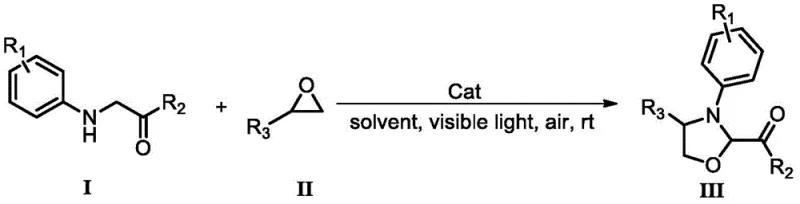
Scalable Visible Light Catalyzed Synthesis of Polysubstituted 1,3-Oxazolidine Intermediates for Commercial Production
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN112939883A, which discloses a novel preparation method for polysubstituted 1,3-oxazolidine compounds. These five-membered nitrogen heterocycles are not merely academic curiosities; they serve as critical skeletons in numerous bioactive molecules and natural products, such as Erchinines and GSK1265744, and function effectively as chiral auxiliaries in asymmetric catalysis. The patented technology introduces a visible light-catalyzed oxidative dehydrogenation [2+3] cyclization strategy that fundamentally shifts the paradigm from resource-intensive traditional methods to a greener, more economical approach. By leveraging cheap and readily available glycine derivatives and epoxy compounds as starting materials, and utilizing inexpensive catalysts like hydroiodic acid or N-bromosuccinimide, this process achieves high efficiency under remarkably mild conditions. For R&D directors and procurement specialists alike, this represents a tangible opportunity to optimize supply chains for high-purity pharmaceutical intermediates while drastically reducing the reliance on precious metals and harsh reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the polysubstituted 1,3-oxazolidine framework has been fraught with significant technical and economic challenges that hinder large-scale adoption. Conventional synthetic strategies often rely on the reaction of aminoalcohols with aldehydes or ketones, ring expansion reactions of strained three- or four-membered rings, or transition metal-catalyzed dipolar [3+2] cycloadditions. These traditional pathways frequently necessitate the use of expensive and potentially toxic transition metal catalysts, which introduce severe complications regarding residual metal removal—a critical quality attribute for any API intermediate intended for human consumption. Furthermore, many existing methods require harsh reaction conditions, including elevated temperatures or the use of stoichiometric amounts of strong oxidants, which not only increase energy consumption but also pose safety risks during scale-up. The complexity of starting materials in some reported methods further exacerbates cost issues, making the final product economically unviable for cost-sensitive applications in agrochemicals or generic pharmaceuticals. Consequently, there has been a persistent industry demand for a methodology that circumvents these limitations without compromising on yield or purity.
The Novel Approach
The innovative process described in the patent data offers a compelling solution by employing a visible light-catalyzed oxidative dehydrogenation-type [2+3] cyclization between glycine derivatives and epoxy compounds. This approach distinguishes itself by utilizing oxygen from the air as a green, inexhaustible oxidant, thereby eliminating the need for hazardous chemical oxidants. The reaction proceeds smoothly at room temperature under the irradiation of visible light, such as from a standard 26W energy-saving lamp or even sunlight, which dramatically lowers the energy footprint of the manufacturing process. Crucially, the system does not require an additional, expensive photocatalyst; instead, it utilizes cheap and easily obtained catalysts like hydroiodic acid, hydrobromic acid, or N-bromosuccinimide. This simplification of the catalytic system not only reduces raw material costs but also streamlines the downstream purification process. The versatility of this method is demonstrated by its ability to accommodate various substituents on the aromatic rings, allowing for the efficient preparation of a diverse series of polysubstituted 1,3-oxazolidine compounds with yields typically ranging from 70% to 80%.
Mechanistic Insights into Visible Light Catalyzed Oxidative Cyclization
The mechanistic underpinning of this transformation involves a sophisticated interplay between photo-excitation and radical chemistry, facilitated by the simple acid catalysts. Upon irradiation with visible light, the reaction mixture initiates a radical process where the glycine derivative and the epoxy compound undergo an oxidative coupling. The presence of air (oxygen) is pivotal, acting as the terminal oxidant to drive the dehydrogenation step necessary for the formation of the stable oxazolidine ring. Unlike traditional transition metal catalysis which often involves complex coordination spheres and ligand exchanges, this metal-free protocol likely proceeds through a radical cation or hydrogen atom transfer mechanism mediated by the halide catalyst and light energy. This pathway avoids the formation of stable metal-substrate complexes that can lead to difficult-to-remove impurities. For the R&D team, understanding this mechanism is vital for troubleshooting and optimization, as it highlights the importance of light intensity and oxygen availability as key process parameters. The absence of heavy metals ensures that the impurity profile is significantly cleaner, primarily consisting of organic by-products that are easier to separate via standard chromatographic techniques compared to trace metal contaminants.
Furthermore, the mildness of the reaction conditions plays a crucial role in impurity control and product stability. Many functional groups that are sensitive to strong acids, bases, or high temperatures remain intact under these room temperature, visible light conditions. This chemoselectivity is particularly advantageous when synthesizing complex intermediates containing multiple reactive sites, as it minimizes side reactions such as polymerization of the epoxide or degradation of the glycine ester moiety. The use of simple solvents like dichloromethane or toluene further enhances the practicality of the process, allowing for easy solvent recovery and recycling. From a quality control perspective, the consistency of the reaction across different substrates—whether electron-rich or electron-deficient aryl groups are present—suggests a robust and forgiving process window. This reliability is essential for maintaining stringent purity specifications required by regulatory bodies for pharmaceutical ingredients, ensuring that the final polysubstituted 1,3-oxazolidine products meet the rigorous standards expected of a reliable pharmaceutical intermediate supplier.
