Advanced Electrochemical Catalytic Oxidative Coupling for Alpha-Disulfide Dicarboxylic Acid Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking sustainable and cost-effective methodologies for synthesizing critical building blocks, particularly those containing disulfide linkages which are ubiquitous in bioactive molecules and polymer additives. Patent CN111809195B introduces a groundbreaking electrochemical catalytic oxidative coupling synthesis method for alpha-disulfide dicarboxylic acid compounds, representing a significant leap forward in green chemistry manufacturing. This technology replaces traditional stoichiometric oxidants with electricity, utilizing a simple single-chamber electrolytic cell setup to drive the transformation of alpha-mercaptocarboxylic acids into valuable disulfide dimers. By leveraging constant current electrolysis with inexpensive graphite electrodes, this process not only mitigates the environmental burden associated with heavy metal waste or halogenated byproducts but also offers a streamlined pathway that is inherently safer and more energy-efficient. For R&D directors and procurement strategists, this patent signals a viable route to high-purity intermediates that aligns with modern regulatory demands for cleaner production processes while maintaining robust economic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-disulfide dicarboxylic acids has relied heavily on chemical oxidative coupling methods that suffer from significant inefficiencies and economic drawbacks. A prominent example found in prior art involves the use of molecular iodine as a stoichiometric oxidant in conjunction with sodium thiosulfate as a nucleophilic reagent, typically requiring reaction temperatures as high as 100°C in aqueous systems. This conventional approach presents multiple critical failure points for large-scale manufacturing: firstly, the reliance on elemental iodine drives up raw material costs substantially, as iodine prices are volatile and the reagent is consumed in equimolar quantities relative to the substrate. Secondly, the high-temperature conditions necessitate energy-intensive heating protocols and pose safety risks related to thermal runaway or solvent evaporation. Furthermore, the chemical complexity of using thiosulfate often leads to the formation of difficult-to-remove sulfur-containing byproducts, complicating the downstream purification process and ultimately depressing the overall isolated yield. These factors combined create a bottleneck for supply chain managers who require consistent, high-volume delivery of intermediates without the variability introduced by complex workup procedures.
The Novel Approach
In stark contrast, the electrochemical methodology disclosed in the patent data revolutionizes this transformation by substituting chemical potential with electrical potential, thereby eliminating the need for expensive stoichiometric oxidants. The novel approach utilizes a single-chamber electrolytic cell where alpha-mercaptocarboxylic acid serves as the substrate and a catalytic amount of sodium iodide acts as the electrolyte and redox mediator. Operating at a remarkably mild temperature of 29°C, this process drastically reduces energy consumption compared to the reflux conditions of traditional methods. The use of graphite sheets for both the anode and cathode ensures that electrode costs remain negligible, a crucial factor for cost reduction in fine chemical manufacturing. Moreover, the constant current electrolysis mode provides precise control over the reaction progress, stopping exactly when 1.0 F/mol of electricity is passed, which minimizes over-oxidation side reactions. This precision translates directly into higher purity profiles and simplified isolation steps, as the reaction mixture primarily contains the target disulfide and easily removable salts, bypassing the need for extensive chromatographic purification that plagues conventional iodine-based syntheses.

Mechanistic Insights into Electrochemical Catalytic Oxidative Coupling
The core of this technological advancement lies in the anodic oxidation mechanism facilitated by the iodide mediator within the electrochemical cell. Upon application of a constant current, iodide ions at the anode surface are oxidized to generate reactive iodine species in situ, which then interact with the thiol group of the alpha-mercaptocarboxylic acid substrate. This interaction promotes the formation of thiyl radicals or sulfenyl iodide intermediates, which rapidly undergo homocoupling to form the stable disulfide bond characteristic of the target alpha-disulfide dicarboxylic acid structure. The beauty of this electrocatalytic cycle is that the iodine species are regenerated at the electrode interface, allowing a sub-stoichiometric amount of sodium iodide (0.5 equivalents) to drive the conversion of the entire substrate load. This catalytic turnover is far more efficient than the single-use consumption of molecular iodine in traditional methods. Simultaneously, at the cathode, protons generated during the oxidation process are reduced to hydrogen gas, maintaining charge balance without introducing foreign reducing agents that could contaminate the product stream. This elegant electron-transfer mechanism ensures that the reaction proceeds with high atom economy, as the only byproducts are hydrogen gas and trace amounts of salt, leaving the organic backbone of the molecule intact and free from halogenation impurities.
