Scaling High-Density Fluorinated Energetic Materials for Advanced Propulsion Systems
Scaling High-Density Fluorinated Energetic Materials for Advanced Propulsion Systems
The development of next-generation energetic materials requires a delicate balance between high energy density, thermal stability, and safety during handling. Patent CN110218164B introduces a significant breakthrough in this domain with the synthesis of 1,3-bis(3,4,5-trifluoro-2,6-dinitrophenyl)urea, commercially designated as ZXC-19. This fluorinated compound represents a new class of insensitive high-energy materials designed to overcome the limitations of traditional explosives like HMX or RDX. The molecular architecture incorporates multiple fluorine atoms and nitro groups onto a urea backbone, resulting in a crystal density of 1.91 g/cm³ and a detonation velocity of 8.26 km/s. These physical properties make it an exceptionally viable candidate for solid rocket propellants where volumetric efficiency is paramount. Furthermore, the compound demonstrates a decomposition temperature of 207.9°C and an impact sensitivity of 24.5 J, indicating a robust safety profile for industrial processing and transportation.
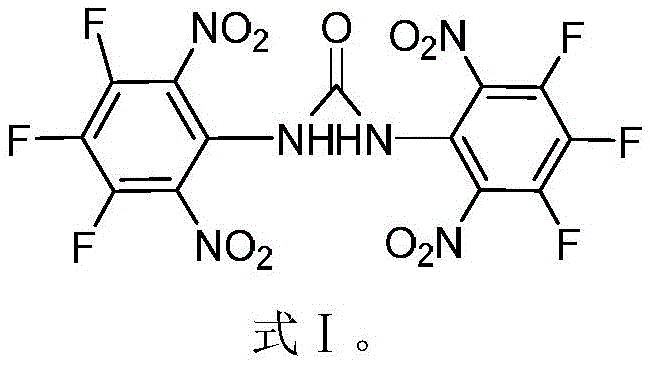
For R&D directors evaluating new candidates for propulsion systems, the structural integrity and synthetic accessibility of ZXC-19 are critical factors. The patent outlines a streamlined two-step synthesis starting from readily available 3,4,5-trifluoroaniline. This approach avoids the hazardous and complex fluorination steps often required in late-stage functionalization, thereby reducing the overall risk profile of the manufacturing process. The introduction of fluorine atoms not only enhances the oxidizing potential of the molecule but also facilitates the formation of intramolecular hydrogen bonds, which significantly improves the thermal stability of the composite material. As a reliable energetic material intermediates supplier, understanding these structural advantages allows for better formulation design in downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the search for high-energy insensitive explosives has been plagued by compounds that offer excellent performance but suffer from poor synthetic feasibility or dangerous sensitivity profiles. For instance, while compounds like HNFX exhibit high crystalline density and thermal stability, their synthesis often involves low-yielding steps and hazardous reagents that complicate scale-up. Traditional nitration methods for poly-nitro aromatic compounds frequently require extreme conditions that can lead to uncontrolled exotherms or the formation of unstable by-products. Additionally, many conventional fluorinated explosives are difficult to purify, requiring extensive chromatography or recrystallization processes that drive up costs and reduce overall throughput. The reliance on expensive transition metal catalysts or specialized fluorinating agents in older methodologies further exacerbates the cost reduction in energetic material manufacturing challenges, making them less attractive for large-scale defense or aerospace applications.
The Novel Approach
The methodology described in CN110218164B offers a transformative solution by utilizing a carbamate protection strategy followed by direct nitration. The process begins with the conversion of 3,4,5-trifluoroaniline into ethyl (3,4,5-trifluorophenyl)carbamate, a stable intermediate that directs subsequent substitution reactions effectively. This intermediate serves as a robust scaffold for the introduction of nitro groups without compromising the integrity of the fluorine substituents. By employing a mixed acid system of concentrated sulfuric acid and potassium nitrate or nitric acid, the reaction proceeds under relatively mild conditions, warming from an ice-water bath to room temperature. This controlled thermal profile minimizes the risk of runaway reactions, a common concern in the commercial scale-up of complex energetic materials. The simplicity of the workup, involving precipitation in ice water and filtration, ensures that the final product can be obtained with high purity and yield, streamlining the supply chain for high-purity energetic material intermediates.
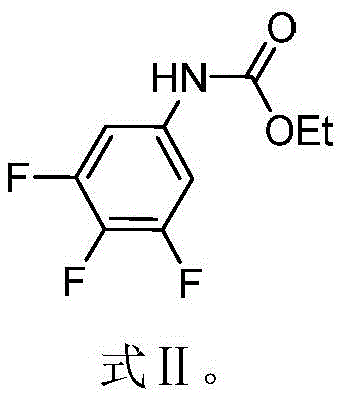
Mechanistic Insights into Nitration of Fluorinated Carbamates
The core chemical transformation in this synthesis is the electrophilic aromatic substitution of the fluorinated phenyl ring. The presence of three fluorine atoms on the benzene ring significantly alters the electron density distribution, making the ring less nucleophilic compared to non-fluorinated analogs. However, the urea linkage acts as an activating group, directing the incoming nitronium ions to the ortho positions relative to the nitrogen attachment. The use of concentrated sulfuric acid serves a dual purpose: it acts as a solvent to dissolve the organic intermediate and as a dehydrating agent to generate the active nitronium species from the nitrate source. The reaction mechanism likely proceeds through a sigma-complex intermediate, where the aromaticity is temporarily lost before the restoration of the aromatic system via proton loss. The steric hindrance provided by the adjacent fluorine atoms ensures high regioselectivity, preventing the formation of unwanted isomers that could compromise the energetic performance of the final material.
