Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H) Ketone Derivatives for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of 3-Alkenyl Quinolin-2(1H) Ketone Derivatives for Pharmaceutical Applications
The landscape of nitrogen-containing heterocyclic synthesis is undergoing a significant transformation driven by the demand for more efficient and sustainable manufacturing processes for bioactive compounds. As detailed in the recent intellectual property disclosure CN114478375A, a novel preparation method for 3-alkenyl quinolin-2(1H) ketone derivatives has been established, addressing critical bottlenecks in the production of these vital pharmaceutical scaffolds. Quinolin-2(1H)-ones are ubiquitous structural motifs found in a vast array of natural products and therapeutic agents, exhibiting potent biological activities ranging from antibiotic and antitumor properties to antiplatelet and endothelin receptor antagonism. The ability to access these complex cores efficiently is paramount for drug discovery and development pipelines. This new methodology leverages a sophisticated palladium-catalyzed reductive aminocarbonylation strategy, utilizing o-nitrobenzaldehyde and allyl aryl ethers as primary building blocks. By integrating molybdenum hexacarbonyl as a safe solid carbon monoxide surrogate, the process circumvents the hazards associated with high-pressure CO gas, thereby enhancing operational safety profiles for industrial applications. 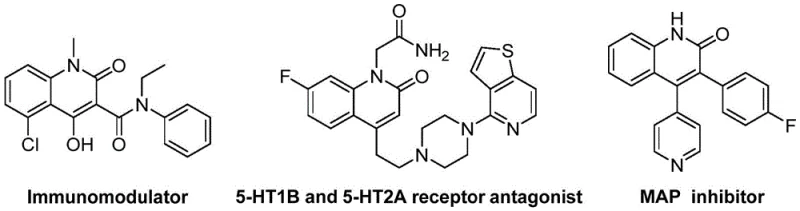
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinolinone frameworks via transition metal-catalyzed carbonylation has relied heavily on the use of highly reactive and often hazardous electrophiles. Traditional protocols frequently employ allyl chlorides, acetates, carbonates, or phosphates as the alkene source, which present significant challenges in terms of storage stability, toxicity, and waste generation. Furthermore, many existing carbonylation reactions require stringent conditions, such as high-pressure carbon monoxide atmospheres, which necessitate specialized autoclave equipment and rigorous safety monitoring, thereby inflating capital expenditure and operational complexity. The substrate scope in these legacy methods is often narrow, struggling to accommodate diverse functional groups without compromising yield or selectivity. Additionally, the reliance on separate nitrogen sources and external carbonyl donors increases the step count and reduces the overall atom economy of the synthesis. These factors collectively contribute to higher production costs and longer lead times, creating friction for procurement managers seeking reliable suppliers of high-purity pharmaceutical intermediates who must balance quality with economic feasibility.
The Novel Approach
The innovative strategy outlined in patent CN114478375A represents a paradigm shift by employing allyl aryl ethers as the electrophilic partner in the carbonylation reaction. Unlike their halide or ester counterparts, allyl aryl ethers are characterized by their natural abundance, low toxicity, and ease of handling, making them ideal candidates for green chemistry initiatives. This method uniquely utilizes o-nitrobenzaldehyde as a dual-function reagent, serving simultaneously as the nitrogen source for the heterocycle and the formyl source for the carbonyl group, effectively consolidating two synthetic steps into one seamless transformation. The reaction proceeds under relatively mild thermal conditions at 100°C in acetonitrile, utilizing a robust catalytic system composed of palladium acetate, tris(3-methoxyphenyl)phosphine, and molybdenum hexacarbonyl. This approach not only simplifies the operational workflow but also dramatically expands the functional group tolerance, allowing for the synthesis of a wide variety of 3-alkenyl quinolin-2(1H) ketone derivatives with excellent yields. 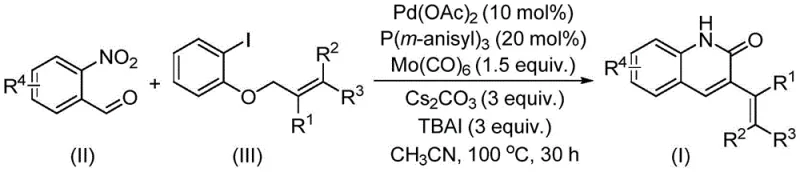
Mechanistic Insights into Palladium-Catalyzed Reductive Aminocarbonylation
The core of this technological advancement lies in the intricate interplay between the palladium catalyst and the molybdenum carbonyl source within the reaction matrix. The mechanism initiates with the oxidative addition of the palladium species to the allyl aryl ether, facilitated by the electron-rich tris(3-methoxyphenyl)phosphine ligand which stabilizes the active catalytic center. Simultaneously, molybdenum hexacarbonyl acts as a controlled release agent for carbon monoxide, generating the necessary CO in situ under the reaction temperature of 100°C, thus avoiding the logistical nightmares of handling gaseous CO. The o-nitrobenzaldehyde undergoes a reductive cyclization process where the nitro group is reduced, likely mediated by the metal-carbonyl species, to form the amine functionality that subsequently attacks the activated carbonyl-palladium intermediate. This cascade results in the formation of the quinolinone ring system with high regioselectivity. The presence of additives such as tetrabutylammonium iodide (TBAI) and cesium carbonate plays a crucial role in modulating the electronic environment and facilitating the leaving group departure, ensuring high conversion rates. Understanding these mechanistic nuances allows R&D directors to appreciate the robustness of the impurity profile, as the specific ligand and additive combination minimizes side reactions such as homocoupling or incomplete cyclization.