From an impurity control perspective, the mild operating conditions of 29°C play a pivotal role in preserving the structural integrity of sensitive functional groups attached to the alpha-carbon. In traditional high-temperature oxidations, there is a persistent risk of over-oxidizing the sulfur moiety to sulfoxides or sulfones, or even degrading the carboxylic acid functionality through decarboxylation pathways. The electrochemical method avoids these thermal degradation pathways entirely by relying on selective electron transfer rather than thermal activation. Additionally, the use of a mixed solvent system comprising methanol and DME (dimethoxyethane) optimizes the solubility of both the organic substrate and the inorganic electrolyte, ensuring a homogeneous reaction environment that prevents localized hot spots or concentration gradients which often lead to side reactions. The result is a product profile with exceptional purity, where the primary impurities are unreacted starting materials that can be easily recycled, rather than complex structural analogs that are difficult to separate. This level of control is essential for producing high-purity pharmaceutical intermediates where strict impurity thresholds must be met for regulatory approval.
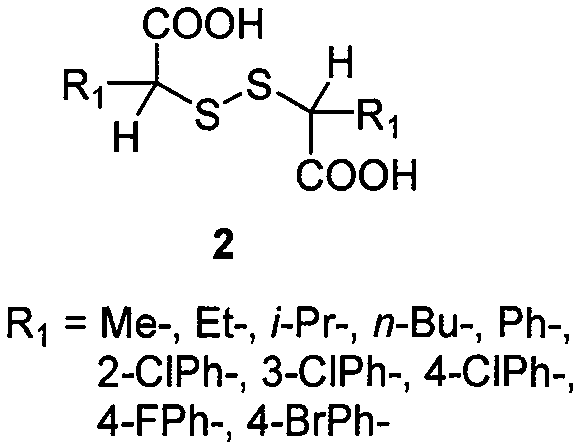
How to Synthesize Alpha-Disulfide Dicarboxylic Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process begins with the preparation of the electrolyte solution in a standard 50mL single-chamber cell, though this geometry is readily adaptable to larger flow cells or tank reactors for commercial production. The key to success lies in the precise control of current density, maintained at approximately 7.9 mA/cm², which ensures optimal reaction kinetics without inducing electrode passivation or excessive gas evolution that could disrupt the mixing. Following the electrolysis, the workup procedure is remarkably straightforward, involving solvent removal followed by a quench with sodium thiosulfate to reduce any residual active iodine species, ensuring the final product is stable and colorless. For detailed operational parameters and specific stoichiometric ratios tailored to different substrate scales, please refer to the standardized synthesis guide below.
- Prepare the electrolyte solution by dissolving alpha-mercaptocarboxylic acid and sodium iodide in a methanol: DME solvent system within a single-chamber electrolytic cell.
- Perform constant current electrolysis using graphite sheet electrodes at a current density of approximately 7.9 mA/cm² until 1.0 F/mol of electricity is passed.
- Execute the workup procedure by removing the solvent, treating with sodium thiosulfate solution, acidifying, extracting with anhydrous ether, and drying to isolate the target disulfide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical synthesis route offers compelling strategic advantages that extend beyond mere technical feasibility. The shift from chemical oxidants to electricity fundamentally alters the cost structure of manufacturing alpha-disulfide dicarboxylic acids, removing the dependency on volatile halogen markets and reducing the inventory burden of hazardous reagents. This transition supports a more resilient supply chain, as electricity is a universally available utility, whereas specialized chemical oxidants may face logistical bottlenecks or regional availability issues. Furthermore, the simplified downstream processing reduces the demand for extensive solvent usage and waste treatment infrastructure, aligning with corporate sustainability goals and reducing the total cost of ownership for the production facility. These factors collectively enhance the reliability of supply for critical intermediates used in drug development and agrochemical formulations.
