strategy that streamlines the formation of the phosphaindole core with exceptional efficiency. By employing a copper catalyst in conjunction with an organic peroxide, the method facilitates the activation of the phosphorus-hydrogen bond and subsequent addition across the alkyne triple bond at moderate temperatures ranging from 50-100°C. This transition metal-catalyzed pathway not only simplifies the operational procedure by eliminating the need for stringent anhydrous conditions or cryogenic temperatures but also significantly expands the scope of compatible substrates to include various alkyl, aryl, and heteroaryl alkynes. The process allows for the direct synthesis of phosphaindole derivatives which can be subsequently derivatized into benzophosphaindole structures through reaction with 2,5-dimethoxytetrahydrofuran, offering a modular platform for material design. 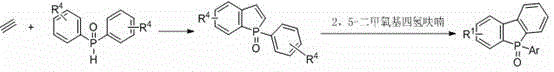
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the intricate interplay between the copper catalyst and the organic peroxide initiator, which drives the formation of the phosphorus-carbon bonds essential for the heterocyclic ring closure. The reaction mechanism likely proceeds through a radical pathway where the copper species mediates the homolytic cleavage of the peroxide, generating reactive radicals that abstract hydrogen from the phosphine oxide reagent. This generates a phosphinoyl radical species that adds regioselectively to the alkyne substrate, followed by an intramolecular cyclization event that constructs the five-membered phosphole ring fused to the aromatic system. The use of copper halides, such as CuCl or CuBr, ensures that the catalytic cycle is sustained efficiently, minimizing the loading of expensive metal while maximizing turnover numbers. 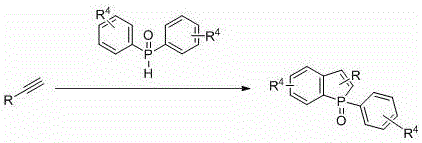
From an impurity control perspective, the mild thermal conditions and the specific choice of oxidants play a pivotal role in maintaining a clean reaction profile. Unlike strong base-mediated condensations that often produce polymeric byproducts or isomeric mixtures, this radical-mediated cyclization exhibits high chemoselectivity, primarily yielding the desired 1-oxide phosphaindole structure. The ability to tune the reaction outcome by selecting specific organic peroxides, such as tert-butyl peroxybenzoate or di-tert-butyl peroxide, allows process chemists to optimize the kinetics of radical generation, thereby suppressing side reactions like alkyne polymerization or over-oxidation. This precise control over the reaction trajectory ensures that the final crude product requires minimal purification effort, typically achievable through standard column chromatography, which is a critical factor for maintaining high purity specifications in electronic grade materials.
How to Synthesize Benzophosphaindole Derivatives Efficiently
The practical implementation of this synthesis route involves a straightforward protocol that begins with the dissolution of the alkyne substrate and phosphorus reagent in a polar protic or aprotic solvent such as methanol or acetonitrile. Following the addition of the copper catalyst and the organic peroxide oxidant, the mixture is heated to the specified temperature range and monitored via thin-layer chromatography to ensure complete consumption of the starting materials. Once the initial phosphaindole intermediate is formed and isolated, it can be further reacted with 2,5-dimethoxytetrahydrofuran in the presence of a Lewis acid catalyst like ZnBr2 to generate the fused benzophosphaindole scaffold. The detailed standardized synthesis steps, including specific molar ratios and workup procedures derived from the patent examples, are outlined below to facilitate immediate technology transfer.
- Dissolve alkyne, phosphorus reagent, copper catalyst, and organic peroxide in a suitable solvent such as methanol or acetonitrile.
- Heat the reaction mixture to a temperature range of 50-100°C and maintain until TLC indicates complete conversion to the phosphaindole intermediate.
- Purify the crude product via column chromatography, optionally reacting the intermediate with 2,5-dimethoxytetrahydrofuran for further functionalization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology offers profound strategic benefits that extend far beyond simple laboratory curiosity. The shift towards using commodity chemicals like acetylene, phenylacetylene, and diphenylphosphine oxide as starting materials drastically reduces the dependency on bespoke, high-cost intermediates that often bottleneck production schedules. By simplifying the synthetic sequence and removing the need for exotic reagents or extreme reaction conditions, manufacturers can achieve substantial cost savings in raw material acquisition and inventory management. Furthermore, the operational simplicity of the process translates directly into reduced utility costs, as the reaction does not require energy-intensive heating or cooling cycles, nor does it demand specialized high-pressure reactor vessels that increase capital expenditure.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts like palladium in the initial cyclization step, replacing them with abundant copper salts, results in a significant decrease in catalyst costs per kilogram of product. Additionally, the high yields reported in the patent examples, often exceeding 80%, mean that less raw material is wasted, directly improving the overall mass balance and reducing the cost of waste disposal. The simplified post-treatment process, which relies on standard extraction and chromatography rather than complex distillation or recrystallization sequences, further lowers the labor and processing time required per batch.
- Enhanced Supply Chain Reliability: Since the key reagents such as alkynes and diarylphosphine oxides are widely available from multiple global suppliers, the risk of supply disruption due to single-source dependency is effectively mitigated. The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is essential for maintaining long-term supply contracts with downstream electronics manufacturers. This reliability allows supply chain planners to forecast production timelines with greater accuracy, reducing the need for excessive safety stock and freeing up working capital.
- Scalability and Environmental Compliance: The use of common organic solvents like ethanol, methanol, and acetonitrile facilitates easy solvent recovery and recycling, aligning the process with modern green chemistry principles and environmental regulations. The absence of heavy metal contaminants in the final product, owing to the ease of removing copper residues compared to noble metals, simplifies the quality control workflow and ensures compliance with strict purity standards for electronic materials. This scalability ensures that the transition from gram-scale laboratory optimization to ton-scale commercial production can be executed smoothly without encountering unforeseen engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis technology for industrial applications. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for process development teams. Understanding these nuances is crucial for successfully adapting the laboratory protocol to a manufacturing environment.
Q: What are the primary advantages of this copper-catalyzed method over traditional synthesis routes?
A: This method utilizes readily available alkynes and mild reaction conditions (50-100°C), avoiding the harsh bases and difficult-to-obtain raw materials required by conventional Wittig or Appel-type reactions, resulting in significantly higher yields.
Q: Can this synthesis protocol be scaled for industrial production of optoelectronic materials?
A: Yes, the process employs common solvents like methanol and acetonitrile and operates at moderate temperatures, making it highly amenable to scale-up without requiring specialized high-pressure or cryogenic equipment.
Q: What types of alkynes are compatible with this phosphorus heterocycle formation?
A: The protocol demonstrates broad substrate scope, successfully accommodating alkyl alkynes, aryl alkynes, and various heteroaryl alkynes, allowing for diverse functionalization of the final benzophosphaindole core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzophosphaindole Derivative Supplier
As the global demand for high-performance organic semiconductors and specialized catalysts continues to surge, having a manufacturing partner with deep expertise in phosphorus heterocycle chemistry is indispensable. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging advanced synthetic capabilities to deliver complex molecules with unmatched consistency and quality. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of even the most demanding multinational corporations. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to unlock the full potential of this innovative copper-catalyzed technology for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project, demonstrating how our optimized processes can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your time to market with reliable, high-quality benzophosphaindole derivatives.