From an impurity control perspective, the choice of o-nitrobenzaldehyde as a dual source is particularly advantageous. By integrating the nitrogen and carbonyl atoms from a single molecule, the potential for mismatched coupling or the formation of byproducts derived from extraneous reagents is significantly reduced. The reaction conditions, specifically the use of acetonitrile as a polar aprotic solvent, ensure good solubility of the organic substrates while maintaining the stability of the ionic additives like cesium carbonate. The moderate temperature of 100°C strikes a balance between providing sufficient activation energy for the challenging C-N and C-C bond formations and preventing thermal degradation of sensitive functional groups on the allyl aryl ether substrate. This precise control over reaction parameters translates directly into a cleaner crude reaction mixture, simplifying downstream purification processes such as column chromatography and reducing the burden on quality control laboratories to identify and quantify trace impurities in the final active pharmaceutical ingredient (API) intermediate.
How to Synthesize 3-Alkenyl Quinolin-2(1H) Ketone Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to a precise sequence of reagent addition and thermal management to maximize yield and reproducibility. The protocol dictates the combination of specific molar ratios of catalysts and additives to ensure the catalytic cycle turns over efficiently without premature deactivation. Detailed below is the standardized procedure derived from the patent examples, which serves as a foundational guideline for process chemists aiming to replicate these high-efficiency results. It is imperative to maintain an inert atmosphere during the setup to protect the sensitive palladium catalyst from oxidation, although the system demonstrates remarkable tolerance once the cycle is initiated. 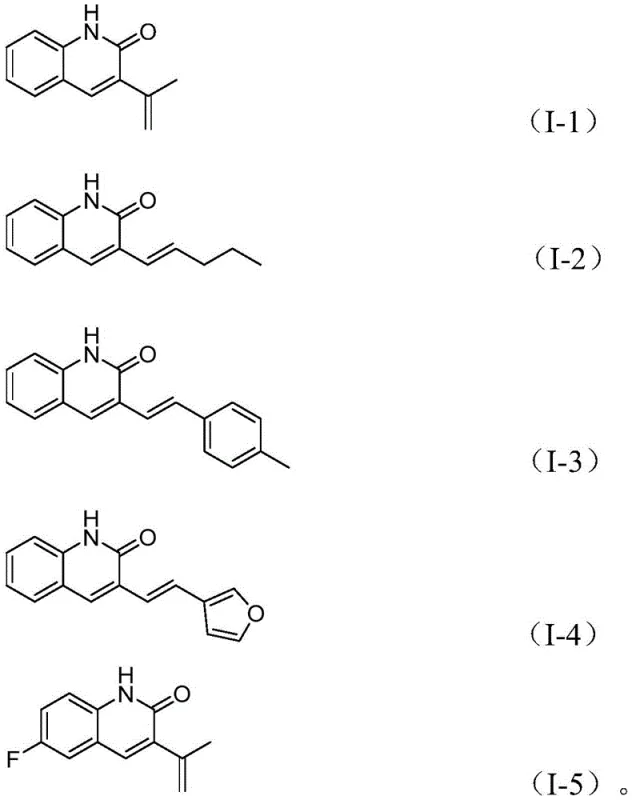
- Combine palladium acetate, tris(3-methoxyphenyl)phosphine, molybdenum hexacarbonyl, cesium carbonate, and tetrabutylammonium iodide in a sealed tube.
- Add o-nitrobenzaldehyde and the specific allyl aryl ether substrate along with acetonitrile solvent to the reaction mixture.