How to Synthesize Polysubstituted 1,3-Oxazolidine Efficiently
The operational simplicity of this synthesis makes it highly attractive for both laboratory discovery and pilot plant operations. The general procedure involves dissolving the glycine derivative and the epoxy compound in a suitable solvent, followed by the addition of the catalyst. The mixture is then stirred at room temperature under visible light irradiation for a period of 10 to 20 hours. After the reaction is complete, the solvent is removed under reduced pressure, and the crude product is purified by column chromatography. This straightforward workflow minimizes the need for specialized equipment, making it accessible for facilities without extensive high-pressure or cryogenic capabilities. The detailed standardized synthesis steps, including specific molar ratios and workup procedures optimized for maximum yield, are outlined below to guide your technical team in replicating this efficient process.
- Dissolve the glycine derivative and epoxy compound in a suitable solvent such as dichloromethane or 1,2-dichloroethane within a reaction flask.
- Add a catalytic amount of hydroiodic acid, hydrobromic acid, or N-bromosuccinimide to the reaction mixture.
- Stir the mixture at room temperature for 10-20 hours under irradiation from a visible light source (e.g., 26W energy-saving lamp or sunlight) in the presence of air.
- Remove the solvent under reduced pressure and purify the crude product via column chromatography to obtain the target polysubstituted 1,3-oxazolidine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light-catalyzed technology translates into direct and substantial strategic benefits. The shift away from expensive transition metals and complex photocatalysts to commodity chemicals like hydrobromic acid represents a fundamental restructuring of the cost base for these intermediates. Moreover, the ability to run reactions at room temperature using ambient air as an oxidant removes the need for energy-intensive heating or cooling systems and specialized gas handling infrastructure. This simplification of the process parameters directly correlates to lower capital expenditure (CAPEX) for new production lines and reduced operational expenditure (OPEX) for existing ones. The robustness of the reaction also implies fewer batch failures and higher overall equipment effectiveness (OEE), ensuring a more consistent and reliable supply of critical intermediates to downstream customers.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and additional photocatalysts results in a drastic reduction in raw material costs. Traditional methods often require palladium, copper, or specialized organic photocatalysts that are not only expensive to purchase but also costly to dispose of or recover. By replacing these with cheap mineral acids or N-bromosuccinimide, the direct material cost per kilogram of product is significantly lowered. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and labor hours, further enhancing the overall cost efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade starting materials such as glycine derivatives and styrene oxides ensures a stable and resilient supply chain. Unlike specialized reagents that may be subject to geopolitical supply disruptions or long lead times, the precursors for this reaction are produced globally in large volumes. This abundance mitigates the risk of production stoppages due to raw material shortages. Furthermore, the mild reaction conditions reduce the wear and tear on reactor vessels and associated equipment, leading to lower maintenance requirements and higher uptime for production facilities, thereby guaranteeing consistent delivery schedules to clients.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with increasingly stringent environmental regulations. Using air as an oxidant and avoiding heavy metals minimizes the generation of hazardous waste, simplifying wastewater treatment and disposal protocols. The room temperature operation reduces the carbon footprint associated with energy consumption for heating or cooling. These factors make the process highly scalable from gram to tonne quantities without encountering the safety and environmental hurdles typical of exothermic or high-pressure reactions. This scalability ensures that the supply can grow in tandem with market demand, supporting the commercial scale-up of complex pharmaceutical intermediates without regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light-catalyzed synthesis. These insights are derived directly from the patent specifications and are designed to clarify the feasibility and advantages of adopting this technology for your specific production needs. Understanding these details is crucial for making informed decisions about process integration and supplier qualification.
Q: What are the primary advantages of this visible light catalyzed method over traditional transition metal catalysis?
A: This method eliminates the need for expensive transition metal catalysts and additional photocatalysts, utilizing cheap acids like HBr or HI instead. It operates under mild room temperature conditions using air as a green oxidant, significantly reducing operational costs and environmental impact compared to harsh traditional methods.
Q: What is the typical yield range for this oxidative dehydrogenation [2+3] cyclization reaction?
A: According to the patent data, the reaction efficiently prepares a series of polysubstituted 1,3-oxazolidine compounds with yields ranging typically between 70% and 80%, demonstrating high efficiency for industrial application.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process is highly suitable for scale-up due to its simple one-step conversion, use of commercially available raw materials, and mild reaction conditions that do not require specialized high-pressure or high-temperature equipment, facilitating easier pilot and commercial scale amplification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 1,3-Oxazolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the visible light-catalyzed synthesis described in patent CN112939883A for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle photochemical reactions safely and efficiently, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to delivering polysubstituted 1,3-oxazolidine compounds that empower your drug discovery and development programs with superior quality and consistency.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free route can optimize your budget. Please contact us today to request specific COA data for our available inventory or to discuss route feasibility assessments for your custom synthesis needs. Let us help you secure a sustainable and cost-effective supply chain for your critical pharmaceutical intermediates.