- Cost Reduction in Manufacturing: The elimination of stoichiometric molecular iodine represents a direct and substantial saving in raw material costs, as iodine is a relatively expensive reagent compared to the catalytic amount of sodium iodide used in this electrochemical process. Additionally, the use of graphite electrodes, which are commodity items with low replacement costs, avoids the capital expenditure associated with specialized noble metal electrodes like platinum or gold often required in other electro-organic syntheses. The mild reaction temperature of 29°C further contributes to cost efficiency by eliminating the need for energy-intensive heating systems and cooling infrastructure required to manage exothermic chemical oxidations. Operational expenditures are also lowered due to the simplified workup procedure, which requires fewer extraction steps and less solvent volume, thereby reducing waste disposal fees and solvent recovery costs significantly.
- Enhanced Supply Chain Reliability: By relying on a continuous electrolysis process rather than batch chemical additions, the manufacturing timeline becomes more predictable and easier to automate, reducing the risk of human error and batch-to-batch variability. The raw materials required, specifically alpha-mercaptocarboxylic acids and sodium iodide, are commercially available in bulk quantities from multiple global suppliers, ensuring that production is not held hostage by single-source dependencies. The stability of the reaction conditions means that the process is less sensitive to minor fluctuations in ambient temperature or pressure, making it suitable for deployment in diverse geographic locations without extensive facility modifications. This robustness ensures a steady flow of high-purity intermediates to downstream customers, minimizing the risk of production delays that could impact the launch timelines of new pharmaceutical products.
- Scalability and Environmental Compliance: The constant current electrolysis method is inherently scalable, as increasing production capacity can be achieved by numbering up electrolytic cells or increasing the electrode surface area in flow reactors without altering the fundamental reaction chemistry. This modularity allows for flexible capacity adjustments to meet fluctuating market demand without the need for massive upfront investment in large-scale batch reactors. From an environmental standpoint, the process generates minimal hazardous waste, primarily consisting of aqueous salt solutions that are easier to treat than the organic halogenated waste streams produced by traditional iodine oxidation. The absence of heavy metal catalysts and the use of green solvent systems position this technology favorably for meeting increasingly stringent environmental regulations, facilitating smoother regulatory approvals for new drug applications that utilize these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These answers are derived directly from the experimental data and process descriptions provided in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is critical for evaluating the fit of this technology within your existing manufacturing portfolio and for assessing the potential return on investment for adopting this greener synthetic route.
Q: What are the primary advantages of this electrochemical method over traditional iodine oxidation?
A: The electrochemical method eliminates the need for stoichiometric amounts of expensive molecular iodine, operates at mild temperatures (29°C) compared to the traditional 100°C requirement, and significantly simplifies post-treatment by reducing byproduct formation.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of constant current electrolysis with inexpensive graphite electrodes makes the process highly suitable for industrial scale-up, offering better control and lower equipment costs compared to batch chemical oxidation methods.
Q: What is the substrate scope for this oxidative coupling reaction?
A: The method demonstrates broad substrate tolerance, successfully converting various alpha-mercaptocarboxylic acids including aliphatic chains (methyl, ethyl, butyl) and aromatic derivatives (phenyl, chlorophenyl, bromophenyl) into their corresponding disulfides with yields ranging from 69% to 95%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Disulfide Dicarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with advanced electrochemical reactors capable of maintaining the precise current densities and temperature controls required by patent CN111809195B, guaranteeing consistent product quality. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch of alpha-disulfide dicarboxylic acid meets the highest international standards for impurity profiles and physical properties.
We invite you to collaborate with us to leverage this innovative technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this electrochemical route for your specific application. Please contact our technical procurement team today to request specific COA data for our available disulfide intermediates and to discuss route feasibility assessments tailored to your unique supply chain requirements. Together, we can drive down costs and accelerate your time to market with sustainable, high-quality chemical solutions.
