Advanced Electrochemical Manufacturing of High-Purity Sulfonyl Nitrogen Heterocycles for Global Pharma
Advanced Electrochemical Manufacturing of High-Purity Sulfonyl Nitrogen Heterocycles for Global Pharma
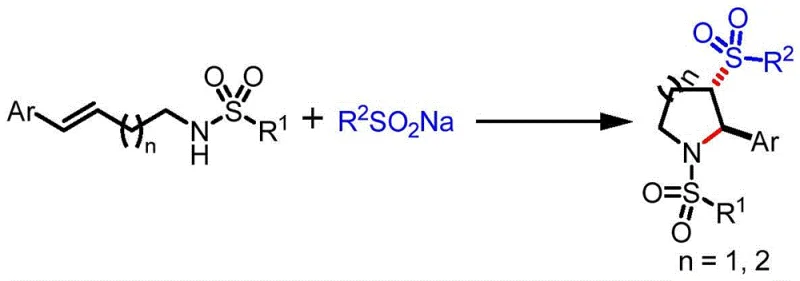
The pharmaceutical and agrochemical industries are constantly seeking more sustainable and efficient pathways to construct complex nitrogen-containing heterocycles, which serve as critical scaffolds for bioactive molecules. A groundbreaking development in this sector is detailed in Chinese Patent CN116356354A, which discloses a novel preparation method for 2-aryl-3-sulfonyl substituted tetrahydropyrrole and piperidine compounds. This technology leverages organic electrosynthesis to achieve a one-pot transformation of unsaturated amines and sodium sulfinates, bypassing the need for harsh chemical oxidants or precious metal catalysts. For R&D directors and procurement specialists, this represents a paradigm shift towards greener chemistry that does not compromise on yield or selectivity. The method utilizes electricity as a clean reagent to drive the oxidative cyclization, offering a distinct advantage in terms of atom economy and environmental compliance. By integrating this electrochemical strategy, manufacturers can access high-value intermediates with reduced operational complexity and enhanced safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 2-aryl-3-sulfonyl substituted piperidine and pyrrole frameworks has relied on synthetic routes that are fraught with significant operational and economic challenges. Traditional approaches often involve (3+2) cycloaddition reactions between aryl vinyl sulfones and 1,3-dipolar reagents, which necessitate the preparation of complex starting materials that are not readily available on a commercial scale. Alternatively, other established methods utilize Grignard reagents in multi-step sequences involving sulfonation and subsequent cyclization. These conventional pathways are inherently problematic because they require the handling of highly reactive and moisture-sensitive organometallic species, posing severe safety risks in large-scale manufacturing environments. Furthermore, the reliance on stoichiometric chemical oxidants generates substantial amounts of toxic waste, creating a heavy burden on wastewater treatment facilities and increasing the overall environmental footprint of the production process. The multi-step nature of these legacy syntheses also leads to cumulative yield losses and extended production timelines, which are unacceptable in the fast-paced landscape of modern drug development.
The Novel Approach
In stark contrast to these cumbersome traditional methods, the electrochemical protocol described in the patent offers a streamlined, one-pot solution that fundamentally simplifies the synthetic logic. By employing an undivided electrolytic cell equipped with a carbon anode and a platinum cathode, the reaction directly couples unsaturated amines with sodium sulfinates under constant current electrolysis. This innovative approach eliminates the requirement for external chemical oxidants, as the anodic oxidation generates the necessary sulfonyl radicals in situ from the inexpensive sulfinate salts. The reaction proceeds under remarkably mild conditions, typically at room temperature and ambient pressure, which drastically reduces energy consumption compared to thermal processes requiring high heat. Moreover, the absence of transition metal catalysts means that the final product is free from heavy metal contamination, a critical quality attribute for pharmaceutical intermediates intended for human consumption. This direct electrified synthesis not only shortens the reaction timeline but also enhances the overall process safety by avoiding hazardous reagents.
