Advanced Synthetic Route for N-butyl-2,2,6,6-tetramethyl-4-piperidylamine: Scaling Safe Production
Advanced Synthetic Route for N-butyl-2,2,6,6-tetramethyl-4-piperidylamine: Scaling Safe Production
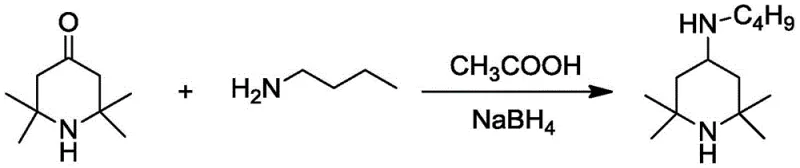
The chemical industry is constantly evolving towards safer, more efficient, and cost-effective manufacturing processes, particularly for critical intermediates used in polymer stabilization. A pivotal advancement in this domain is detailed in patent CN106631991B, which discloses a novel, simple synthesis method for N-butyl-2, 2,6, 6-tetramethyl-4-piperidylamine. This compound serves as a vital precursor for hindered amine light stabilizers (HALS), which are essential for enhancing the photo-thermal stability of polyolefins and other resin systems used in medical and food packaging. The patented technology represents a significant departure from legacy manufacturing protocols by substituting hazardous high-pressure hydrogenation with a mild, chemically driven reductive amination pathway. For R&D directors and procurement strategists, understanding this shift is crucial, as it not only mitigates safety risks associated with high-pressure reactors but also opens avenues for substantial cost optimization in the supply chain of polymer additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of N-butyl-2, 2,6, 6-tetramethyl-4-piperidylamine has relied heavily on catalytic hydrogenation techniques that impose severe operational constraints. Traditional methods, as referenced in prior art such as European patent EP 302020, typically require the condensation product of 2,2,6, 6-tetramethyl piperidone and N-butylamine to undergo hydrogenation in the presence of expensive platinum or platinum oxide catalysts. This process necessitates rigorous conditions, specifically temperatures ranging from 70°C to 80°C and pressures as high as 50 bar. The reliance on specialized high-pressure autoclaves and hydrogen gas introduces significant safety hazards and capital expenditure requirements for manufacturing facilities. Furthermore, the use of precious metal catalysts like platinum creates a dependency on volatile commodity markets, leading to unpredictable raw material costs and potential supply chain bottlenecks. The complexity of managing high-pressure systems also demands highly skilled labor and stringent maintenance protocols, which collectively inflate the operational expenditure (OPEX) of producing this key intermediate.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the innovative approach outlined in patent CN106631991B utilizes a chemical reducing agent reductive amination method that operates under remarkably mild conditions. By employing acetic acid as a catalyst and a metal hydride, preferably sodium borohydride, the reaction proceeds efficiently at temperatures between 0°C and 60°C without the need for pressurized equipment. This fundamental shift eliminates the requirement for hydrogen gas and expensive platinum catalysts, thereby simplifying the reactor setup to standard glass-lined or stainless steel vessels capable of handling atmospheric pressure. The process involves dissolving the ketone and amine in common organic solvents such as methanol, ethanol, or dichloromethane, followed by the controlled addition of the reducing agent. This methodology not only enhances operator safety by removing high-pressure and high-temperature risks but also streamlines the workflow, making it accessible for a broader range of chemical manufacturers. The result is a robust, scalable process that maintains high purity standards while drastically reducing the technological barriers to entry for production.
Mechanistic Insights into Acetic Acid-Catalyzed Reductive Amination
The core of this technological breakthrough lies in the precise mechanistic execution of reductive amination using a weak acid catalyst and a hydride source. The reaction initiates with the nucleophilic attack of n-butylamine on the carbonyl carbon of 2,2,6, 6-tetramethyl piperidone, facilitated by the protonation of the carbonyl oxygen by acetic acid. This acid catalysis accelerates the formation of the imine or iminium ion intermediate, which is the critical electrophilic species required for the subsequent reduction step. Unlike strong mineral acids that might promote side reactions or degradation of the sensitive piperidine ring, acetic acid provides a buffered acidic environment that optimizes imine formation without compromising the structural integrity of the substrate. Once the imine intermediate is generated, the sodium borohydride acts as a source of hydride ions (H-), which selectively reduce the C=N double bond to a C-N single bond. This selectivity is paramount in ensuring that the ketone functionality is fully converted to the amine without affecting other parts of the molecule, thereby securing the high chemical purity required for downstream polymer applications.
Furthermore, the control of impurities in this synthetic route is inherently superior due to the mildness of the reagents and the absence of harsh thermal stress. In high-pressure hydrogenation, there is always a risk of over-reduction or hydrogenolysis of the C-N bond, which can lead to ring-opening byproducts or de-alkylation, complicating the purification process. The chemical reduction method described in the patent avoids these pitfalls by operating at lower temperatures (0-60°C), which kinetically favors the desired reduction pathway while suppressing thermal decomposition. The use of sodium borohydride also allows for precise stoichiometric control; by adding the hydride in batches, the exothermic nature of the reduction can be managed effectively, preventing local hot spots that could generate impurities. Post-reaction workup involving aqueous extraction and distillation further ensures that boron salts and unreacted starting materials are efficiently removed, yielding a final product with purity levels comparable to, or exceeding, those obtained via platinum catalysis. This mechanistic elegance translates directly into a cleaner crude product profile, reducing the burden on downstream purification units.
