Advanced 2-Step Synthesis of Gastrodin Hemihydrate for Commercial Scale-Up
Introduction to Next-Generation Gastrodin Synthesis
The pharmaceutical landscape for neuroprotective agents is constantly evolving, driven by the need for cleaner, more efficient manufacturing processes. Patent CN111018928A introduces a transformative approach to synthesizing gastrodin hemihydrate, a critical active pharmaceutical ingredient (API) intermediate widely used in treating neurological disorders. This technology addresses long-standing inefficiencies in the industry by replacing hazardous, multi-step protocols with a streamlined, two-step sequence that prioritizes both atomic economy and environmental safety. The core innovation lies in the direct glycosylation of 4-hydroxymethyl phenol using a specifically prepared bromo-sugar intermediate under phase transfer catalysis. This method not only simplifies the operational workflow but also ensures the production of high-purity gastrodin hemihydrate, meeting the stringent quality standards required by global regulatory bodies.
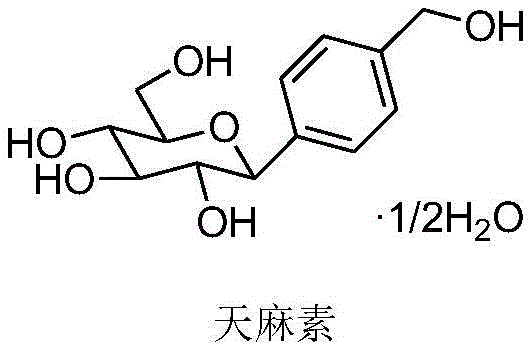
For R&D directors and procurement specialists, understanding the structural integrity and synthesis pathway of high-purity gastrodin hemihydrate is paramount. The molecule, chemically defined as 4-hydroxymethyl phenyl-β-D-glucopyranoside hemihydrate, possesses a specific stereochemistry that is crucial for its biological activity. Traditional methods often struggled to maintain this stereochemical purity while managing toxic byproducts. The disclosed patent overcomes these hurdles by utilizing a Koenigs-Knorr type modification that ensures the correct β-configuration is achieved with high selectivity. This technical breakthrough positions the technology as a cornerstone for any organization seeking a reliable gastrodin intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of gastrodin has been plagued by significant technical and environmental bottlenecks. Early synthetic routes, such as those described in literature from the 1980s, relied heavily on the use of red phosphorus and bromine. These reagents are not only highly toxic and corrosive but also generate substantial amounts of hazardous waste, creating a severe burden on three-waste treatment facilities. Furthermore, these legacy processes typically suffered from low total yields, often hovering around 24%, which translates to massive raw material wastage and inflated production costs. Subsequent improvements attempted to mitigate these issues by introducing catalytic hydrogenation using Raney Nickel. While this offered a marginal yield increase to approximately 31%, it introduced a new set of problems related to heavy metal contamination. The presence of nickel residues necessitates complex and costly purification steps to meet pharmaceutical grade specifications, and the disposal of spent nickel catalysts poses ongoing environmental compliance challenges.
More recent patents, such as CN102516329B and CN104072549B, have attempted to optimize the route through Lewis acid catalysis or multi-step protection-deprotection sequences. However, these methods often extend the synthetic timeline to five or more distinct steps. Each additional step in a chemical synthesis introduces a multiplicative loss in yield; consequently, even with moderate individual step yields, the overall process efficiency remains suboptimal, rarely exceeding 44%. Additionally, many of these routes still rely on harsh conditions or expensive reagents that hinder cost reduction in pharmaceutical intermediates manufacturing. The cumulative effect of these limitations is a supply chain that is fragile, expensive, and environmentally unsustainable, failing to meet the modern demands of green chemistry and efficient large-scale production.
The Novel Approach
In stark contrast to the convoluted pathways of the past, the method disclosed in CN111018928A offers a elegant solution by condensing the entire synthesis into just two robust steps. The first step involves the controlled bromination of β-D-glucose pentaacetate to form 2-bromo-α-D-glucose tetraacetate. This intermediate is then directly coupled with 4-hydroxymethyl phenol in a biphasic system. This novel approach eliminates the need for toxic red phosphorus and heavy metal catalysts entirely. By operating under mild alkaline conditions with a phase transfer catalyst, the reaction proceeds smoothly at temperatures between 15°C and 45°C. This mildness is a critical advantage, as it reduces energy consumption and minimizes the formation of thermal degradation byproducts. The result is a process that delivers a finished product with a purity exceeding 99.5% after simple recrystallization.