Mechanistic Insights into Anodic Oxidative Cyclization
The core of this technological breakthrough lies in the precise control of electron transfer at the electrode surface, which initiates a radical cascade leading to the desired heterocyclic products. Mechanistically, the process begins with the anodic oxidation of the sodium sulfinate salt, which loses an electron to form a sulfonyl radical species. This highly reactive radical then undergoes a regioselective addition to the carbon-carbon double bond of the unsaturated amine substrate, generating a carbon-centered radical intermediate. Subsequently, an intramolecular cyclization occurs where the nitrogen lone pair attacks the radical center, closing the ring to form either a five-membered tetrahydropyrrole or a six-membered piperidine structure, depending on the chain length of the starting amine. The final step involves the loss of a proton and potentially another electron to restore aromaticity or stability, completing the catalytic cycle without the need for sacrificial reagents. Understanding this mechanism is crucial for R&D teams as it highlights the tunability of the reaction; by adjusting the current density or electrode potential, chemists can fine-tune the radical generation rate to optimize selectivity and minimize side reactions.
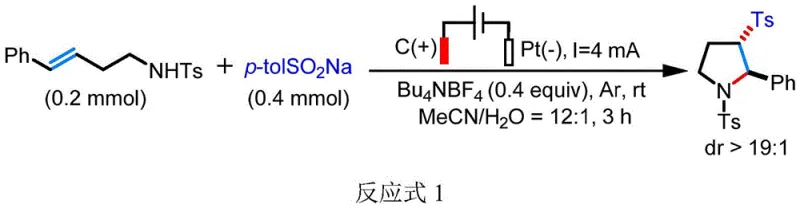
Furthermore, the electrochemical environment provides a unique medium for controlling stereochemistry, which is often a challenge in free-radical reactions. The patent data indicates that this method achieves exceptional diastereoselectivity, with dr values exceeding 19:1 in many examples, suggesting that the cyclization transition state is highly organized, possibly due to coordination with the electrolyte or solvent effects. The use of tetrabutylammonium tetrafluoroborate as the supporting electrolyte plays a dual role: it ensures sufficient conductivity for the electric current while potentially stabilizing the charged intermediates through ion-pairing interactions. This level of control over the impurity profile is vital for regulatory compliance, as it minimizes the formation of difficult-to-remove stereoisomers. For process chemists, this mechanistic clarity offers a robust platform for scaling up, as the reaction parameters (current, time, electrode area) are easily measurable and controllable variables that translate well from laboratory benchtop to industrial pilot plants.
How to Synthesize 2-Aryl-3-Sulfonyl Tetrahydropyrroles Efficiently
Implementing this electrochemical synthesis requires careful attention to the setup of the electrolytic cell and the selection of reaction parameters to ensure reproducibility and high yield. The protocol dictates a specific molar ratio of substrates to electrolyte, typically favoring a slight excess of the sulfinate reagent to drive the reaction to completion while maintaining efficient current usage. The choice of solvent system, specifically a mixture of acetonitrile and water, is critical for solubilizing both the organic substrates and the inorganic electrolyte while facilitating the proton transfer steps. Operators must ensure that the electrode materials, preferably a carbon rod anode and a platinum sheet cathode, are properly positioned to maintain a consistent electric field throughout the reaction volume. While the general procedure is straightforward, strict adherence to the inert atmosphere (argon protection) is necessary to prevent the quenching of radical intermediates by oxygen, which could lead to oxidative byproducts. For a detailed, step-by-step operational guide including specific quantities and workup procedures, please refer to the standardized synthesis instructions below.
- Prepare the electrolytic cell by adding unsaturated amine substrate, aryl or alkyl sodium sulfinate, and tetrabutylammonium tetrafluoroborate electrolyte into a mixture of acetonitrile and water.
- Insert carbon rod anode and platinum sheet cathode, then purge the system with argon gas to establish an inert atmosphere.
- Apply a constant current of 4 mA at room temperature for 3 hours, followed by standard extraction and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrochemical methodology offers profound advantages that directly address the pain points of procurement managers and supply chain directors. The most significant benefit is the drastic simplification of the raw material supply chain; sodium sulfinates and unsaturated amines are commodity chemicals that are widely available and inexpensive compared to the specialized vinyl sulfones or Grignard reagents required by older methods. This shift to abundant feedstocks mitigates the risk of supply disruptions and price volatility, ensuring a more stable and predictable cost structure for long-term manufacturing contracts. Additionally, the elimination of transition metal catalysts removes the need for costly metal scavenging resins and the associated validation testing for residual metals, which translates into tangible savings in both material costs and quality control labor. The simplified workflow also means shorter batch cycles, allowing manufacturing facilities to increase throughput and respond more rapidly to market demand fluctuations without the need for significant capital investment in new equipment.