- Heat the mixture at 100°C for 30 hours, then filter and purify via silica gel column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the substantial reduction of raw material costs and logistical complexity. By shifting from hazardous allyl halides to stable, commercially abundant allyl aryl ethers, companies can mitigate risks associated with the storage and transport of volatile reagents. Furthermore, the elimination of high-pressure carbon monoxide cylinders removes a significant safety liability and reduces the need for specialized infrastructure, leading to lower overhead costs in manufacturing facilities. The simplicity of the post-treatment process, which involves basic filtration and standard silica gel chromatography, ensures that production throughput can be maintained at high levels without bottlenecks in the purification stage. This streamlined workflow directly contributes to enhanced supply chain reliability, as the dependency on niche or hard-to-source reagents is minimized, allowing for more consistent inventory planning and shorter lead times for delivering high-purity pharmaceutical intermediates to downstream clients.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive and readily available starting materials such as o-nitrobenzaldehyde and various substituted allyl aryl ethers. The catalytic system, while utilizing palladium, operates with high efficiency, meaning the loading of this precious metal can be optimized to minimize expense without sacrificing yield. Moreover, the avoidance of exotic ligands or complex multi-step sequences drastically lowers the overall cost of goods sold (COGS). The qualitative improvement in atom economy, achieved by using a single reagent for both nitrogen and carbonyl insertion, means less waste is generated per kilogram of product, which in turn reduces waste disposal costs and environmental compliance fees. This holistic approach to cost optimization makes the technology highly attractive for large-scale commercial production where margin preservation is critical.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the broad availability of the requisite substrates. Allyl aryl ethers and nitrobenzaldehydes are commodity chemicals produced by numerous global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of functional groups including halogens, methoxy groups, and trifluoromethyl substituents, means that the same manufacturing line can be easily adapted to produce a diverse library of derivatives. This flexibility allows manufacturers to respond rapidly to changing market demands or specific client requests for custom analogs without requiring extensive process re-validation. Consequently, the lead time for high-purity pharmaceutical intermediates can be consistently met, fostering stronger partnerships with international pharmaceutical companies that prioritize dependable delivery schedules.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns perfectly with modern green chemistry principles. The use of solid molybdenum hexacarbonyl as a CO source eliminates the release of toxic gases, improving the workplace safety environment and simplifying regulatory compliance regarding emissions. The reaction generates minimal hazardous byproducts, and the solvent system (acetonitrile) is well-established for recovery and recycling in industrial settings. The straightforward workup procedure reduces the consumption of water and energy typically associated with complex extraction and washing protocols. As regulatory bodies worldwide tighten restrictions on pharmaceutical manufacturing waste, adopting such a clean and efficient process future-proofs the production facility against evolving environmental standards, ensuring long-term operational continuity and sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical decision-makers evaluating this technology for integration into their existing portfolios. Understanding these specifics is crucial for assessing the feasibility of scaling this route for commercial API intermediate production.
Q: What are the key advantages of using allyl aryl ethers over traditional allyl halides in this synthesis?
A: Allyl aryl ethers offer superior operational safety and stability compared to reactive allyl halides or carbonates. They are naturally occurring, low-toxicity electrophiles that simplify handling protocols while maintaining high reaction efficiency in the palladium-catalyzed system described in patent CN114478375A.
Q: How does the dual role of o-nitrobenzaldehyde impact the synthetic route complexity?
A: Utilizing o-nitrobenzaldehyde as both the nitrogen source and the formyl (carbonyl) source significantly streamlines the synthetic pathway. This atom-economical approach eliminates the need for separate nitrogen donors and external carbon monoxide gas, reducing reagent costs and simplifying the overall process workflow for manufacturing teams.
Q: Is this catalytic system suitable for large-scale commercial production of quinolinone intermediates?
A: Yes, the protocol utilizes commercially available catalysts like palladium acetate and robust conditions (100°C in acetonitrile) that are amenable to scale-up. The broad substrate tolerance allows for the production of diverse derivatives without requiring extensive re-optimization, supporting reliable supply chain continuity for API manufacturers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkenyl Quinolin-2(1H) Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN114478375A for accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of batch size. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with the utmost precision, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee that every batch of 3-alkenyl quinolin-2(1H) ketone derivative meets the highest industry standards. We are committed to bridging the gap between innovative academic research and practical industrial application, providing a seamless transition from gram-scale discovery to multi-ton manufacturing.
We invite forward-thinking pharmaceutical and agrochemical companies to collaborate with us to leverage this efficient synthetic route for their specific project needs. By partnering with our technical team, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you optimize your supply chain and reduce your time-to-market with our reliable 3-alkenyl quinolin-2(1H) ketone supplier capabilities.