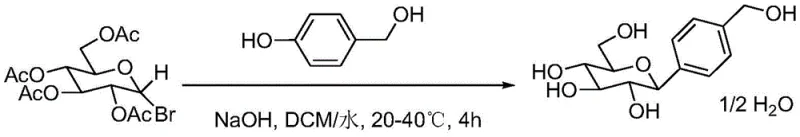
The strategic value of this novel approach extends beyond mere yield improvements. By shortening the synthetic route from five steps to two, the process dramatically reduces the accumulation of impurities that typically occur at each stage of a longer synthesis. This inherent purity simplifies downstream processing, removing the need for extensive chromatography or complex extraction protocols. For supply chain managers, this translates to a more predictable and reliable production schedule. The ability to source readily available starting materials like glucose pentaacetate and 4-hydroxymethyl phenol, combined with a simplified workflow, ensures that the commercial scale-up of complex glycosides becomes a viable and economically attractive proposition. This methodology represents a paradigm shift towards sustainable, high-efficiency API intermediate manufacturing.
Mechanistic Insights into Phase Transfer Catalyzed Glycosylation
The success of this synthesis hinges on the precise mechanistic interplay between the bromo-sugar donor and the phenolic acceptor under phase transfer conditions. The reaction utilizes tetrabutylammonium bromide (TBAB) as a crucial phase transfer catalyst. In the biphasic system consisting of an organic solvent (preferably dichloromethane) and an aqueous alkaline solution, the base (NaOH or KOH) deprotonates the 4-hydroxymethyl phenol to form the corresponding phenoxide anion in the aqueous phase. The quaternary ammonium cation of the TBAB facilitates the transport of this lipophilic phenoxide ion into the organic phase, where the 2-bromo-α-D-glucose tetraacetate resides. Once in the organic phase, the phenoxide acts as a potent nucleophile, attacking the anomeric carbon of the sugar moiety. This nucleophilic substitution proceeds via an SN2-like mechanism, resulting in the inversion of configuration from the α-bromide to the desired β-glycoside linkage. This stereoselective control is vital for ensuring the biological efficacy of the final gastrodin product.
Impurity control is another critical aspect managed by this mechanistic design. In traditional hydrogenation routes, side reactions such as over-reduction or incomplete deprotection can lead to difficult-to-remove impurities. In this phase transfer system, the primary byproducts are inorganic salts (sodium bromide) and the quaternary ammonium salt, which are easily separated during the aqueous workup. The use of mild temperatures (20-40°C) further suppresses elimination reactions that could lead to glycal formation. Moreover, the specific molar ratio of reactants—optimized at roughly 1:1.3 for the sugar bromide to phenol—ensures that the limiting reagent is fully consumed while minimizing the presence of unreacted phenol in the final matrix. This rigorous control over reaction parameters allows manufacturers to consistently achieve the high-purity gastrodin hemihydrate specifications required for downstream pharmaceutical formulation, thereby reducing the risk of batch rejection and ensuring supply continuity.
How to Synthesize Gastrodin Hemihydrate Efficiently
Implementing this synthesis requires careful attention to the preparation of the key intermediate, 2-bromo-α-D-glucose tetraacetate, followed by the optimized coupling reaction. The process begins with the bromination of glucose pentaacetate using hydrobromic acid in acetic acid, a reaction that must be kept cool to prevent degradation. Once the intermediate is isolated, it is immediately subjected to the glycosylation conditions described in the patent. The simplicity of the workup—involving basic washing, solvent evaporation, and recrystallization—makes this route particularly amenable to standard chemical manufacturing equipment. Detailed standard operating procedures regarding stoichiometry, temperature profiles, and purification techniques are essential for replicating the high yields reported in the patent data.