Impurity control is another critical aspect of this mechanistic pathway. The patent highlights the use of Thin Layer Chromatography (TLC) to monitor the consumption of the starting carbamate, ensuring that the reaction is driven to completion before quenching. This real-time monitoring prevents the accumulation of partially nitrated species which could act as sensitizers in the final product. Furthermore, the low solubility of ZXC-19 in water but high solubility in most organic solvents facilitates an efficient purification strategy. The product precipitates cleanly upon dilution with ice water, leaving behind soluble inorganic salts and acidic by-products in the aqueous phase. This inherent difference in solubility parameters simplifies the isolation process, reducing the need for energy-intensive distillation or complex extraction protocols, which is a key consideration for reducing lead time for high-purity energetic material intermediates in a commercial setting.
How to Synthesize 1,3-bis(3,4,5-trifluoro-2,6-dinitrophenyl)urea Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production. The process is characterized by its operational simplicity, requiring standard glassware and magnetic stirring equipment rather than specialized high-pressure reactors. The initial preparation of the nitrating mixture must be conducted under strict temperature control to manage the exotherm associated with mixing strong acids and salts. Once the intermediate is added, the gradual warming to room temperature allows the kinetic energy to overcome the activation barrier for nitration without triggering decomposition. This balance of thermodynamics and kinetics is essential for achieving the reported yields of nearly 90%. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Prepare the nitrating mixture by slowly adding potassium nitrate or nitric acid into concentrated sulfuric acid (95-98%) under ice-water bath cooling with magnetic stirring.
- Introduce the intermediate ethyl (3,4,5-trifluorophenyl)carbamate into the acid mixture while maintaining low temperature, then allow the system to warm to room temperature for reaction completion.
- Quench the reaction by pouring into vigorously stirred ice water, filter the precipitated solid, wash thoroughly with water, and dry to obtain the pure energetic material.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift towards this specific synthetic route offers substantial strategic benefits. The reliance on commodity chemicals such as sulfuric acid, potassium nitrate, and trifluoroaniline ensures a stable and diversified supply base, mitigating the risks associated with single-source specialty reagents. The elimination of precious metal catalysts removes a significant cost driver and simplifies the regulatory compliance landscape regarding heavy metal residues in the final product. This aligns perfectly with the global trend towards greener chemistry in the defense and aerospace sectors. Moreover, the high yield and straightforward purification process translate directly into improved material throughput, allowing manufacturers to meet demanding delivery schedules without compromising on quality standards.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive fluorinating agents or transition metal catalysts, which are often cost-prohibitive at scale. By utilizing a direct nitration strategy on a pre-fluorinated scaffold, the process significantly reduces raw material expenses. The simplified workup procedure, which relies on precipitation and filtration rather than column chromatography, drastically lowers solvent consumption and waste disposal costs. These efficiencies compound over large production runs, resulting in a highly competitive cost structure for the final energetic material.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis are widely available in the global chemical market, reducing the vulnerability of the supply chain to geopolitical disruptions or logistical bottlenecks. The robustness of the reaction conditions means that production can be maintained consistently across different facilities without the need for highly specialized equipment or operator expertise. This flexibility ensures continuous availability of critical intermediates, supporting just-in-time manufacturing models for downstream propellant formulators.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from gram quantities to multi-kilogram batches with consistent results, indicating strong potential for ton-scale production. The aqueous workup generates waste streams that are primarily acidic and saline, which are easier to treat and neutralize compared to organic-heavy waste from alternative synthetic routes. This environmental compatibility facilitates easier permitting and adherence to increasingly stringent industrial safety and environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of ZXC-19. These insights are derived directly from the experimental data and performance metrics provided in the patent literature. Understanding these details is crucial for stakeholders evaluating the integration of this material into existing supply chains or formulation pipelines.
Q: What are the thermal stability characteristics of ZXC-19 compared to traditional explosives?
A: ZXC-19 exhibits a thermal decomposition temperature of 207.9°C, which is comparable to RDX (210.0°C) and demonstrates excellent thermal stability suitable for high-performance propulsion systems.
Q: Is the synthesis process for this fluorinated urea scalable for industrial production?
A: Yes, the patent describes a method using safe raw materials and simple equipment that has been successfully demonstrated from gram-scale to multi-kilogram batches with consistent yields around 87-89%.
Q: How does the density of ZXC-19 impact its performance as a propellant?
A: With a crystal density of approximately 1.91 g/cm³, ZXC-19 offers superior volumetric energy density compared to many conventional CHNO explosives, enhancing the efficiency of rocket propellant formulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-bis(3,4,5-trifluoro-2,6-dinitrophenyl)urea Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance energetic materials play in advancing aerospace and defense technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of ZXC-19 performs consistently in your final applications. Our infrastructure is designed to handle complex fluorinated chemistries safely and efficiently, providing a secure foundation for your long-term supply requirements.
We invite you to collaborate with us to optimize your material sourcing strategy. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and production planning. Let us be your partner in driving innovation through superior chemical manufacturing.