- Cost Reduction in Manufacturing: The economic impact of replacing chemical oxidants with electricity cannot be overstated, as it removes the cost of purchasing, storing, and disposing of hazardous oxidizing agents. By utilizing electrons as the primary oxidant, the process inherently reduces the mass intensity of the reaction, leading to less waste generation and lower disposal fees. Furthermore, the one-pot nature of the synthesis eliminates intermediate isolation steps, which saves on solvent usage, filtration media, and drying time. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), making the final API intermediates more competitive in the global marketplace. The avoidance of cryogenic conditions or high-pressure reactors also reduces energy overheads, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the electrochemical process, which tolerates a wide range of functional groups on the aromatic rings without requiring protective group strategies. This versatility means that a single manufacturing line can be adapted to produce a diverse library of analogues by simply swapping the starting amine or sulfinate, providing agility in portfolio management. The use of standard electrolytic cells and common electrode materials like carbon and platinum ensures that equipment sourcing is not a bottleneck, unlike specialized reactors required for high-pressure hydrogenation or cryogenic lithiation. Consequently, lead times for custom synthesis projects can be significantly compressed, enabling faster delivery of clinical trial materials and commercial batches to clients.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is increasingly well-understood, with modern flow electrolysis cells offering linear scalability from gram to tonne scales. This technology aligns perfectly with global sustainability goals by minimizing the E-factor (mass of waste per mass of product), as the primary byproduct is often just hydrogen gas at the cathode. The reduction in hazardous waste streams simplifies environmental permitting and reduces the liability associated with chemical storage and transport. For multinational corporations with strict ESG (Environmental, Social, and Governance) mandates, partnering with suppliers who utilize such green chemistry technologies enhances their own sustainability reporting and brand reputation.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled answers to common questions regarding the feasibility and scope of this electrochemical transformation. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's potential in your specific application. Whether you are concerned about substrate compatibility, purification requirements, or regulatory status, the following information addresses the key considerations for integrating this method into your supply chain. We encourage technical teams to review these details to assess the fit for their current development pipelines.
Q: What are the primary advantages of this electrochemical method over traditional Grignard routes?
A: Unlike traditional methods requiring hazardous Grignard reagents and multiple steps, this electrochemical approach operates in a single pot under mild conditions, eliminating the need for stoichiometric chemical oxidants and transition metal catalysts.
Q: Does this process support the synthesis of both five-membered and six-membered rings?
A: Yes, the methodology is highly versatile, successfully demonstrating the formation of both tetrahydropyrrole (five-membered) and piperidine (six-membered) scaffolds with excellent diastereoselectivity.
Q: How does the removal of metal catalysts impact downstream purification costs?
A: By avoiding transition metal catalysts, the process removes the necessity for expensive and time-consuming heavy metal scavenging steps, significantly simplifying the purification workflow and reducing overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl-3-Sulfonyl Tetrahydropyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in delivering high-quality pharmaceutical intermediates with superior efficiency. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to industrial reality. Our state-of-the-art facilities are equipped to handle electrochemical processes safely and effectively, adhering to stringent purity specifications and rigorous QC labs to guarantee product consistency. We understand that every molecule presents unique challenges, and our team of expert chemists is ready to optimize the reaction parameters outlined in CN116356354A to meet your specific yield and purity targets. By leveraging our infrastructure, you can mitigate the risks associated with process development and accelerate your time to market.
We invite you to engage with our technical procurement team to discuss how this innovative electrochemical route can be tailored to your specific needs. Contact us today to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener synthesis method. We are prepared to provide specific COA data and route feasibility assessments to support your decision-making process. Let us collaborate to build a more sustainable and efficient supply chain for your next-generation therapeutic agents.