- Prepare 2-bromo-α-D-glucose tetraacetate by reacting β-D-glucose pentaacetate with hydrobromic acid in acetic acid within an organic solvent like dichloromethane at 15-25°C.
- React the resulting bromo-intermediate with 4-hydroxymethyl phenol in a biphasic system using sodium hydroxide and tetrabutylammonium bromide as a phase transfer catalyst.
- Isolate the crude product by solvent evaporation and purify via recrystallization using a dichloromethane and ethanol mixture to achieve >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers tangible strategic advantages that go beyond simple technical metrics. The primary benefit lies in the drastic simplification of the manufacturing workflow. By reducing the number of unit operations from five to two, the facility throughput is effectively doubled without the need for additional capital investment in reactors or separation columns. This efficiency gain directly correlates to a significant reduction in operational expenditures, including labor, energy, and solvent consumption. Furthermore, the elimination of heavy metal catalysts like Raney Nickel removes the logistical and financial burden associated with sourcing, handling, and disposing of hazardous materials. This not only lowers the direct cost of goods sold but also mitigates regulatory risks associated with heavy metal limits in final drug products.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the convergence of high yield and low input costs. Traditional methods suffer from compounding yield losses across multiple steps, whereas this two-step route maintains high efficiency throughout. The absence of expensive noble metal catalysts or specialized high-pressure hydrogenation equipment means that the barrier to entry for production is lower. Additionally, the solvents used, such as dichloromethane and ethanol, are commodity chemicals with stable pricing and wide availability. The qualitative reduction in waste generation also translates to lower waste disposal fees, contributing to a leaner and more cost-effective production model that enhances overall profit margins for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, commercially available starting materials. Glucose pentaacetate and 4-hydroxymethyl phenol are bulk chemicals produced by numerous suppliers globally, reducing the risk of single-source dependency. The mild reaction conditions (15-45°C) mean that the process is less susceptible to disruptions caused by utility failures or equipment limitations compared to high-temperature or high-pressure alternatives. This reliability ensures that production schedules can be met consistently, reducing lead time for high-purity pharmaceutical intermediates and allowing manufacturers to respond more agilely to market demand fluctuations. The simplified purification process further accelerates the release of batches, shortening the cash-to-cash cycle time.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior. The avoidance of red phosphorus and bromine gas eliminates the risk of acute toxicity incidents and the generation of corrosive waste streams. The aqueous workup generates salt waste that is far easier to treat than the heavy metal sludge produced by nickel-catalyzed routes. This alignment with green chemistry principles facilitates smoother regulatory approvals and permits for plant expansion. The process is inherently scalable; the exothermic nature of the reactions is manageable at large scales due to the moderate temperature range, allowing for safe operation in multi-ton reactors. This scalability ensures that the supply of gastrodin can grow in tandem with the increasing global demand for neuroprotective therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in patent CN111018928A. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios. The answers highlight the specific advantages in terms of purity, yield, and operational safety that distinguish this method from legacy technologies.
Q: How does this new synthesis method improve upon traditional gastrodin production?
A: Unlike traditional methods requiring 5 steps and toxic reagents like red phosphorus or heavy metal catalysts like Raney Nickel, this patented route achieves synthesis in only 2 steps with mild conditions, eliminating heavy metal residues and significantly boosting total yield.
Q: What represents the primary cost-saving driver in this manufacturing process?
A: The primary cost driver is the reduction in unit operations. By condensing the synthesis into two high-yield steps and removing the need for expensive hydrogenation equipment and heavy metal catalyst removal processes, operational expenditures are drastically lowered.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available starting materials and operates at mild temperatures (15-45°C) without requiring high-pressure hydrogenation, making it inherently safer and easier to scale from pilot plants to multi-ton commercial reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gastrodin Hemihydrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the CN111018928A patent and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant is seamless and efficient. Our state-of-the-art facilities are designed to handle the specific solvent systems and reaction conditions required for this glycosylation process, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch to ensure compliance with international pharmacopoeia standards.
We invite potential partners to engage with us to explore how this technology can optimize your supply chain. By leveraging our expertise, you can secure a stable supply of high-purity gastrodin hemihydrate while achieving substantial cost savings. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Let us provide you with specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the neuroprotective therapeutic sector.