How to Synthesize N-butyl-2,2,6,6-tetramethyl-4-piperidylamine Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and addition sequences to maximize yield and safety. The patent specifies a molar ratio of 2,2,6, 6-tetramethyl piperidone to N-butylamine between 1.0:1.0 and 1.0:1.5, ensuring that the amine is present in slight excess to drive the equilibrium towards imine formation. Acetic acid is introduced at 5-20% molar amount relative to the ketone to provide sufficient catalytic activity without causing excessive acidity. The critical step involves the batch-wise addition of the metal hydride (sodium borohydride, potassium borohydride, or lithium borohydride) to control the reaction rate and heat evolution. Detailed standardized synthesis steps, including specific solvent choices like methanol or ethanol and precise temperature controls for each stage, are essential for reproducible results. For a comprehensive, step-by-step protocol optimized for laboratory and pilot-scale execution, please refer to the technical guide below.
- Dissolve 2,2,6,6-tetramethylpiperidone and n-butylamine in an organic solvent such as methanol or ethanol, then add acetic acid as a catalyst.
- Stir the mixture at a controlled temperature between 0°C and 60°C to facilitate imine formation before reduction.
- Add sodium borohydride in batches to the reaction system, maintain stirring for 5-12 hours, and proceed with aqueous workup and distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from high-pressure hydrogenation to this chemical reduction method offers transformative economic and logistical benefits. The most immediate impact is the elimination of capital-intensive infrastructure; facilities no longer need to invest in or maintain high-pressure autoclaves rated for 50 bar operations, which significantly lowers the barrier to scaling production. Additionally, the removal of platinum catalysts decouples the production cost from the fluctuating prices of precious metals, leading to a more stable and predictable cost structure. The use of commodity chemicals like sodium borohydride and acetic acid, which are widely available and inexpensive compared to specialized heterogeneous catalysts, further drives down the variable costs of manufacturing. This shift not only improves the margin profile for suppliers but also provides buyers with a more resilient supply source that is less susceptible to disruptions in the precious metals market or specialized equipment maintenance issues.
- Cost Reduction in Manufacturing: The economic advantages of this process are profound, primarily driven by the substitution of expensive catalytic systems with affordable chemical reagents. By avoiding the use of platinum or platinum oxide, manufacturers eliminate the high upfront cost of catalyst procurement and the recurring costs associated with catalyst recovery or disposal. Furthermore, the energy consumption is significantly reduced because the reaction operates at much lower temperatures (0-60°C) compared to the 70-80°C required for hydrogenation, and without the energy-intensive compression of hydrogen gas. The simplified equipment requirements mean that depreciation costs are lower, and the overall operational expenditure is streamlined. These factors combine to create a manufacturing process that is inherently more cost-efficient, allowing for competitive pricing strategies in the global market for polymer additives without sacrificing quality or yield.
- Enhanced Supply Chain Reliability: From a logistics and continuity perspective, this synthetic route offers superior reliability by utilizing widely available raw materials. Sodium borohydride, acetic acid, and common solvents like methanol and ethanol are bulk commodities with robust global supply chains, unlike specialized high-pressure hydrogenation catalysts which may have limited suppliers. The safety profile of the process also enhances supply chain resilience; without the need to transport or store large quantities of high-pressure hydrogen, the regulatory burden and insurance costs for the manufacturing site are reduced. This makes the facility less prone to shutdowns due to safety inspections or regulatory compliance issues related to hazardous gases. Consequently, buyers can expect more consistent delivery schedules and a lower risk of force majeure events disrupting the supply of this critical light stabilizer intermediate.
- Scalability and Environmental Compliance: The environmental and scalability footprint of this method aligns perfectly with modern green chemistry principles and increasing regulatory pressures. The mild reaction conditions generate fewer hazardous byproducts and eliminate the risk of high-pressure explosions, making the process safer for workers and the surrounding community. Waste management is simplified as the process avoids heavy metal contamination associated with platinum catalysts, reducing the complexity and cost of wastewater treatment and solid waste disposal. The scalability is excellent because the reaction kinetics are manageable in large vessels without the engineering challenges of heat transfer and pressure containment found in hydrogenation reactors. This ease of scale-up ensures that manufacturers can rapidly increase production capacity to meet surging demand from the polyolefin and packaging industries, ensuring a steady flow of high-purity intermediates to the market.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is vital for stakeholders evaluating its adoption or sourcing implications. The following questions address common concerns regarding safety, yield, and scalability, drawing directly from the experimental data and technical disclosures within the patent documentation. These insights are designed to clarify how this chemical reduction strategy outperforms traditional methods in both laboratory and industrial settings. For more specific technical inquiries or custom feasibility studies, our team is prepared to provide detailed analysis based on your specific production requirements.
Q: How does the new synthesis method improve safety compared to traditional platinum catalysis?
A: The novel method replaces high-pressure hydrogenation (50 bar) requiring expensive platinum catalysts and autoclaves with a mild chemical reduction using sodium borohydride at atmospheric pressure, drastically reducing operational risks.
Q: What yields can be expected from this reductive amination process?
A: According to patent data, the process achieves yields comparable to traditional methods, reaching up to 95.2% under optimized conditions with methanol solvent and sodium borohydride.
Q: Is this process scalable for industrial production of light stabilizer intermediates?
A: Yes, the elimination of high-pressure equipment and the use of common solvents like ethanol and methanol make the process highly scalable and suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-butyl-2,2,6,6-tetramethyl-4-piperidylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of final polymer products. As a leading CDMO and manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of N-butyl-2,2,6,6-tetramethyl-4-piperidylamine. Our facilities are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the demanding requirements of the hindered amine light stabilizer market. We are committed to leveraging advanced synthetic technologies, such as the platinum-free reductive amination process, to deliver products that offer superior value and performance.
We invite procurement leaders and R&D teams to collaborate with us to optimize their supply chains and reduce manufacturing costs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and quality standards. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us help you secure a sustainable and cost-effective source for your polymer additive intermediates, driving efficiency and innovation in your production processes.
